In Vitro Propagation of Gymnocalycium mihanovichii LB2178 Agua Dulce (Cactaceae) and Genetic Stability Assessment Using Simple Sequence Repeat (SSR) Markers
DOI:
https://doi.org/10.48048/tis.2025.9827Keywords:
Gymnocalycium mihanovichii LB2178 agua dulce, In vitro, Genetic stability, SSR markers, 2,4 Dichlorophenoxyacetic acid, Anthocyanin, HistologicalAbstract
The Gymnocalycium mihanovichii LB2178 Agua Dulce is a popular variety of cactus, known for its slightly raised skin known as “stripes” or bone pattern. It is also in high commercial demand. Therefore, the objective of this study were to study the effect of plant growth regulators and various explants fragmentation on callus induction and plant regeneration of G. mihanovichii LB2178 Agua Dulce and assessment genetic stability by SSR markers. The 1 cm seedlings of G. mihanovichii LB2178 Agua Dulce explant and callus were used as plant material. After 3 months of culture, the results showed that the explant sectioned longitudinally were cultured on MS medium supplemented with 2 mg/L 2,4-D, which gave the highest survival rate, 100 %, the highest callus induction, 100 % and the highest callus fresh weight, 2.72 g. The explant sectioned transversally were cultured on MS medium supplemented with 1 mg/L 2,4-D gave the highest anthocyanin contents, 0.00149 mg/gFW. The anatomical features of the callus derived from the tissue properties of the callus generated from transversely sectioned plant tissue culture, the epidermis layer is clearly visible, and there are vascular bundles arranged around the stem. The callus fragments were cultured on MS medium supplemented with 0.5 mg/L NAA and 3.5 mg/L BA gave the highest survival rate, 100 %, shoot induction, 90 %, a number of shoots, 2.20 shoots/explant and the shoot length, 0.28 cm. Assessment of genetic fidelity was performed on the G. mihanovichii LB2178 Agua Dulce micro propagated plantlets using SSR analysis with 8 SSR primers. The range of the chosen primers was 100 - 1,500 bp. The 4 of the 8 examined primers exhibited no polymorphism, whereas primer 4 displayed 83.33 % polymorphism.
HIGHLIGHTS
- Effect of different explant fragmentation and different concentrations of 2,4 Dichlorophenoxyacetic acid (2,4-D) on callus induction of mihanovichii LB2178 Agua Dulce.
- Quantification of anthocyanins from callus of mihanovichii LB2178 Agua Dulce.
- Genetic stability of the mihanovichii LB2178 Agua Dulce using SSR markers.
GRAPHICAL ABSTRACT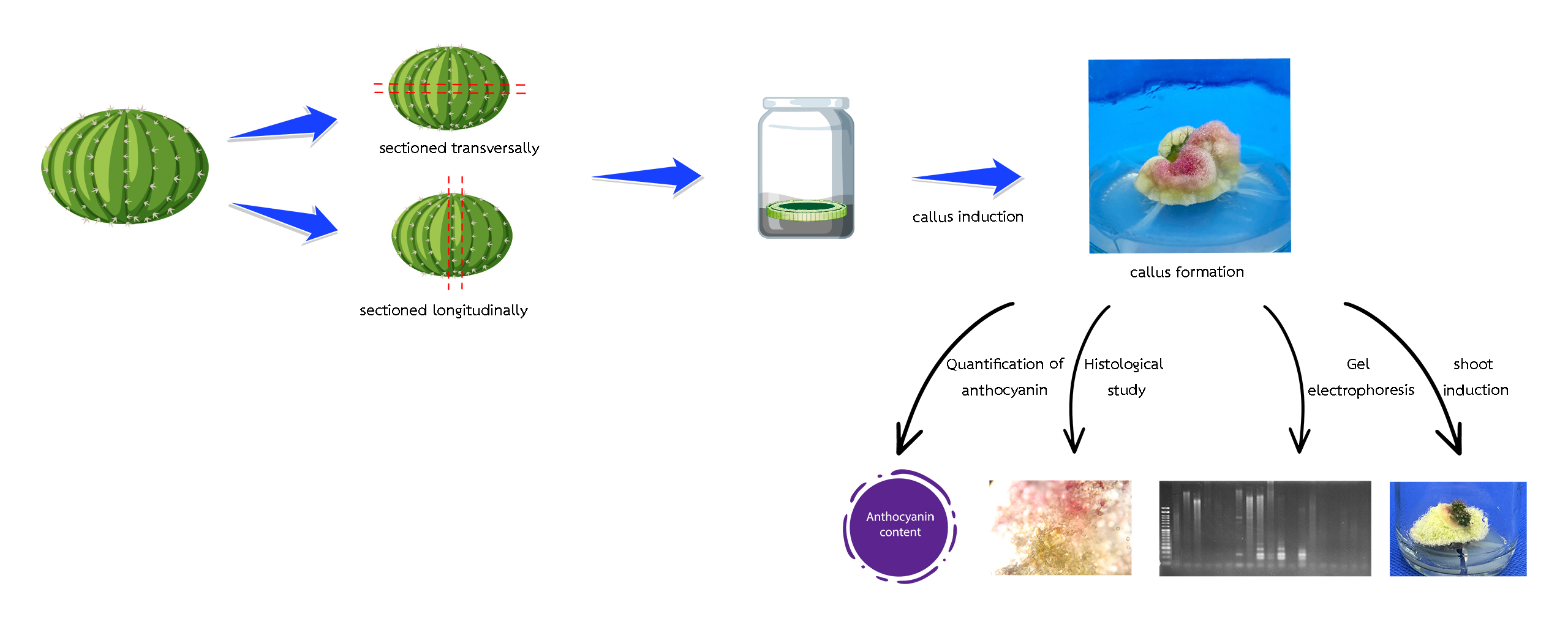
Downloads
References
G Rowley and M Kimnach. The new cactus lexicon. Cactus and Succulent Journal 2006; 78(6), 318-319.
M Arakaki, PA Christin, R Nyffeler, A Lendel, U Eggli, RM Ogburn, E Spriggsa, MJ Moored and EJ Edwards. Contemporaneous and recent radiations of the world’s major succulent plant lineages. Proceedings of the National Academy of Sciences 2011; 108(20), 8379-8384.
TH Hernández, HM Hernández, JAD Nova, R Puente, LE Eguiarte and S Magallón. Phylogenetic relationships and evolution of growth form in Cactaceae (Caryophyllales, Eudicotyledoneae). American Journal of Botany 2011; 98(1), 44-61.
EF Anderson. The cactus family. Timber Press, Oregon, 2001, p. 562-564.
B Goettsch, C Hilton-Taylor, G Cruz-Piñón, JP Duffy, A Frances, HM Hernández, R Inger, C Pollock, J Schipper, M Superina, NP Taylor, M Tognelli, AM Abba, S Arias, HJ Arreola-Nava, MA Baker, RT Bárcenas, D Barrios, P Braun, CA Butterworth, …, KJ Gaston. High proportion of cactus species threatened with extinction. Nature Plants 2015; 1, 15142.
S Boonyang. Business plan for building a new business sb cactus garden: Small and medium-sized enterprises. Bangkok University, Bangkok, Thailand, 2016.
MO Brutsch and MB Scott. Extending the fruiting season of spineless prickly pear (Opuntia ficus-indica). Journal of the South African Society for Horticultural Science 1991; 1, 73-76.
JHC Sánchez and DS Croutch. Notes on the floral biology of Praecereus euchlorus subsp. euchlorus (Cactaceae). Schumannia 2008; 5, 102-103.
T Galili. Dendextend: An R package for visualizing, adjusting and comparing trees of hierarchical clustering. Bioinformatics 2015; 31(22), 3718-3720.
A Novoa, J Rodr´ıguez, AL Nogueira, DM Richardson and L Gonzalez. Seed characteristics in Cactaceae: Useful f,diagnostic features for screening species for invasiveness. South African Journal of Botany 2016; 105, 61-65.
NL Britton and JN Rose. The Cactaceae: Descriptions and illustrations of plants of the cactus family. The Carnegie Institution, Washington, 1922.
P Supanantananon. Cactus. Amarin Printing and Publishing, Bangkok, Thailand, 2019, p. 7-10.
S Kanchanakul. Roiphan phrueksa cactus. Amarin Printing and Publishing, Bangkok, Thailand, 2014, p. 105-108.
S Ramasoot. Textbook on plant tissue culture. Nakhon Si Thammarat Rajabhat University, Nakhon Si Thammarat, Thailand, 2016, p. 106-108.
MSS Díaz, RM Ontiveros, AA Gómez and MDLS Diaz. In vitro organogenesis of Pelecyphora aselliformis Erhenberg (Cactaceae). In vitro Cellular and Developmental Biology - Plant 2003; 39, 480-484.
EPM Balch and CAD Figueroa. In vitro propagation of Pelecyphora aselliformis Ehrenberg and P. strobiliformis Werdermann (Cactaceae). In vitro Cellular and Development Biology - Plant 2002; 38, 73-78.
JL Rumińska and D Kulus. Micropropagation of cacti: A review. Haseltonia 2014; 2014(19), 46-63.
JL Rumińska and A Fijalkowska. The rate of callus growth and regeneration of embryogenic structures in cactus of Gymnocalycium genus depending on the light conditions. Zeszyty Problemowe Postepw Nauk Rolniczych 2006; 510, 325-331.
JL Rumińska and D Kulus. Micropropagation of cacti: A review. Haseltonia 2014; 2014(19), 46-63.
LA Medeiros, RCS Ribeiro, LA Gallo, ET Oliveira and MESP Dematte. In vitro propagation of Notocactus magnificus. Plant Cell, Tissue and Organ Culture 2006; 84, 165-169.
JL Müller. Auxin conjugates: Their role for plant development and in the evolution of land plants. Journal of Experimental Botany 2011; 62(6), 1757-1773.
GT Silva, SV Resende, AL Brito, HB Bezerra, JRF Santana and AS Schnadelbach. In vitro shoot production, morphological alterations and genetic instability of Melocactus glaucescens (Cactaceae), an endangered species endemic to eastern Brazil. South African Journal of Botany 2018; 115, 100-107.
MT Hamblin, ML Warburton and ES Buckler. Empirical comparison of simple sequence repeats and single nucleotide polymorphisms in assessment of maize diversity and relatedness. PLoS One 2007; 2(12), e1367.
D Csencsics, S Brodbeck and R Holderegger. Cost-effective, species-specific microsatellite development for the endangered dwarf bulrush (Typha minima) using next generation sequencing technology. Journal of Heredity 2010; 101(6), 789-793.
S Dutta, G Kumawat, BP Singh, DK Gupta, S Singh, V Dogra, K Gaikwad, TR Sharma, RS Raje, TK Bandhopadhya, S Datta, MN Singh, F Bashasab, P Kulwal, KB Wanjari, RK Varshney, DR Cook and NK Singh. Development of genic-SSR markers by deep transcriptome sequencing in pigeonpea [Cajanus cajan (L.) Millspaugh]. BMC Plant Biology 2011; 11, 17.
M Caruso, S Curr and CG Las. Microsatellite markers help to assess genetic diversity among Opuntia Ficusindica cultivated genotypes and their relation with related species. Plant Systematics and Evolution 2010; 290, 85-97.
WS Fava, VGN Gomes, AP Lorenz and GM Paggi. Cross amplification of microsatellite loci in the cacti species from Brazilian chaco. Molecular Biology Reports 2020; 47(2), 1535-1542.
JM Tsholofelo, FM Mathabatha, F Herman and MC Gesine. Genetic diversity and differentiation of South African cactus pear cultivars (Opuntia spp.) based on simple sequence repeat (SSR) markers. Genetic Resources and Crop Evolution 2023; 71(1), 373-384.
KI Ereifej, H Feng, T Rababah, A Almajwal, M Alu’datt, S Gammoh and LI Oweis. Chemical composition, phenolics, anthocyanins concentration and antioxidant activity of tenwild edible plants. Food and Nutrition Sciences 2015; 6(7), 581-590.
I Rabino and AL Mancinelli. Light, temperature, and anthocyanin production. Plant Physiology 1986; 81(3), 922-924.
B Habibeh, A Ghasemnezhad, M Alizadeh and AS Mahonak. Optimizing callus induction and analyzing in vitro phytochemicals in San Pedro cactus (Echinopsis pachanoi). Journal of Horticulture and Postharvest Research 2024; 7(2), 197-212.
MR Martıinez, APBL Rosa, F Gueraud, AN Salvayre, M Rossigno and MDSS Diaz. Establishment of callus and cell suspensions of wild and domesticated Opuntia species: Study on their potential as a source of metabolite production. Plant Cell, Tissue and Organ Culture 2016; 124, 181-189.
TL Ho, S Te-Chato and S Yenchon. Callus induction and plantlet regeneration systems in Indica rice (Oryza sativa L.) Cultivar Sangyod. Walailak Journal of Science and Technology 2018; 15(10), 753-763.
A Libin, PJH King, KH Ong, JK Chubo and P Sipen. Callus induction and plant regeneration of Sarawak rice (Oryza sativa L.) variety Biris. African Journal of Agricultural Research 2012; 7(30), 4260-4265.
K Tongtape, S Te-chato and S Yenchon. Somatic Embryo (SE) formation from culturing floral explants of rubber tree (Hevea brasiliensis Muell. Arg.) and assessment of genetic stability by RAPD and SSR markers. Trends in Sciences 2023; 20(9), 6728.
N Maharik, SE Elgengaihib and H Taha. Anthocyanin production in callus cultures of Crataegus Sinaica Boiss. International Journal of Academic Research 2009; 1, 30-34.
DAL Valler. In-vitro production of anthocyanin in Sesbania grandiflora (Red Katuray) as influenced by varying concentrations of 2,4-D and BA added on MS medium. American Journal of Plant Sciences 2016; 7(15), 2297-2306.
K Matsumoto. Micro-propagation of bananas. In: SM Jain and K Ishii (Eds.). Micropropagation of woody trees and fruits. Kluwer Academic Publishers, Alphen aan den Rijn, Netherlands, 2003, p. 353-380.
A Karthikeyan, SK Pandian and M Ramesh. Agrobacterium-mediated transformation of leaf base derived callus tissues of popular indica rice (Oryza sativa L. sub sp. indica cv. ADT 43). Plant Science 2011; 181(3), 258-268.
R Kawi. Plant tissue culture: Principles and techniques, type II 3. Agronomy Department, Faculty of Agriculture, Kasetsart University, Bangkok, Thailand, 2002.
Y Meksuwan1 and P Sutthinon. In vitro propagation and histochemical analysis of Launaea sarmentosa (Willd.) Kuntze. Trends in Sciences 2023; 20(5), 5729.

Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






