Genetic Stability Assessment of In Vitro Plantlets of Sang Mon Bamboo (Dendrocalamus sp.) Derived from Axillary Bud Proliferation Using RAPD and SCoT Markers
DOI:
https://doi.org/10.48048/tis.2025.9961Keywords:
Somaclonal variations, Genetic fidelity, Molecular markers, Bamboo, In vitro multiplication, Plant regeneration, Start Codon Target Polymorphism, Random Amplified Polymorphic DNA, Bamboo, Genetic fidelity, In vitro multiplication, Molecular markers, Plant regeneration, Random Amplified Polymorphic DNA, Somaclonal variations, Start Codon Target PolymorphismAbstract
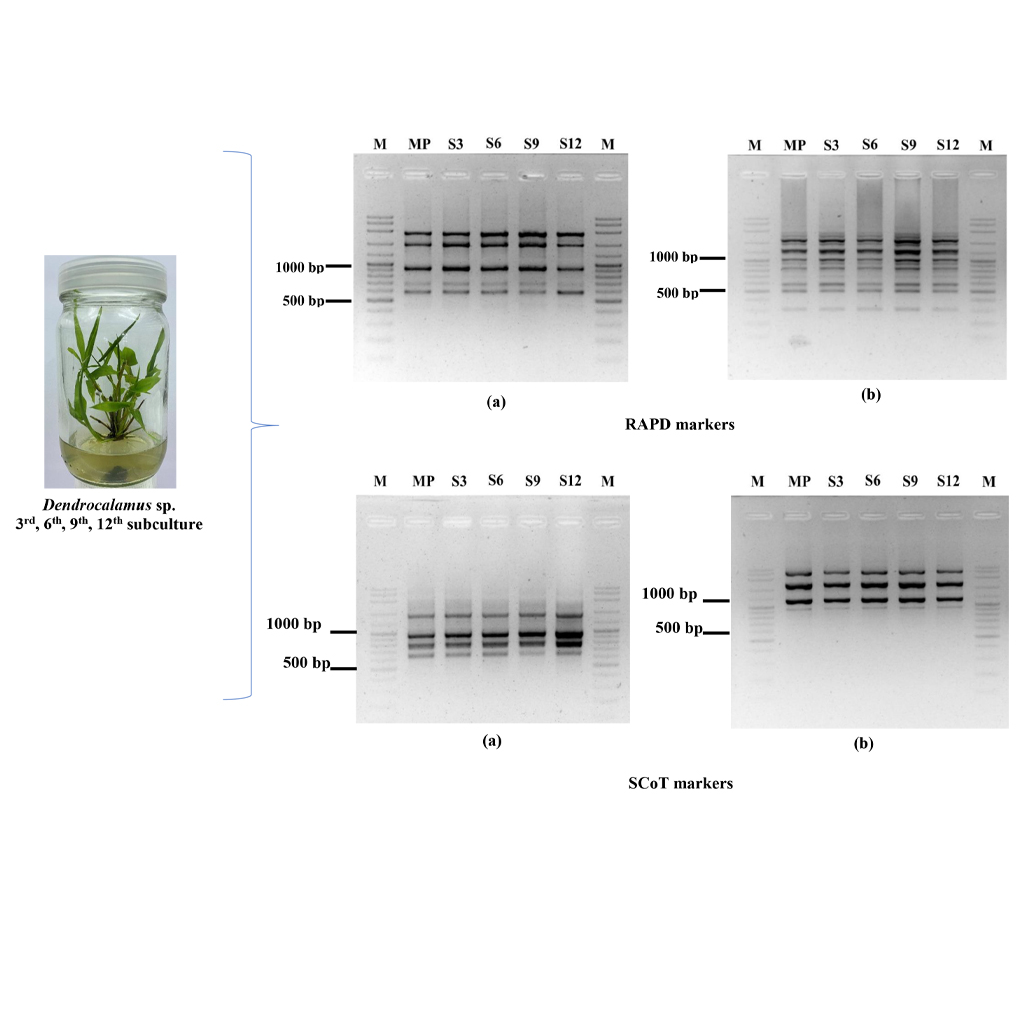
Sang Mon bamboo (Dendrocalamus sp.) is an important bamboo species with significant potential for various applications due to its straight, thick, and strong culm. Its ability to thrive in diverse environments has led to high market demand. Plant tissue culture technology provides an efficient method for the large-scale propagation of various bamboo species. However, the possibility of soma-clonal variation must be considered. The objectives of this research were to investigate the effects of various plant growth regulators, including TDZ, BA, and NAA, at different concentrations on shoot multiplication in Sang Mon bamboo for large-scale propagation. Additionally, the genetic stability of in vitro-raised plantlets was assessed using Random Amplified Polymorphic DNA (RAPD) and Start Codon Targeted (SCoT) markers. Genomic DNA was extracted from both the mother plant and in vitro-raised plants. The results revealed that the highest number of shoots (143.8) was obtained during the 12th subculture on Murashige and Skoog (MS) medium set 3, containing 0.05 mg/L TDZ combined with 0.5 or 1.0 mg/L BA and 0.05 mg/L NAA. Additionally, shoot clusters exhibited the longest shoot length (4.39 cm) and were stronger and more consistent across all medium formulations and subculture cycles. Eleven RAPD primers amplified 90 DNA fragments ranging from 300 to 3,000 bp, all of which were monomorphic across the 3rd, 6th, 9th, and 12th subcultures. The number of monomorphic bands per primer ranged from 7 to 12, with an average of 8.18 bands. Similarly, 11 SCoT primers generated 77 monomorphic DNA fragments ranging from 350 to 3,000 bp, with the number of bands per primer ranging from 4 to 10, averaging 7 bands. This study successfully established an efficient protocol for the large-scale propagation of Sang Mon bamboo through in vitro culture. The genetic stability of in vitro cultures was confirmed, indicating the absence of soma-clonal variation in plantlets derived from axillary bud proliferation.
HIGHLIGHTS
- The combination of TDZ in combination with BA and NAA resulted in efficient shoot multiplication of Sang Mon bamboo (Dendrocalamus ).
- Somaclonal variations are typically caused by mutations that arise during the tissue culture process, potentially affecting the genetic stability of plantlets.
- Genetic stability in micropropagated Sang Mon bamboo (Dendrocalamus) was assessed using RAPD and SCoT markers, revealing no variability among plantlets after the 3rd, 6th, 9th, and 12th subcultures, as well as when compared to the mother plant.
- Molecular markers, such as RAPD and SCoT, have proven effective in confirming the genetic stability of in vitro plantlets through axillary bud proliferation, ensuring the production of true-to-type plants.
- Tissue culture methods for bamboo propagation have been shown to be both reliable and sustainable, with strong evidence supporting their use for large-scale, consistent bamboo production.
GRAPHICAL ABSTRACT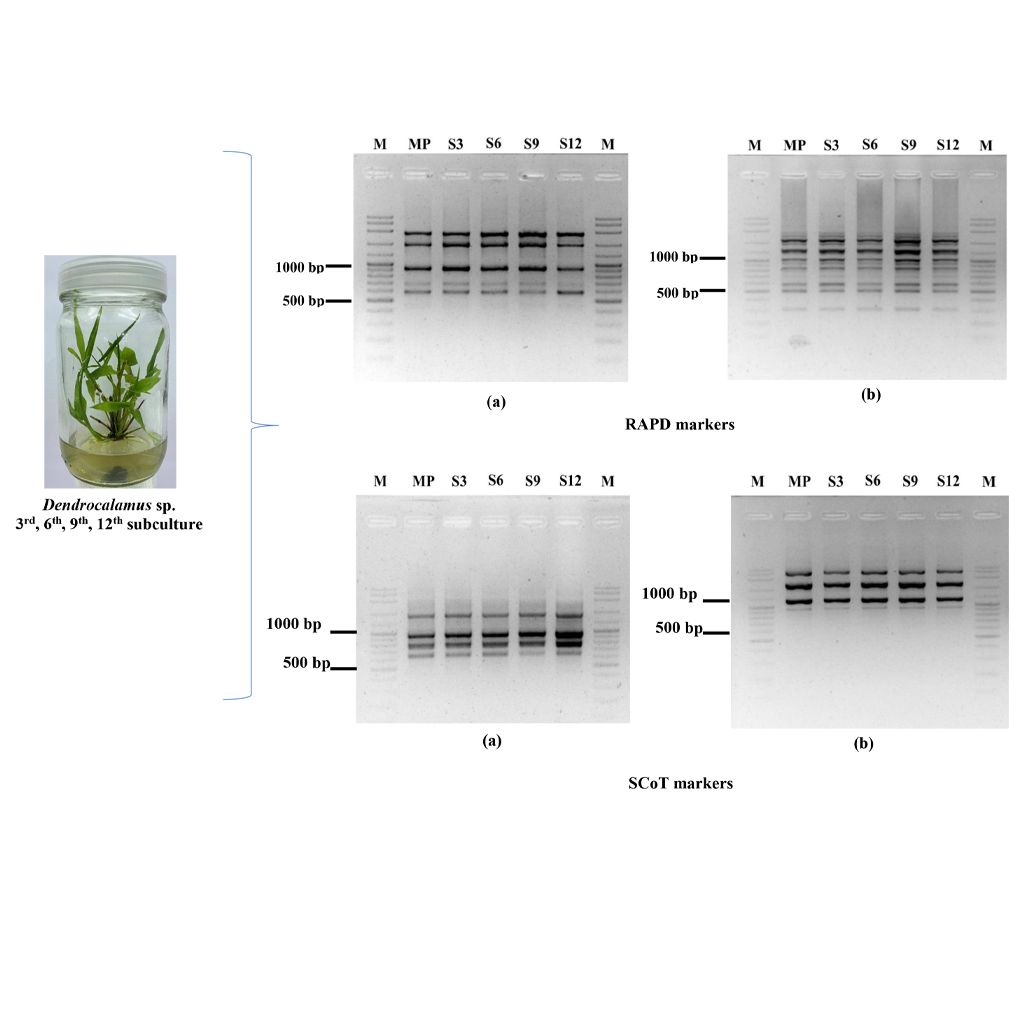
Downloads
References
ER Konzen, R Peron, MA Ito, GE Brondani and SM Tsai. Molecular identification of bamboo genera and species based on RAPD-RFLP markers. Silva Fennica 2017; 51(4), 1691.
M Das, S Bhattacharya, P Singh, TS Filgueiras and A Pal. Bamboo taxonomy and diversity in the era of molecular markers. Advances in Botanical Research 2008; 47, 225-268.
S Sungkaew, A Teerawatananon and K Jindawong. Bamboo of Thailand. Amarin Printing and Publishing, Bangkok, Thailand, 2011.
H Lalhruaitluanga and MNV Prasad. Comparative results of RAPD and ISSR markers for genetic diversity assessment in Melocanna baccifera Roxb. growing in Mizoram State of India. African Journal of Biotechnology 2009; 8(22), 6053-6062.
H Nilkanta, T Amom, L Tikendra, H Rahaman and P Nongdam. ISSR marker based population genetic study of Melocanna baccifera (Roxb.) Kurz: A commercially important bamboo of Manipur, North-East India. Scientifica 2017; 2017, 3757238.
T Puangchick, B Harakotr and N Sriskulpair. Effects of formula and rates of chemical fertilizer on growth of bamboo (Dendrocalamus sericeus) (in Thai). Thai Journal of Science and Technology 2018; 7(2), 113-122.
S Pattanaik, P Das, E Borah, H Kaur and K Borah. Vegetative multiplication of Bambusa balcooa Roxb. using branch cuttings. Journal of Bamboo and Rattan 2004; 3(4), 365-374.
SR Singh, S Dalal, R Singh, AK Dhawan and RK Kalia. Ascertaining clonal fidelity of micropropagated plants of Dendrocalamus hamiltonii Nees et Arn. ex Munro using molecular markers. In Vitro Cellular and Developmental Biology - Plant 2013; 49, 572-583.
AA Mustafa, MR Derise, WTL Yong and KF Rodrigues. A concise review of Dendrocalamus asper and related bamboos: Germplasm conservation, propagation and molecular biology. Plants 2021; 10(9), 1897.
OA Oumer, K Dagne, T Feyissa, K Tesfaye, J Durai and MZ Hyder. Genetic diversity, population structure, and gene flow analysis of lowland bamboo [Oxytenanthera abyssinica (A. Rich.) Munro] in Ethiopia. Ecology and Evolution 2020; 10(20), 11217-11236.
P Chowdhury, M Das, S Sikdar and A Pal. Influence of the physiological age and position of the nodal explants on micropropagation of field-grown Dendrocalamus strictus Nees. Plant Cell Biotechnology and Molecular Biology 2004; 5(1-2), 45-50.
S Kapruwan, M Bakshi and M Kaur. Effect of growth regulators on the in vitro multiplication of Dendrocalamus hamiltonii. International Journal of Engineering Research and Applications 2014; 4(11), 83-86.
BN Pandey and NB Singh. Micropropagation of Dendrocalamus strictus Nees from mature nodal explants. Journal of Applied and Natural Science 2012; 4(1), 5-9.
SR Singh, S Dalal, R Singh, AK Dhawan and RK Kalia. Micropropagation of Dendrocalamus asper {Schult. & Schult. F.} Backer ex k. Heyne): An exotic edible bamboo. Plant Biochemistry and Biotechnology 2012; 21, 220-228.
L Zailiu and H Chaomao. Study on tissue culture of Dendrocalamus sinicus. Scientia Silvae Sinica 2006; 42(2), 43-49.
P Venkatachalam, K Kalaiarasi and S Sreeramanan. Influence of plant growth regulators (PGRs) and various additives on in vitro plant propagation of Bambusa arundinacea (Retz.) Wild: A recalcitrant bamboo species. Journal of Genetic Engineering and Biotechnology 2015; 13(2), 193-200.
MDKM Gunasena, PH Chandrasena and WTPSK Senarath. In vitro mass propagation of Dendrocalamus asper (Giant bamboo) through direct organogenesis. Advances in Bamboo Science 2024; 8, 100090.
BNS Murthy, SJ Murch and PK Saxena. Thidiazuron: A potent regulator of in vitro plant morphogenesis. In Vitro Cellular and Developmental Biology - Plant 1998; 34, 267-275.
SR Pai and NS Desai. Effect of TDZ on various plant cultures. In: N Ahmad and M Faisal (Eds.). Thidiazuron: From urea derivative to plant growth regulator. Springer Nature, Singapore, 2018, p. 439-454.
S Bordoloi, BL Singha, PB Goswami and INA Hazarika. Improved clonal propagation of superior Dendrocalamus hamiltonii Nees germplasm through in vitro techniques. Global Journal of Bio-science and Biotechnology 2018; 7, 537.
RI Raju and SK Roy. Mass propagation of Bambusa bambos (L.) Voss through in vitro culture. Jahangirnagar University Journal of Biological Sciences 2016; 5(2), 15-26.
PJ Larkin and WR Scowcroft. Somaclonal variation - a novel source of variability from cell cultures for plant improvement. Theoretical and Applied Genetics 1981; 60, 197-214.
M Sato, M Hosokawa and M Doi. Somaclonal variation is induced de novo via the tissue culture process: A study quantifying mutated cells in Saintpaulia. PLoS One 2011; 6(8), e23541.
N Abdalla, H El-Ramady, MK Seliem, ME El-Mahrouk, N Taha, Y Bayoumi, TA Shalaby and J Dobránszki. An academic and technical overview on plant micropropagation challenges. Horticulturae 2022; 8(8), 677.
H Krishna, M Alizadeh, D Singh, U Singh, N Chauhan, M Eftekhari and RK Sadh. Somaclonal variations and their applications in horticultural crops improvement. 3 Biotech 2016; 6(1), 54.
H Mehbub, A Akter, MA Akter, MSH Mandal, MA Hoque, M Tuleja and H Mehraj. Tissue culture in ornamentals: Cultivation factors, propagation techniques, and its application. Plants 2022; 11(23), 3208.
R Mehta, V Sharma, A Sood, M Sharma and RK Sharma. Induction of somatic embryogenesis and analysis of genetic fidelity of in vitro-derived plantlets of Bambusa nutans Wall., using AFLP markers. European Journal of Forest Research 2010; 130(5), 729-736.
T Dey, S Saha and PD Ghosh. Somaclonal variation among somatic embryo derived plants - Evaluation of agronomically important somaclones and detection of genetic changes by RAPD in Cymbopogon winterianus. South African Journal of Botany 2015; 96, 112-121.
J Mamgain, A Mujib, B Ejaz, B Gulzar, MQ Malik and R Syeed. Flow cytometry and start codon targeted (SCoT) genetic fidelity assessment of regenerated plantlets in Tylophora indica (Burm. f.) Merrill. Plant Cell, Tissue and Organ Culture 2022; 150(1), 129-140.
R Tisarum, T Samphumphuang, W Prommee, C Mongkolsiriwatana and S Cha-Um. True-to-type micropropagated plants of para rubber (Hevea brasiliensis Müll. Arg.) via somatic embryogenesis. Notulae Botanicae Horti Agrobotanici Cluj-Napoca 2020; 48(4), 1901-1914.
S Pradhan, YP Paudel, W Qin and B Pant. Genetic fidelity assessment of wild and tissue cultured regenerants of a threatened orchid, Cymbidium aloifolium using molecular markers. Plant Gene 2023; 34, 100418.
GK Rohela, P Jogam, MY Mir, AA Shabnam, P Shukla, S Abbagani and AN Kamili. Indirect regeneration and genetic fidelity analysis of acclimated plantlets through SCoT and ISSR markers in Morus alba L. cv. Chinese white. Biotechnology Reports 2020; 25, e00417.
T Siringam and O Vanijajiva. The effect of plant growth regulators on micropropagation of Melientha suavis Pierre. and assessment of genetic fidelity of regenerants based on iPBS and SRAP markers. Biodiversitas Journal of Biological Diversity 2023; 24(9), 4628-4634.
AK Goyal, S Pradhan, BC Basistha and A Sen. Micropropagation and assessment of genetic fidelity of Dendrocalamus strictus (Roxb.) Nees using RAPD and ISSR markers. 3 Biotech 2015; 5(4), 473-482.
SR Singh, S Dalal, R Singh, AK Dhawan and RK Kalia. Evaluation of genetic fidelity of in vitro raised plants of Dendrocalamus asper (Schult. & Schult. F.) Backer ex K. Heyne using DNA-based markers. Acta Physiologiae Plantarum 2013; 35, 419-430.
K Tongtape, S Te-Chato and S Yenchon. Somatic Embryo (SE) formation from culturing floral explants of rubber tree (Hevea brasiliensis Muell. Arg.) and assessment of genetic stability by RAPD and SSR markers. Trends in Sciences 2023; 20(9), 6728.
Çetin. Evaluation of the genetic fidelity of in vitro raised plants of Origanum majorana L. using Random Amplified Polymorphic DNA. Celal Bayar University Journal of Science 2018; 14(2), 237-239.
S Sadhu, P Jogam, RK Thampu, S Abbagani, S Penna and V Peddaboina. High efficiency plant regeneration and genetic fidelity of regenerants by SCoT and ISSR markers in chickpea (Cicer arietinum L.). Plant Cell, Tissue and Organ Culture 2020; 141, 465-477.
MK Rai. Start Codon Targeted (SCoT) polymorphism marker in plant genome analysis: Current status and prospects. Planta 2023; 257(2), 34.
T Amom, L Tikendra, N Apana, M Goutam, P Sonia, AS Koijam, AM Potshangbam, H Rahaman and P Nongdam. Efficiency of RAPD, ISSR, iPBS, SCoT and phytochemical markers in the genetic relationship study of five native and economical important bamboos of North-East India. Phytochemistry 2020; 174, 112330.
TVD Huynh, TCH Cao, HTT Nguyen, TCV Tran, MQL Huynh, PL Tran and PV Nguyen. Somatic embryogenesis and plant regeneration from leaf callus with genetic stability validation using SCoT markers in Paramignya trimera, a medicinal plant native to Vietnam. Notulae Botanicae Horti Agrobotanici Cluj-Napoca 2024; 52(2), 13886.
H Kudikala, P Jogam, A Sirikonda, K Mood and VR Allini. In vitro micropropagation and genetic fidelity studies using SCoT and ISSR primers in Annona reticulata L.: An important medicinal plant. Vegetos 2020; 33, 446-457.
T Murashige and F Skoog. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiologia Plantarum 1962; 15(3), 473-497.
JJ Doyle and JL Doyle. A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochemical Bulletin 1987; 19, 11-15.
M Thakur, Rakshandha, V Sharma and A Chauhan. Genetic fidelity assessment of long term in vitro shoot cultures and regenerated plants in Japanese plum cvs Santa Rosa and Frontier through RAPD, ISSR and SCoT markers. South African Journal of Botany 2021; 140, 428-433.
JG Williams, AR Kubelik, KJ Livak, JA Rafalski and SV Tingey. DNA polymorphisms amplified by arbitrary primers are useful as genetic markers. Nucleic Acids Research 1990; 18(22), 6531-6535.
B Guo, BH Abbasi, A Zeb, LL Xu and YH Wei. Thidiazuron: A multi-dimensional plant growth regulator. African Journal of Biotechnology 2011; 10(45), 8984-9000.
HM Ali, T Khan, MA Khan and N Ullah. The multipotent thidiazuron: A mechanistic overview of its roles in callogenesis and other plant cultures in vitro. Biotechnology and Applied Biochemistry 2022; 69(6), 2624-2640.
FG Celikel, Q Zhang, Y Zhang, MS Reid and CZ Jiang. A cytokinin analog thidiazuron suppresses shoot growth in potted rose plants via the gibberellic acid pathway. Frontiers in Plant Science 2021; 12, 639717.
LA Erland, RT Giebelhaus, JMR Victor, SJ Murch and PK Saxena. The morphoregulatory role of thidiazuron: Metabolomics-guided hypothesis generation for mechanisms of activity. Biomolecules 2020; 10(9), 1253.
M Vescovi, M Riefler, M Gessuti, O Novak, T Schmulling and FL Schiavo. Programmed cell death induced by high levels of cytokinin in Arabidopsis cultured cells is mediated by the cytokinin receptor CRE1/AHK4. Journal of Experimental Botany 2012; 63(7), 2825-2832.
S Nurhayani, R Megia and R Purnamaningsih. In vitro propagation of Bambusa balcooa as alternative material of wood. Biosaintifika: Journal of Biology and Biology Education 2018; 10(1), 198-204.
KD Mudoi, SP Saikia, A Goswami, A Gogoi, D Bora and M Borthakur. Micropropagation of important bamboos: A review. African Journal of Biotechnology 2013; 12(20), 2770-2785.
S Arya, S Sharma, R Kaur and ID Arya. Micropropagation of Dendrocalamus asper by shoot proliferation using seeds. Plant Cell Reports 1999; 18, 879-882.
SMSD Ramanayake, VN Meemaduma and TE Weerawardene. In vitro shoot proliferation and enhancement of rooting for the large-scale propagation of yellow bamboo (Bambusa vulgaris ‘Striata’). Scientia Horticulturae 2006; 110(1), 109-113.
QM Wang and L Wang. An evolutionary view of plant tissue culture: Somaclonal variation and selection. Plant Cell Reports 2012; 31(9), 1535-1547.
S Sun, JQ Zhong, SH Li and XJ Wang. Tissue culture-induced somaclonal variation of decreased pollen viability in torenia (Torenia fournieri Lind.). Botanical Studies 2013; 54, 36.
T Arumugam, G Jayapriya and T Sekar. Molecular fingerprinting of the Indian medicinal plant Strychnos minor Dennst. Biotechnology Reports 2019; 21, e00318.
K Sharma, AK Mishra and RS Misra. The genetic structure of taro: A comparison of RAPD and isozyme markers. Plant Biotechnology Reports 2008; 2, 191-198.
BCY Collard and DJ Mackill. Start codon targeted (SCoT) polymorphism: A simple, novel DNA marker technique for generating gene-targeted markers in plants. Plant Molecular Biology Reporter 2009; 27, 86-93.
SR Singh, S Dalal, R Singh, AK Dhawan and RK Kalia. Evaluation of genetic fidelity of in vitro raised plants of Dendrocalamus asper (Schult. & Schult. F.) Backer ex K. Heyne using DNA-based markers. Acta Physiologiae Plantarum 2013; 35, 419-430.
D Negi and S Saxena. Ascertaining clonal fidelity of tissue culture raised plants of Bambusa balcooa Roxb. using inter simple sequence repeat markers. New Forests 2010; 40, 1-8.
MM Suwal, J Lamichhane and DP Gauchan. Assessment of genetic stability of micropropagated Bambusa balcooa Roxb. using RAPD marker. Plant Tissue Culture and Biotechnology 2021; 31(1), 81-95.
O Borsai, M Hârța, K Szabo, CD Kelemen, FA Andrecan, MM Codrea and D Clapa. Evaluation of genetic fidelity of in vitro-propagated blackberry plants using RAPD and SRAP molecular markers. Horticultural Science 2020; 47(1), 21-27.
J Brar, A Shafi, P Sood, A Sood and M Anand. Micropropagation of Dendrocalamus membranaceus Munro. through axillary shoot proliferation and confirmation of clonal fidelity of in vitro raised plants. Journal Bamboo Rattan 2012; 11(1-2), 13-28.
K Kalaiarasi, P Sangeetha, S Subramaniam and P Venkatachalam. Development of an efficient protocol for plant regeneration from nodal explants of recalcitrant bamboo (Bambusa arundinacea Retz. Willd) and assessment of genetic fidelity by DNA markers. Agroforestry Systems 2014; 88(3), 527-537.
HK Nadha, R Kumar, RK Sharma, M Anand and A Sood. Evaluation of clonal fidelity of in vitro raised plants of Guadua angustifolia Kunth using DNA-based markers. Journal of Medicinal Plants Research 2011; 5(23), 5636-5641.
RK Agnihotri, J Mishra and SK Nandi. Improved in vitro shoot multiplication and rooting of Dendrocalamus hamiltonii Nees et Arn. Ex Munro: Production of genetically uniform plants and field evaluation. Acta Physiologiae Plantarum 2009; 31, 961-967.
D Sarmah, PP Mohapatra, MF Seleiman, T Mandal, N Mandal, K Pramanik, C Jane, S Sow, BA Alhammad, N Ali, S Ranjan and DO Wasonga. Efficient regeneration of in vitro derived plants and genetic fidelity assessment of Phalaenopsis orchid. Frontiers in Sustainable Food Systems 2024; 8, 1359486.
SS Kadapatti and HN Murthy. Rapid plant regeneration, analysis of genetic fidelity, and neoandrographolide content of micropropagated plants of Andrographis alata (Vahl) Nees. Journal of Genetic Engineering and Biotechnology 2021; 19(1), 20.
FMK Aldabbagh, IH AL-Zaidi and MA ALshamari. Micropropagation and assessment of genetic fidelity of regenerate by RAPD markers of Solanum nigrum. Iraqi Journal of Agricultural Sciences 2024; 55(1), 432-439.
A Kader, SN Sinha and P Ghosh. Clonal fidelity investigation of micropropagated hardened plants of jackfruit tree (Artocarpus heterophyllus L.) with RAPD markers. Journal of Genetic Engineering and Biotechnology 2022; 20(1), 145.
M Rani, MAH Miah, MT Hasan, MHO Rashid, S Yasmin and MS Haque. Clonal propagation of turmeric (Curcuma longa) and confirmation of genetic fidelity of the micropropagated shoots by RAPD markers. Plant Tissue Culture and Biotechnology 2024; 34(1), 55-69.
PR Joshi, S Pandey, L Maharjan and B Pant. Micropropagation and assessment of genetic stability of Dendrobium transparens wall. Ex Lindl. using RAPD and ISSR markers. Frontiers in Conservation Science 2023; 3, 1083933.
S Pandey, L Maharjan and B Pant. In vitro propagation and assessment of genetic homogeneity using RAPD and ISSR markers in Tinospora cordifolia (Wild.) Hook. F. & Thoms, an important medicinal plant of Nepal. Journal of Nepal Biotechnology Association 2023; 4(1), 27-36.
D Swain, G Pushpalatha and P Das. In vitro micro-propagation of Typhonium flagelliforme (Lodd.) Blume and its genetic fidelity using ISSR and RAPD markers. Journal of Pharmacognosy and Phytochemistry 2020; 9(4), 3484-3488.
BS Rajput, M Jani, K Ramesh, M Manokari, P Jogam, VR Allini, MM Kher and MS Shekhawat. Large-scale clonal propagation of Bambusa balcooa Roxb.: An industrially important bamboo species. Industrial Crops and Products 2020; 157, 112905.
MK Badhepuri, PR Beeravelli, RG Arolla, P Jogam, GK Rohela and NR Singisala. Micropropagation and genetic fidelity analysis using SCoT and ISSR markers in Muehlenbeckia platyclada (F. Muell.) meisn. Plant Cell, Tissue and Organ Culture 2024; 157(3), 51.
P Chirumamilla, C Gopu, P Jogam and S Taduri. Highly efficient rapid micropropagation and assessment of genetic fidelity of regenerants by ISSR and SCoT markers of Solanum khasianum Clarke. Plant Cell, Tissue and Organ Culture 2021; 144, 397-407.
D Clapa and M Hârţa. Evaluation of genetic fidelity of in vitro growth plants of highbush blueberry (Vaccinium corymbosum L.) cultivars using scot molecular markers. Scientific Papers. Series B, Horticulture 2022; 66(1), 67-74.
N Kaushal, A Alok, M Kajal and K Singh. Regeneration and genetic fidelity analysis of Chlorophytum borivilianum using flower stalk as explant source. Advances in Bioscience and Biotechnology 2021; 12, 95-107.
AA Al-Aizari, YH Dewir, AH Ghazy, A Al-Doss and RS Al-Obeed. Micropropagation and genetic fidelity of fegra fig (Ficus palmata Forssk.) and grafting compatibility of the regenerated plants with Ficus carica. Plants 2024; 13(9), 1278.
V Bisht, JM Rawat, KS Gaira, S Purohit, J Anand, S Sinha, D Mitra, FS Ataya, AM Elgazzar, GES Batiha and B Rawat. Assessment of genetic homogeneity of in-vitro propagated apple root stock MM 104 using ISSR and SCoT primers. BMC Plant Biology 2024; 24(1), 240.
KW Sultana, S Das, I Chandra and A Roy. Efficient micropropagation of Thunbergia coccinea Wall. and genetic homogeneity assessment through RAPD and ISSR markers. Scientific Reports 2022; 12(1), 1683.
MS Alwahibi, AA Alawaadh, YH Dewir, DA Soliman and MK Seliem. Assessment of genetic fidelity of lacy tree philodendron (Philodendron bipinnatifidum Schott ex Endl.) micro propagated plants. Revis Bionatura 2022; 7(1), 10.
W Muslihatin, TB Saputro, AT Wibowo and YSW Manuhara. Assessment of genetic stability of micropropagated Curculigo latifolia from Indonesia by RAPD and ISSR. Journal of Applied Biology and Biotechnology 2025; 13(1), 229-234.
L Tikendra, AM Potshangbam, A Dey, TR Devi, MR Sahoo and P Nongdam. RAPD, ISSR, and SCoT markers based genetic stability assessment of micropropagated Dendrobium fimbriatum Lindl. var. oculatum Hk. f. - an important endangered orchid. Physiology and Molecular Biology of Plants 2021; 27, 341-357.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






