NASICON-PEO (Polyethylene Oxide) Polymer-in-Ceramic Composite Electrolytes: Thermal, Structural and Electrical Properties
DOI:
https://doi.org/10.48048/tis.2025.9672Keywords:
Composite electrolyte, NASICON (LSPO), Polymer electrolyte, PEO-LiTFSI electrochemical impedance spectroscopy (EIS), Thermal stability, Bulk conductivityAbstract
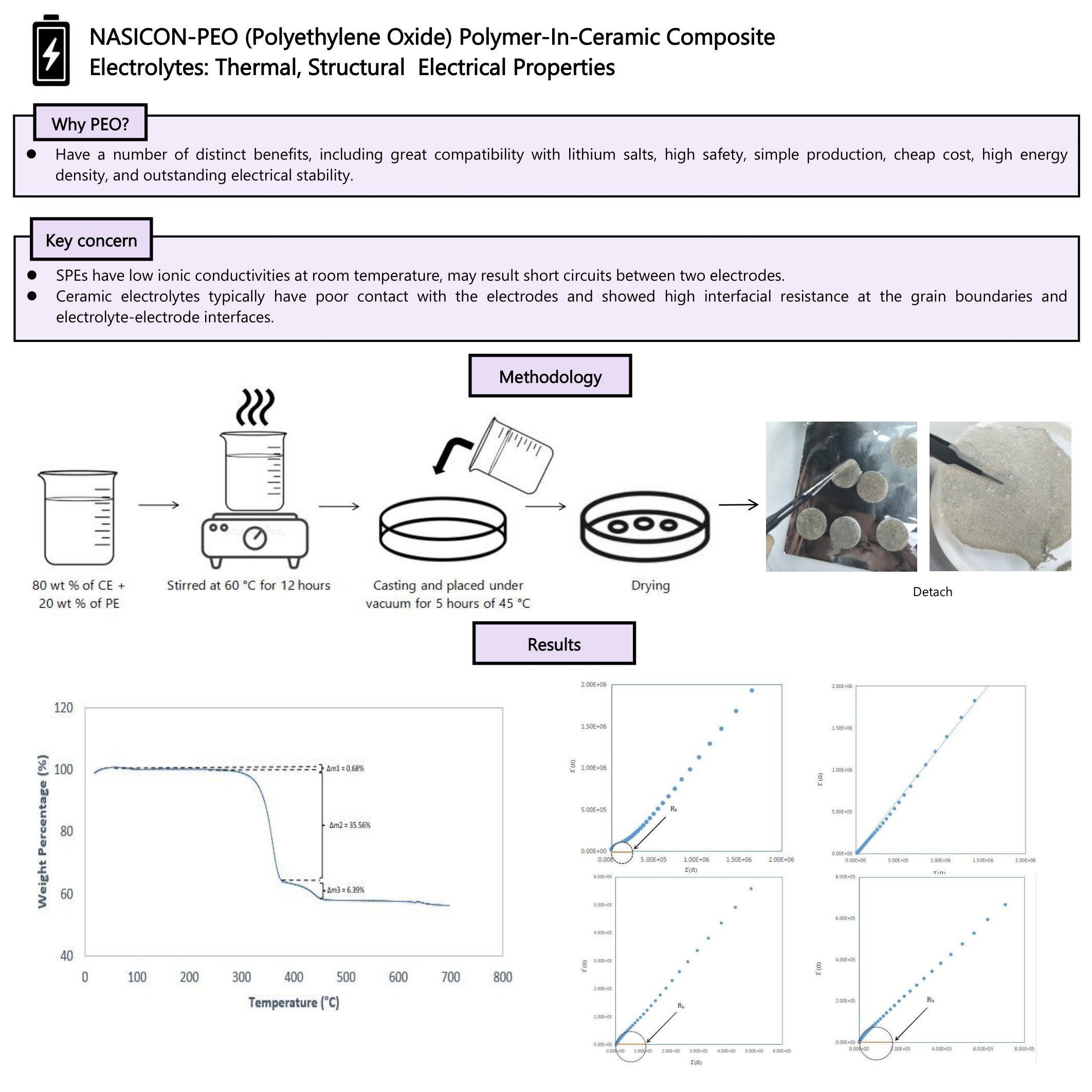
A composite electrolyte consisting of sodium super ionic conductor (NASICON) LiSn2P3O12 (LSPO) and a polymer electrolyte (80:20 polyethylene oxide (PEO) with LiTFSI salt) was prepared using the solution casting method. The thermal stability of the composite was assessed, revealing a second-stage decomposition temperature range between 375 and 462 °C, with a glass transition temperature (Tg) of 28 °C. X-ray diffraction (XRD) analysis indicated low crystallinity in PEO-LiTFSI, with no significant structural changes, despite the emergence of a new peak at 20.7 °C. Electrochemical impedance spectroscopy (EIS) was used to investigate the electrical properties at seven different temperatures (30, 40, 50, 60, 70, 80 and 90 °C), with the highest bulk conductivity of 6.2825×10−5 S/cm observed at 50 °C. The results demonstrate that the addition of LSPO ceramic electrolyte and temperature significantly influence the thermal, structural, and electrical properties of the composite electrolyte.
HIGHLIGHTS
- This article focused on composite electrolyte consisting of NASICON, LiSn2P3O12 (LSPO) and a polymer
electrolyte (80:20 PEO with LiTFSI salt) prepared using the solution casting method.
- The thermal and structural properties of the material indicate good thermal stability and a predominantly crystalline structure, which are favourable characteristics for solid-state electrolyte applications.
- Electrochemical studies found the highest ionic conductivity of 6.2825 × 10⁻⁵ S/cm at 50 °C, highlighting the role of LSPO and temperature on electrolyte performance.
- These findings suggest that the LSPO–PEO composite electrolyte is a promising candidate for solid-state sodium-ion battery applications due to its enhanced thermal and electrochemical properties.
GRAPHICAL ABSTRACT
Downloads
References
SA Ahmed, T Pareek, S Dwivedi, M Badole and S Kumar. Polymer-in-ceramic composite electrolyte with high ionic conductivity for all-solid-state lithium batteries. Journal of Solid-State Electrochemistry 2020; 24, 2407-2417.
P Yao, H Yu, Z Ding, Y Liu, J Lu, M Lavorgna, J Wu and X Liu. Review on polymer-based composite electrolytes for lithium batteries. Frontiers in Chemistry 2019; 7, 522.
FP Nkosi, M Valvo, J Mindemark, NA Dzulkurnain, G Hernández, A Mahun, S Abbrent, J Brus, L Kobera and K Edström. Garnet-poly(ε-caprolactone-co-trimethylene carbonate) polymer-in-ceramic composite electrolyte for all-solid-state lithium-ion batteries. ACS Applied Energy Materials 2021; 4(3), 2531-2542.
Z Wei, Y Ren, M Wang, J He, W Huo and H Tang. Improving the conductivity of solid polymer electrolyte by grain reforming. Nanoscale Research Letters 2020; 15, 122.
Y Chen, Y Kang, Y Zhao, L Wang, J Liu, Y Li, Z Liang, X He, X Li, N Tavajohi and B Li. A review of lithium-ion battery safety concerns: The issues, strategies, and testing standards. Journal of Energy Chemistry 2020; 59, 83-99.
J Zhang, B Li, Q Wang, X Wei, W Feng, Y Chen, P Huang and Z Wang. Application of fourier transform infrared spectroscopy with chemometrics on postmortem interval estimation based on pericardial fluids. Scientific Reports 2017; 7(1), 18013.
A Nandiyanto, R Oktiani and R Ragadhita. How to read and interpret FTIR spectroscopy of organic material. Indonesian Journal of Science and Technology 2019; 4(1), 97-118.
AK Manohar, O Bretschger, KH Nealson and F Mansfeld. The use of electrochemical impedance spectroscopy (EIS) in the evaluation of the electrochemical properties of a microbial fuel cell. Bioelectrochemistry 2008; 72(2), 149-154.
NA Mustaffa, S Adnan, M Sulaiman and NS Mohamed. Low-temperature sintering effects on NASICON-structured solid electrolytes prepared via citric acid-assisted sol-gel method. Ionics 2015; 21, 955-965.
L Chen, Y Li, SP Li, LZ Fan, CW Nan and JB Goodenough. PEO/garnet composite electrolytes for solid-state lithium batteries: From “ceramic-in-polymer” to “polymer-in-ceramic”. Nano Energy 2018; 46, 176-184.
B Zhang, Y Liu, J Liu, L Sun, L Cong, F Fu, A Mauger, CM Julien, H Xie and X Pan. Polymer-in-ceramic based poly(ε-caprolactone)/ceramic composite electrolyte for all-solid-state batteries. Journal of Energy Chemistry 2021; 52, 318-325.
JH Choi, CH Lee, JH Yu, CH Doh and SM Lee. Enhancement of ionic conductivity of composite membranes for all-solid-state lithium rechargeable batteries incorporating tetragonal LLZO into a polyethylene oxide matrix. Journal of Power Sources 2015; 274, 458-463.
MS Park, YC Jung and DW Kim. Hybrid solid electrolytes composed of poly(1,4-butylene adipate) and lithium aluminum germanium phosphate for all-solid-state Li/Li cells. Solid State Ionics 2018; 315, 65-70.
AS Pandian, XC Chen, J Chen, BS Lokitz, RE Ruther, G Yang, K Lou, J Nanda, FM Delnick and NJ Dudney. Facile and scalable fabrication of polymer-ceramic composite electrolyte with high ceramic loadings. Journal of Power Sources 2018; 390, 153-164.
RC Agrawal and GP Pandey. Solid polymer electrolytes: Materials designing and all-solid-state battery applications. Journal of Physics D: Applied Physics 2008; 41(22), 223001.
PG Bruce, B Scrosati and JM Tarascon. Nanomaterials for rechargeable lithium batteries. Angewandte Chemie International Edition 2008; 47(16), 2930-2946.
B Kumar and LG Scanlon. Polymer-ceramic composite electrolytes. Journal of Power Sources 1994; 52(2), 261-268.
D Zhou, D Shanmukaraj, A Tkacheva, M Armand and G Wang. Polymer electrolytes for lithium-based batteries: Advances and prospects. Chem 2019; 5(9), 2326-2352.
JR MacCallum and CA Vincent. Polymer electrolyte reviews. Elsevier Applied Science, London, 1987.
V Aravindan, J Gnanaraj, S Madhavi and HK Liu. Lithium-ion conducting electrolyte salts for lithium batteries. Chemistry - A European Journal 2011; 17(51), 14326-14346.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






