Direct Comparison between Ring-Opening Polymerization and Hydrolysis-Condensation Methods in the Synthesis of Polydimethylsiloxane as a Vitreous Substitute: A Systematic Literature Review
DOI:
https://doi.org/10.48048/tis.2025.9668Keywords:
Dichlorodimethylsilane, Hydrolysis-condensation, Octamethylcyclotetrasiloxane, Polydimethylsiloxane, Ring-opening polymerization, Synthesis, Vitreous substituteAbstract
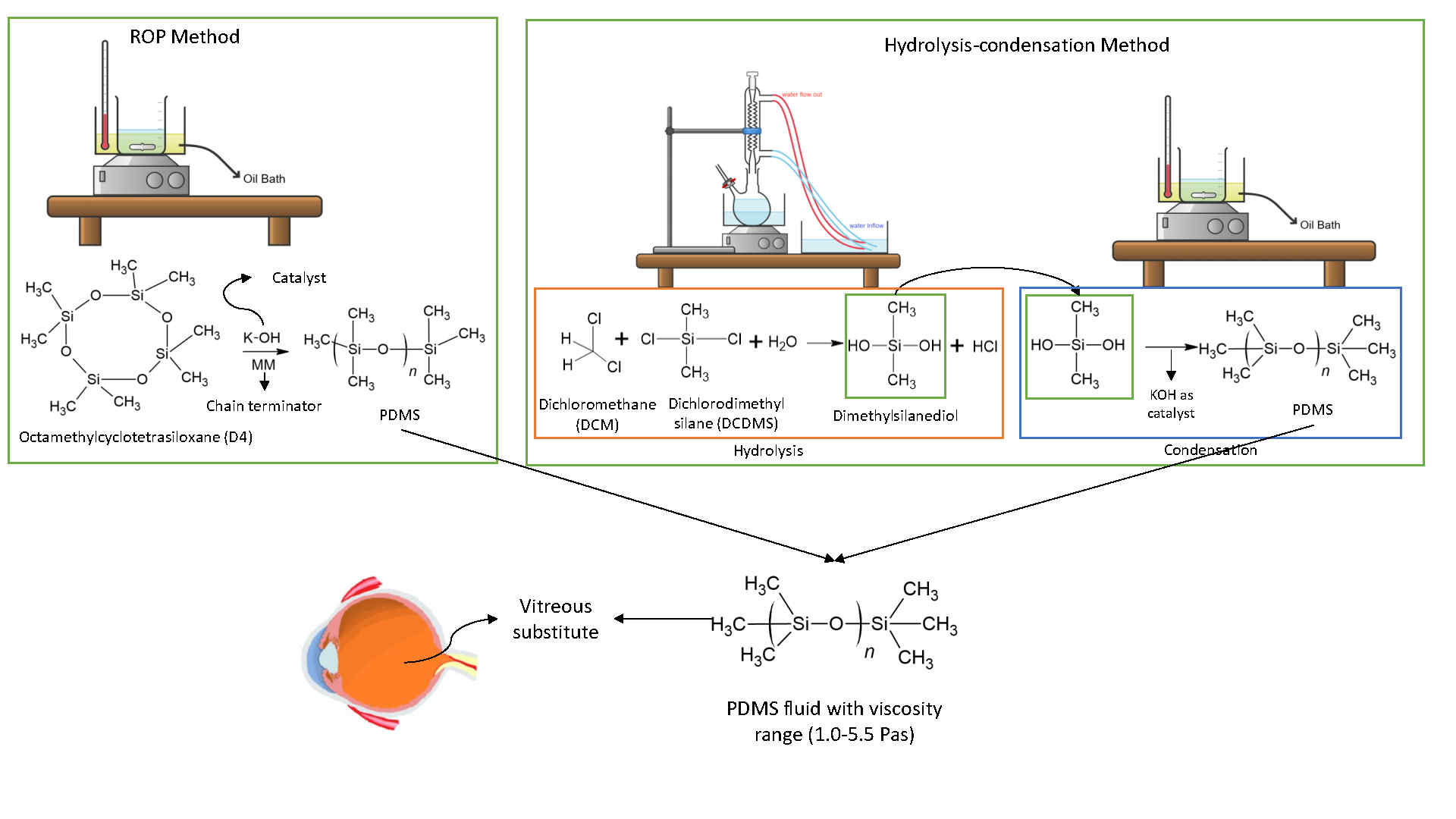
Polydimethylsiloxane (PDMS) is a polymer with outstanding biocompatibility, chemical stability, gas permeability, and optical clarity properties. It is suitable for various applications, including as a vitreous substitute in retinal surgery. PDMS provides optical stability and transparency, essential for maintaining adhesion between the retina and the retinal pigment epithelium. The main methods for synthesizing PDMS are ring-opening polymerization (ROP) and hydrolysis-condensation, both of which produce PDMS with a wide range of properties that can be tailored by adjusting the synthesis parameters. While ROP is typically performed with cyclic siloxanes such as octamethylcyclotetrasiloxane (D4), hydrolysis-condensation uses precursors such as dichlorodimethylsilane (DCDMS). This study compares the ROP and hydrolysis-condensation methods for PDMS synthesis, focusing on their effectiveness in achieving a specific viscosity range, a key parameter for vitreous replacement applications. The effectiveness of each method in achieving specific viscosity ranges for vitreous substitute applications is compared. The results indicate that ROP requires only 1 synthesis step and operates at higher temperatures (150 - 200 °C), yielding PDMS with viscosities ranging from 0.58 to 9.36 Pa·s, influenced by factors such as KOH concentration, reaction time, and D4 purity. In contrast, hydrolysis-condensation involves a 2-step process with viscosities between 0.57 and 4.49 Pa·s, affected by more complex parameters, including the DCDMS:DCM volume ratio, reaction time, and temperature during hydrolysis. Both methods achieved close refractive index values (1.3989 - 1.4048), although the surface tension varied with sample viscosity (19 - 23 mN/m). Comparative analysis reveals that although ROP is more suitable for producing high-purity PDMS efficiently. These findings contribute to understanding PDMS synthesis methods and their implications for vitreous substitute applications. Recommendations for future research on this topic include the need to conduct further biocompatibility tests such as in vivo tests and stability tests of PDMS samples.
HIGHLIGHTS
- Polydimethylsiloxane (PDMS) stands out for its biocompatibility, chemical stability, optical clarity, and gas permeability, making it ideal for vitreous substitute applications in retinal surgery.
- PDMS serves as an intraocular tamponade agent, offering optical clarity, inertness, and maintaining adhesion between the retina and retinal pigment epithelium, mimicking native vitreous properties.
- The study compares ring-opening polymerization (ROP) and hydrolysis-condensation methods, enabling tunable PDMS properties for specific applications.
- Efficient synthesis of high-performance PDMS tailored for biomedical applications.
GRAPHICAL ABSTRACT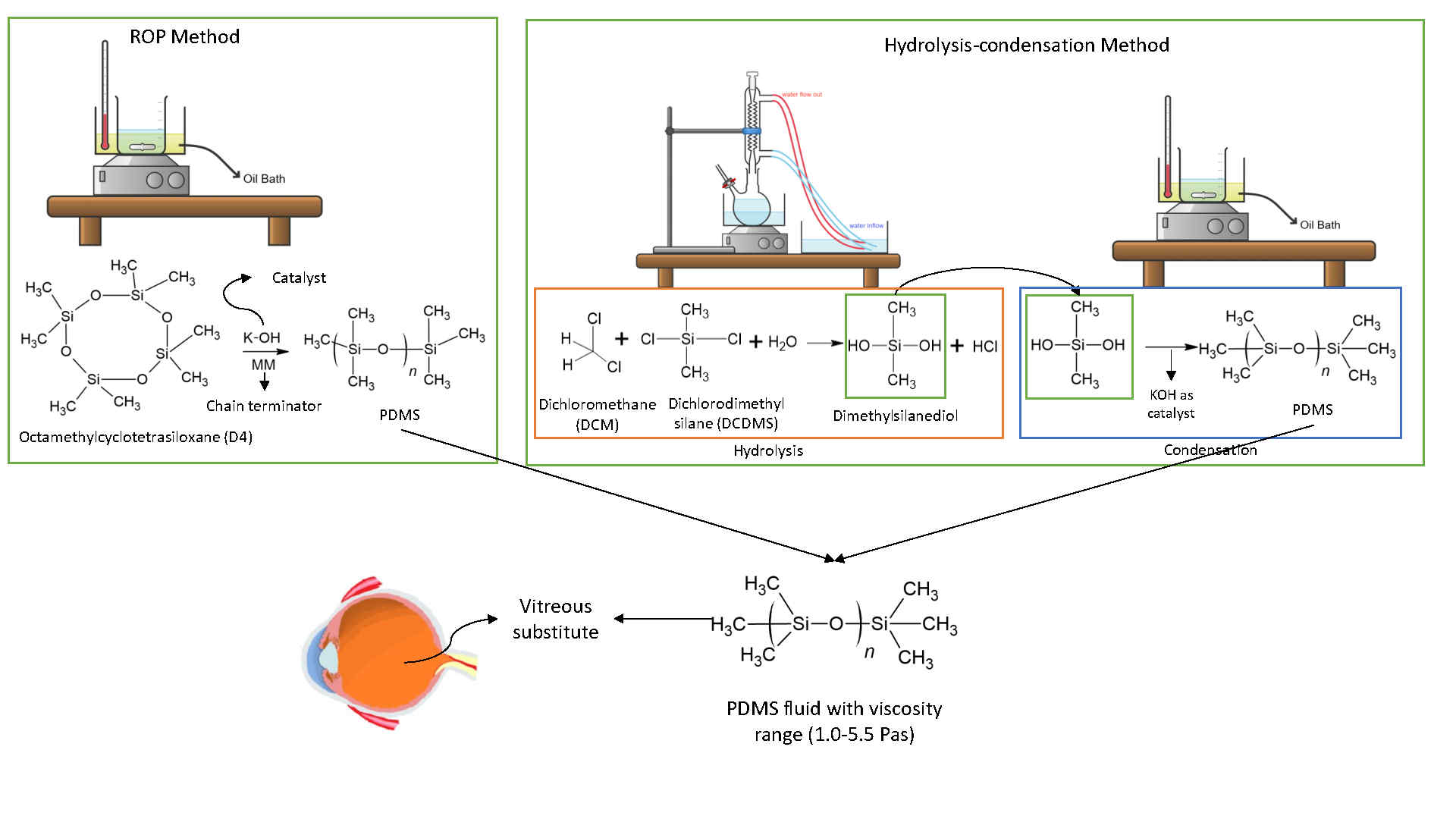
Downloads
References
I Miranda, A Souza, P Sousa, J Ribeiro, EMS Castanheira, R Lima and G Minas. Properties and applications of PDMS for biomedical engineering: A review. Journal of Functional Biomaterials 2022; 13(1), 2.
K Mojsiewicz-Pieńkowska, M Jamrógiewicz, K Szymkowska and D Krenczkowska. Direct human contact with siloxanes (silicones) - safety or risk part 1. Characteristics of siloxanes (silicones). Frontiers in Pharmacology 2016; 7, 132.
TT Kleinberg, RT Tzekov, L Stein, N Ravi and S Kaushal. Vitreous substitutes: A comprehensive review. Survey of Ophthalmology 2011; 56(4), 300-323.
H Hu, S Li, C Ying, R Zhang, Y Li, W Qian, L Zheng, X Fu, Q Liu, S Hu and CP Wong. Hydrophilic PDMS with a sandwich-like structure and no loss of mechanical properties and optical transparency. Applied Surface Science 2020; 503, 144126.
AK Khurana. Comprehensive ophthalmology. 4th ed. New Age International, New Delhi, India, 2007.
QY Gao, Y Fu and YN Hui. Vitreous substitutes: Challenges and directions. International Journal of Ophthalmology 2015; 8(3), 437.
M Thacker, CL Tseng and FH Lin. Substitutes and colloidal system for vitreous replacement and drug delivery: Recent progress and future prospective. Polymers 2020; 13(1), 121.
S Donati, SM Caprani, G Airaghi, R Vinciguerra, L Bartalena, F Testa, C Mariotti, G Porta, F Simonelli and C Azzolini. Vitreous substitutes: The present and the future. BioMed Research International 2014; 2014(1), 351804.
C Alovisi, C Panico, U Sanctis and CM Eandi. Vitreous substitutes: Old and new materials in vitreoretinal surgery. Journal of Ophthalmology 2017; 2017(1), 3172138.
MR Romano, X Xu and KKW Li. Vitreous substitutes: From tamponade effect to intraocular inflammation. BioMed Research International 2014; 2014, 159832.
X Su, MJ Tan, Z Li, M Wong, L Rajamani, G Lingam and XJ Loh. Recent progress in using biomaterials as vitreous substitutes. Biomacromolecules 2015; 16(10), 3093-3102.
R Ariati, F Sales, A Souza, RA Lima and J Ribeiro. Polydimethylsiloxane composites characterization and its applications: A review. Polymers 2021; 13(23), 4258.
KA Bezlepkina, SA Milenin, NG Vasilenko and AM Muzafarov. Ring-opening polymerization (ROP) and catalytic rearrangement as a way to obtain siloxane mono- and telechelics, as well as well-organized branching centers: History and prospects. Polymers 2022; 14(12), 2408.
S Penczek, J Pretula and S Slomkowski. Ring-opening polymerization. Chemistry Teacher International 2021; 3(2), 33-57.
ZG Coban, HC Kiliclar and Y Yagci. Photoinitiated cationic ring-opening polymerization of octamethylcyclotetrasiloxane. Molecules 2023; 28(3), 1299.
T Köhler, A Gutacker and E Mejía. Industrial synthesis of reactive silicones: Reaction mechanisms and processes. Organic Chemistry Frontiers 2020; 7(24), 4108-4120.
A Kartasasmita, W Kusdiono, R Virgana and S Boesorie. In vivo emulsification analysis of 1,000 cs and 5,000 cs silicone oil after rhegmatogenous retinal detachment vitrectomy surgery. Open Journal of Ophthalmology 2017; 7(04), 231.
HJ Cleaves. Hydrolysis. In: M Gargaud, WM Irvine, R Amils, P Claeys, HJ Cleaves, M Gerin, D Rouan, T Spohn, S Tirard and M Viso (Eds.). Encyclopedia of astrobiology. Springer, Berlin, Germany, 2011.
B Raval and I Banerjee. Functionalized graphene nanocomposite in gas sensing. In: M Jawaid, R Bouhfid and AK Qaiss (Eds.). Functionalized graphene nanocomposites and their derivatives. Elsevier, Amsterdam, The Netherlands, 2019.
DG Auliya, U Fauziah, VF Arini, S Setiadji, F Fitrilawati, AS Kartasasmita and R Risdiana. Use of dichlorodimethylsilane to produce polydimethylsiloxane as a substitute for vitreous humour: Characteristics and in vitro toxicity. Journal of Functional Biomaterials 2023; 14(8), 425.
S Sikdar and S Majumdar. Reactive silicones as multifacetic materials. In: TJ Gutiérrez (Ed.). Reactive and functional polymers volume one: Biopolymers, polyesters, polyurethanes, resins and silicones. Springer International Publishing, Cham, Switzerland, 2020.
Q Zaman, KM Zia, M Zuber, YN Mabkhot, F Almalki and TB Hadda. A comprehensive review on synthesis, characterization, and applications of polydimethylsiloxane and copolymers. International Journal of Plastics Technology 2019; 23, 261-282.
DG Auliya, S Setiadji, F Fitrilawati and R Risdiana. Physical characterization and in vitro toxicity test of PDMS synthesized from low-grade D4 monomer as a vitreous substitute in the human eyes. Journal of Functional Biomaterials 2022; 13(1), 3.
O Nuyken and SD Pask. Ring-opening polymerization - an introductory review. Polymers 2013; 5(2), 361-403.
J Chojnowski. Kinetically controlled siloxane ring-opening polymerization. Journal of Inorganic and Organometallic Polymers 1991; 1, 299-323.
L Shi, A Boulègue-Mondière, D Blanc, A Baceiredo, V Branchadell and T Kato. Ring-opening polymerization of cyclic oligosiloxanes without producing cyclic oligomers. Science 2023; 381(6661), 1011-1014.
M Barrere, F Ganachaud, D Bendejacq, MA Dourges, C Maitre and P Hémery. Anionic polymerization of octamethylcyclotetrasiloxane in miniemulsion II. Molar mass analyses and mechanism scheme. Polymer 2001; 42(17), 7239-7246.
S Penczek, M Cypryk, A Duda, P Kubisa and S Słomkowski. Living ring-opening polymerizations of heterocyclic monomers. Progress in Polymer Science 2007; 32(2), 247-282.
J Currie, P Griffith, W Herron and R Taylor. 1999, Process for producing a silicone polymer, U.S. Patent 6,054,548.
JF Berbert and W Herbert. 1948, Production of organosiloxane, U.S. Patent 2,443,353.
DG Auliya, S Setiadji, ZM Agasa, F Fitrilawati, N Syakir and R Risdiana. Synthesis of low viscosity polydimethylsiloxane using low grade of octamethylcyclotetrasiloxane. Materials Science Forum 2021; 1028, 365-370.
S Setiadji, ZM Agasa, DG Auliya, F Fitrilawati, N Syakir, AR Noviyanti, I Rahayu, A Supriadin and R Risdiana. Synthesis and characterization of polydimethylsiloxane (PDMS) with medium viscosity via ring-opening polymerization. Materials Science Forum 2021; 1028, 346-351.
S Setiadji, F Fitrilawati, AN Fauza, A Ardi, R Novianti, N Syakir, W Waslaluddin, I Rahayu, AS Kartasasmita and R Risdiana. Optimization of polydimethylsiloxane synthesized parameters as vitreous humour substitutes. Materials Science Forum 2019; 966, 189-193.
AN Fauza, A Ardi, RM Novianti, N Syakir and AS Kartasasmita. Effect of KOH concentration on characteristics of polydimethylsiloxane synthesized by ring opening polymerization method. Journal of Physics: Conference Series 2018; 1080(1), 012016.
W Waslaluddin, W Wiendartun, S Setiadji, AT Rahayu and R Risdiana. Formulation, process, and scale-up engineering of silicone oil. Materials Science Forum 2021; 1028, 377-382.
RM Hill. Silicone surfactants. 1st ed. Routledge, New York, 1999.
Z Rappoport and Y Apeloig. The chemistry of organic silicon compounds. John Wiley & Sons, New York, 1998.
SJ Clarson, JJ Fitzgerald, MJ Owen and SD Smith. Silicones and silicone-modified materials. ACS Publications, Washington DC, 2000.
W Noll. Chemistry and technology of silicones. Elsevier Science, Amsterdam, The Netherlands, 2012.
JBP Soares and V Touloupidis. Polymerization kinetics and the effect of reactor residence time on polymer microstructure. In: AR Albunia, F Prades and D Jeremic (Eds.). Multimodal polymers with supported catalysts: Design and production. Springer International Publishing, Cham, Switzerland, 2019.
G Odian. Principles of polymerization. John Wiley & Sons, Chicago, 2004.
IH Malitson. Interspecimen comparison of the refractive index of fused silica. Journal of the Optical Society of America 1965; 55(10), 1205-1209.
SS Batsanov, ED Ruchkin, IA Poroshina, SS Batsanov, ED Ruchkin and IA Poroshina. Chemical bonding and refractive indices. In: SS Batsanov, ED Ruchkin and IA Poroshina (Eds.). Refractive indices of solids. Springer Singapore, 2016.
A Colas and J Curtis. 7 - silicones. In: K Modjarrad and SBT Ebnesajjad (Eds.). Plastics design library. William Andrew Publishing, Oxford, 2013.
FL Pedrotti, LM Pedrotti and LS Pedrotti. Introduction to optics. 3rd ed. Cambridge University Press, Cambridge, 2017.
P Mardina, HA Prathama and DM Hayati. Pengaruh waktu hidrolisis dan konsentrasi katalisator asam sulfat terhadap sintesis furfural dari jerami padi (in Indonesian). Konversi 2016; 3(2), 37-44.
J Chojnowski and M Cypryk. Synthesis of linear polysiloxanes. In: RG Jones, W Ando and J Chojnowski (Eds.). Silicon-containing polymers: The science and technology of their synthesis and applications. Springer, Dordrecht, The Netherlands, 2000.
M Kar, Y Chourasiya, R Maheshwari and RK Tekade. Chapter 2 - current developments in excipient science: Implication of quantitative selection of each excipient in product development. In: R Tekade (Ed.). Advances in pharmaceutical product development and research. Academic Press, New York, 2019.
D Seyferth. Dimethyldichlorosilane and the direct synthesis of methylchlorosilanes. The key to the silicones industry. Organometallics 2001; 20(24), 4978-4992.
B Zhuang, G Ramanauskaite, ZY Koa and ZG Wang. Like dissolves like: A first-principles theory for predicting liquid miscibility and mixture dielectric constant. Science Advances 2024; 7(7), eabe7275.
CAM Afonso, NR Candeias, DP Simão, AF Trindade, JAS Coelho, B Tan and R Franzén. Comprehensive organic chemistry experiments for the laboratory classroom. Royal Society of Chemistry, London, 2016.
Y Wang. 2015, Synthesis, characterization and surface modification of synthesis, characterization and surface modification of polydimethylsiloxane and its composites polydimethylsiloxane and its composites. Ph. D. Dissertation. University of Massachusetts Amherst, United States.
PJ Flory. Fundamental principles of condensation polymerization. Chemical Reviews 1946; 39(1), 137-197.
S Fakirov. Condensation polymers: Their chemical peculiarities offer great opportunities. Progress in Polymer Science 2019; 89, 1-18.
U Fauziah, D Sandi, VF Arini, DG Aulia and S Setiadji. Synthesis of polydimethylsiloxane with hydrolysis and condensation methods using monomer of dichlorodimethylsilane as vitreous humour substitute. Journal of Physics: Conference Series 2022; 2165(1), 012026.
W Steiling, M Bracher, P Courtellemont and O De Silva. The HET-CAM, a useful in vitro assay for assessing the eye irritation properties of cosmetic formulations and ingredients. Toxicology in Vitro 1999; 13(2), 385-384.
P Budai, J Lehel, J Tavaszi and É Kormos. HET-CAM test for determining the possible eye irritancy of pesticides. Acta Veterinaria Hungarica 2010; 58(3), 369-377.
P Budai, É Kormos, I Buda, G Somody and J Lehel. Comparative evaluation of HET-CAM and ICE methods for objective assessment of ocular irritation caused by selected pesticide products. Toxicology In Vitro 2021; 74, 105150.
FA Barile. Validating and troubleshooting ocular in vitro toxicology tests. Journal of Pharmacological and Toxicological Methods 2010; 61(2), 136-145.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






