Development and Applications of a Kinetic Model for Resveratrol Production in Escherichia coli Cell Factories
DOI:
https://doi.org/10.48048/tis.2025.9529Keywords:
Kinetic model, Resveratrol, Cell factory, E. coliAbstract
Resveratrol is a compound with significant medical and nutraceutical benefits, offering great economic value due to its diverse therapeutic effects. Traditional production of resveratrol from plants is limited by slow growth rates and challenges in maintaining consistent purity. Heterologous production in microbial systems has emerged as a promising alternative. Here, we developed a chemical kinetic model to improve our understanding of the factors influencing resveratrol production in engineered Escherichia coli. While computational models have been widely used in microbial cell factories, most focus on flux balance analyses, which are useful for identifying gene knockout targets and optimizing media. However, these models often overlook enzyme expression levels and kinetic parameters, which are critical for optimizing production. Our model specifically incorporates these parameters, providing a more detailed understanding of how changes in enzyme kinetics and expression affect production outcomes. Our model successfully explained experimental observations from our engineered E. coli strain, and other reported results in the literature. Despite limited data, we demonstrate that the model narrows down possible ranges for key kinetic parameters such as stilbene synthase expression level, especially when resveratrol production is high. Further validation and refinement of the model will enhance its predictive power, aiding the design of future microbial cell factories for resveratrol and other polyphenolic compounds.
HIGHLIGHTS
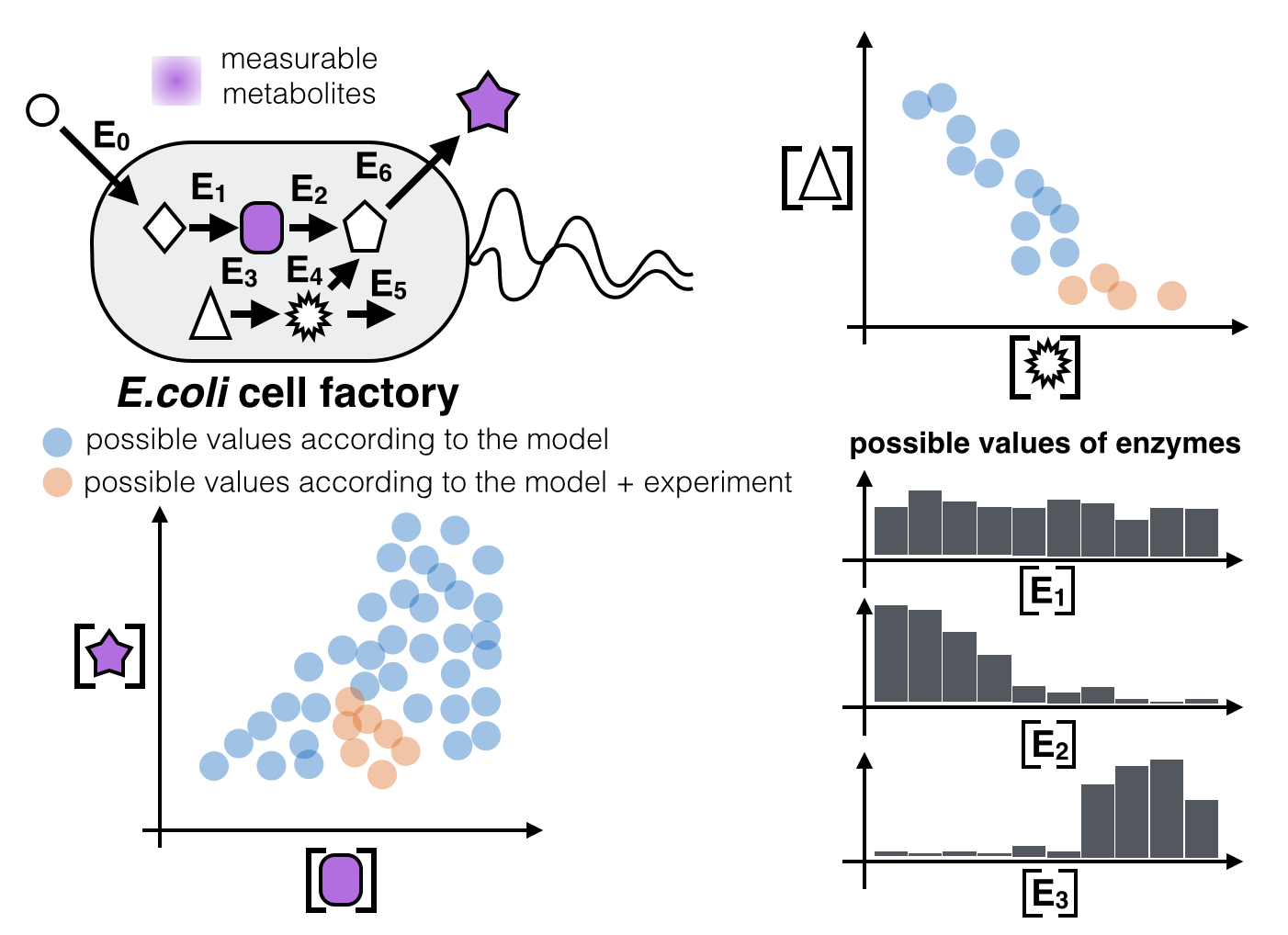
- We built a kinetic model for resveratrol production in engineered coli.
- The model predicts possible ranges of resveratrol outputs and intermediate metabolites.
- Experimental results help narrow down possible ranges of intermediate metabolites and enzyme expression levels.
- Higher resveratrol productivity corresponds to narrower ranges of possible enzyme expression levels.
GRAPHICAL ABSTRACT
Downloads
References
AP Singh, R Singh, SS Verma, V Rai, CH Kaschula, P Maiti and SC Gupta. Health benefits of resveratrol: Evidence from clinical studies. Medicinal Research Reviews 2019; 39(5), 1851-1891.
X Meng, J Zhou, C Zhao, RY Gan and HB Li. Health benefits and molecular mechanisms of resveratrol: A narrative review. Foods 2020; 9(3), 340.
Future Market Insights. Global Resveratrol Market is projected to reach US$ 278.3 million by 2033 at a moderate CAGR of 8 %, Available at: https://www.globenewswire.com/news-release/2023/05/09/2664312/0/en/Global-Resveratrol-Market-is-projected-to-reach-US-278-3-Million-by-2033-at-a-moderate-CAGR-of-8-Get-In-depth-Report-by-Future-Market-Insights-Inc.html, accessed December 2024.
B Tian and J Liu. Resveratrol: A review of plant sources, synthesis, stability, modification and food application. Journal of the Science of Food and Agriculture 2020; 100(4), 1392-1404.
C Feng, J Chen, W Ye, K Liao, Z Wang, X Song and M Qiao. Synthetic biology-driven microbial production of resveratrol: Advances and perspectives. Frontiers in Bioengineering and Biotechnology 2022; 10, 833920.
J Beekwilder, R Wolswinkel, H Jonker, R Hall, CH De Rie Vos and A Bovy. Production of resveratrol in recombinant microorganisms. Applied and Environmental Microbiology 2006; 72(8), 5670-5672.
M Cvijovic, S Bordel and J Nielsen. Mathematical models of cell factories: Moving towards the core of industrial biotechnology. Microbial Biotechnology 2011; 4(5), 572-584.
N Bhan, P Xu, O Khalidi and MAG Koffas. Redirecting carbon flux into malonyl-CoA to improve resveratrol titers: Proof of concept for genetic interventions predicted by OptForce computational framework. Chemical Engineering Science 2013; 103, 109-114.
H Kuwahara, M Alazmi, X Cui and X Gao. MRE: A web tool to suggest foreign enzymes for the biosynthesis pathway design with competing endogenous reactions in mind. Nucleic Acids Research 2016; 44(1), W217-W225.
J Hong, DK Im and MK Oh. Investigating E. coli coculture for resveratrol production with 13C metabolic flux analysis. Journal of Agricultural and Food Chemistry 2020; 68(11), 3466-3473.
M Cotner, J Zhan and Z Zhang. A computational metabolic model for engineered production of resveratrol in Escherichia coli. ACS Synthetic Biology 2021; 10(8), 1992-2001.
S Hoops, S Sahle, R Gauges, C Lee, J Pahle, N Simus, M Singhal, L Xu, P Mendes and U Kummer. COPASI - A complex pathway simulator. Bioinformatics 2006; 22(24), 3067-3074.
KE Brechun, D Zhen, A Jaikaran, V Borisenko, M Kumauchi, WD Hoff, KM Arndt and GA Woolley. Detection of incorporation of p-coumaric acid into photoactive yellow protein variants in vivo. Biochemistry 2019; 58(23), 2682-2694.
KT Watts, PC Lee and C Schmidt-Dannert. Biosynthesis of plant-specific stilbene polyketides in metabolically engineered Escherichia coli. BMC Biotechnology 2006; 6, 22.
BD Bennett, EH Kimball, M Gao, R Osterhout, SJ Van Dien and JD Rabinowitz. Absolute metabolite concentrations and implied enzyme active site occupancy in Escherichia coli. Nature Chemical Biology 2009; 5(8), 593-599.
J Wu, P Zhou, X Zhang and M Dong. Efficient de novo synthesis of resveratrol by metabolically engineered Escherichia coli. Journal of Industrial Microbiology and Biotechnology 2017; 44(7), 1083-1095.
JM Camacho-Zaragoza, G Hernández-Chávez, F Moreno-Avitia, R Ramírez-Iñiguez, A Martínez, F Bolívar and G Gosset. Engineering of a microbial coculture of Escherichia coli strains for the biosynthesis of resveratrol. Microbial Cell Factories 2016; 15, 163.
NJ Linden, B Kramer and P Rangamani. Bayesian parameter estimation for dynamical models in systems biology. PLoS Computational Biology 2022; 18(10), e1010651.
MD Lynch. The bioprocess TEA calculator: An online technoeconomic analysis tool to evaluate the commercial competitiveness of potential bioprocesses. Metabolic Engineering 2021; 65, 42-51.
CE Costa, A Romaní and L Domingues. Overview of resveratrol properties, applications, and advances in microbial precision fermentation. Critical Reviews in Biotechnology 2024. https://doi.org/10.1080/07388551.2024.2424362
A Arias, CE Costa, G Feijoo, MT Moreira and L Domingues. Process modeling, environmental and economic sustainability of the valorization of whey and eucalyptus residues for resveratrol biosynthesis. Waste Management 2023; 172, 226-234.
A Arias, CE Costa, MT Moreira, G Feijoo and L Domingues. Environmental and techno-economic assessment on the valorization of vine-side streams to produce resveratrol. Journal of Cleaner Production 2023; 429, 139622.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






