Role of Blood and Urine-Based Novel Biomarkers in Cervical Cancer Detection, Screening, Prognosis: Current Advances and Future Directions
DOI:
https://doi.org/10.48048/tis.2025.9522Keywords:
Cervical cancer, Non-invasive biomarkers, Blood based biomarker, Urine based biomarkers, Early detectionAbstract
Cervical cancer (CC) is one of the most common cancers among women worldwide, and early detection is crucial step for increasing survival rates. Blood and urine-based biomarkers provide non-invasive, very sensitive detection of cervical cancer, allowing for earlier diagnosis and surveillance than traditional procedures such as the Pap test or visual inspection with acetic acid (VIA). These biomarkers enhance precision by detecting molecular alterations, lowering false negatives, and facilitating large-scale screening systems. This review focuses on recent advances in non-invasive biomarkers for prediction and diagnosis of cervical cancer. This review analyzes a decade of literature on blood- and urine-based noninvasive biomarkers for cervical cancer detection, screening, prognosis emphasizing recent advancements, innovative approaches, and emerging prospects in biomarker applications to enhance diagnostic precision and effectiveness.
HIGHLIGHTS
- Blood and urine biomarkers are more sensitive and scalable than traditional methods for detecting cervical cancer.
- Advancements in proteomics, mass spectrometry, and miRNA analysis enable early detection and surveillance of cervical cancer utilizing biomarkers including SCC-Ag, CA-125, and cfDNA.
- Urine-based biomarkers such miRNA-34a-5p, MMRN1, and LRG1 have promising sensitivity and specificity, making them suitable for cost-effective cervical cancer screening.
- Omics technologies facilitate the discovery of biomarkers, leading to the creation of personalized cervical cancer treatments.
- Combining blood and urine biomarkers with modern diagnostic methods improves accuracy and lowers false positives, enabling integrated cervical cancer detection and surveillance.
GRAPHICAL ABSTRACT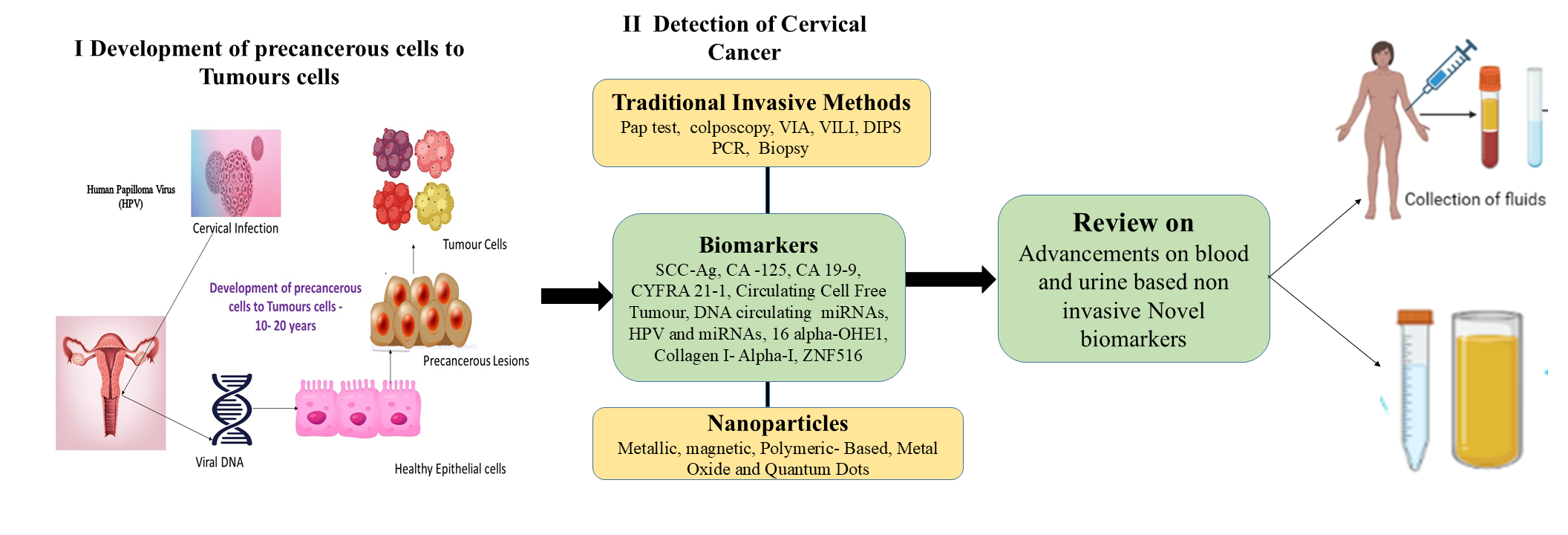
Downloads
References
WHO. Cervical cancer elimination initiative: From call to action to global movement. World Health Organization, Geneva, Switzerland, 2023.
P Duangkaew, S Tapaneeyakorn, C Apiwat, T Dharakul, S Laiwejpithaya, P Kanatharana and R Laocharoensuk. Ultrasensitive electrochemical immunosensor based on dual signal amplification process for p16ink4a cervical cancer detection in clinical samples. Biosensors and Bioelectronics 2015; 74, 673-679.
M Salvatici, MT Achilarre, MT Sandri, S Boveri, Z Vanna and F Landoni. Squamous cell carcinoma antigen (SCC-Ag) during follow-up of cervical cancer patients: Role in the early diagnosis of recurrence. Gynecologic Oncology 2016; 142(1), 115-119.
N Lekskul, C Charakorn, A Lertkhachonsuk, S Rattanasiri and NIN Ayudhya. The level of squamous cell carcinoma antigen and lymph node metastasis in locally advanced cervical cancer. Asian Pacific Journal of Cancer Prevention 2015; 16(11), 4719-4722.
Q Li, S Liu, H Liu, J Zhang, S Guo and L Wang. Significance and implication on changes of serum squamous cell carcinoma antigen in the diagnosis of recurrence squamous cell carcinoma of cervix. Zhonghua Fu Chan Ke Za Zhi 2015; 50(2), 131-136.
Y Bocheva, P Bochev, B Chaushev and S Ivanov. Squamous cell carcinoma (SCC)–nature and usage in patients with cervical carcinoma. Akusherstvo I Ginekologiia 2015; 54(2), 29-34.
K Al-Musalhi, M Al-Kindi, F Ramadhan, T Al-Rawahi, K Al-Hatali and WA Mula-Abed. Validity of cancer antigen-125 (CA-125) and risk of malignancy index (RMI) in the diagnosis of ovarian cancer. Oman Medical Journal 2015; 30(6), 428-434.
H Pradjatmo. Impact of preoperative serum levels of CA 125 on epithelial ovarian cancer survival. Asian Pacific Journal of Cancer Prevention 2016; 17(4), 1881-1886.
PJ Hoskins, N Le and R Correa. CA 125 normalization with chemotherapy is independently predictive of survival in advanced endometrial cancer. Gynecologic Oncology 2011; 120(1), 52-55.
T Jiang, L Huang and S Zhang. Preoperative serum CA125: A useful marker for surgical management of endometrial cancer. BMC Cancer 2015; 15(1), 396.
S Du, Y Zhao, C Lv, M Wei, Z Gao and X Meng. Applying serum proteins and microrna as novel biomarkers for early-stage cervical cancer detection. Scientific Reports 2020; 10(1), 9033.
LV Volkova, AI Pashov and NN Omelchuk. Cervical carcinoma: Oncobiology and biomarkers. International Journal of Molecular Sciences 2021; 22(22), 12571.
AM Mazurek, A Fiszer-Kierzkowska, T Rutkowski, K Skladowski, M Pierzyna, D Scieglinska, G Wozniak, G Glowacki, R Kawczynski and E Malusecka. Optimization of circulating cell-free DNA recovery for kras mutation and HPV detection in plasma. Cancer Biomarkers 2013; 13(5), 385-394.
JH Ginkel, MM Huibers, RJJV Es, RD Bree and SM Willems. Droplet digital PCR for detection and quantification of circulating tumor DNA in plasma of head and neck cancer patients. BMC Cancer 2017; 17(1), 428.
KR Dahlstrom, G Li, CS Hussey, JT Vo, Q Wei, C Zhao and EM Sturgis. Circulating human papillomavirus DNA as a marker for disease extent and recurrence among patients with oropharyngeal cancer. Cancer 2015; 121(19), 3455-3464.
L Liu, W Xie, P Xue, Z Wei, X Liang and N Chen. Diagnostic accuracy and prognostic applications of cyfra 21-1 in head and neck cancer: A systematic review and meta-analysis. PLoS One 2019; 14(5), e0216561.
E Peeters, N Wentzensen, C Bergeron and M Arbyn. Meta-analysis of the accuracy of p16 or p16/ki-67 immunocytochemistry versus HPV testing for the detection of CIN2+/CIN3+ in triage of women with minor abnormal cytology. Cancer Cytopathology 2019; 127(3), 169-180.
HZ Hausen. Papillomaviruses and cancer: From basic studies to clinical application. Nature Reviews Cancer 2002; 2(5), 342-350.
MA Molina, LC Diatricch, MC Quintana, MJ Melchers and KM Andralojc. Cervical cancer risk profiling: Molecular biomarkers predicting the outcome of HRHPV infection. Expert Review of Molecular Diagnostics 2020; 20(11), 1099-1120.
X Piao, T Kong, S Chang, J Paek, M chun and H Ryu. Pretreatment serum CYFRA 21-1 level correlates significantly with survival of cervical cancer patients: A Multivariate analysis of 506 cases. Gynecologic Oncology 2015; 138(1), 89-93.
A Chmura, A Wojcieszek, J Mrochem, A Walaszek-Gruska, R Deja, B Maslyk, W Bartnik and K Sodowski. Usefulness of the SCC, CEA, CYFRA 21.1, and CRP markers for the diagnosis and monitoring of cervical squamous cell carcinoma. Ginekologia Polska 2009; 80(5), 361-366.
E Pras, PH Willemse, AA Canrinus, HWAD Bruijn, WJ Sluiter, KAT Hoor, JG Aalders, BG Szabo and EGED Vries. Serum squamous cell carcinoma antigen and cyfra 21-1 in cervical cancer treatment. International Journal of Radiation Oncology Biology Physics. 2002; 52(1), 23-32.
Y Suzuki, T Nakano, T Ohno, A Abe, S Morita and H Tsujii. Serum CYFRA 21-1 in cervical cancer patients treated with radiation therapy. Journal of Cancer Research and Clinical Oncology 2000; 126(6), 332-336.
SP Mathur, RS Mathur, EA Gray, D Lane, PG Underwood, M Kohler and WT Creasman. Serum vascular endothelial growth factor C (VEGF-C) as a specific biomarker for advanced cervical cancer: Relationship to insulin-like growth factor II (IGF-II), IGF binding protein 3 (IGF-BP3) and VEGF-A [corrected]. Gynecologic Oncology 2005; 98(3), 467-483.
J An, Y Lee, JY Park, CH Choi, TJ Kim, J Lee, B Kim and D Bae. Prognostic value of pre-treatment SCC-AG level in patients with cervical cancer. Korean Journal of Obstetrics & Gynecology 2011; 54(8), 428-434.
R Fujiwaki, K Hata, M Moriyama, O Iwanari, H Katabuchi, H Okamura and K Miyazaki. Clinical value of thymidine kinase in patients with cervical carcinoma. Oncology 2001; 61(1), 47-54.
AG Gebrehiwot. 2019, Human serum n-glycans as highly sensitive cancer biomarkers: Potential benefits and the risks. Ph. D. Dissertation. Bahir Dar University, Ethiopia.
K Nustad, RCB Jr, TJ Brien, O Nilsson, P Seguin, MR Suresh, T Saga, S Nozawa, OP Børmer, HWD Bruijn, M Nap, A Vitali, M Gadnell, J Clark, K Shigemasa, B Karlsson, FT Kreutz, D Jette, H Sakahara, K Endo, ..., J Hilgers. Specificity and affinity of 26 monoclonal antibodies against the ca 125 antigen: First report from the isobmtd-1 workshop. International society for oncodevelopmental biology and medicine. Tumor Biology 1996; 17(4), 196-219.
L Mussolin, R Burnelli, M Pillon, E Carraro, P Farruggia, A Todesco, M mascarin and A Rosolen. Plasma cell-free DNA in pediatric lymphomas. Journal of Cancer 2013; 4(4), 323-329.
M Yanaranop, N Jantarateptewan, J Tiyayon and S Nakrangsee. Significance of serum human epididymis protein 4 and cancer antigen 125 in distinguishing type I and type II epithelial ovarian cancers. International Journal of Gynecological Cancer 2018; 28(6), 1058-1065.
GL Goldberg, A Sklar, KA O’Hanlan, PA Levine and CD Runowicz. CA-125: A potential prognostic indicator in patients with cervical cancer? Gynecologic Oncology 1990; 40(3), 222-224.
N Kim, W Park, WK Cho, DS Bae, BG Kim, JW Lee, CH Choi, TJ Kim and YY Lee. Significance of serum CA125 level in surgically resected cervical adenocarcinoma with adverse features. Journal of Gynecologic Oncology 2021; 32(5), e72.
C Ran, J Sun, Y Qu and N Long. Clinical value of MRI, serum SCCA, and CA125 levels in the diagnosis of lymph node metastasis and para-uterine infiltration in cervical cancer. World Journal of Surgical Oncology 2021; 19, 343.
SP Lee, IK Sung, JH Kim, SY Lee, HS Park and CS Shim. Usefulness of carbohydrate antigen 19-9 test in healthy people and necessity of medical follow-up in individuals with elevated carbohydrate antigen 19-9 level. Korean Journal of Family Medicine 2019; 40(5), 314-322.
Z Hernadi, L Gazdag, K Szoke, T Sapy, ZT Krasznai and J Konya. Duration of HPV-associated risk for high-grade cervical intraepithelial neoplasia. European Journal of Obstetrics & Gynecology and Reproductive Biology 2006; 125(1), 114-119.
LD Marco, A Gillio-Tos, L Bonello, V Ghisetti, G Ronco and F Merletti. Detection of human papillomavirus type 16 integration in pre-neoplastic cervical lesions and confirmation by dips-PCR and sequencing. Journal of Clinical Virology 2007; 38(1), 7-13.
H Shi, Y Ma, Y Shao, Y Zhang and B Lu. Cytology, high-risk human papillomavirus testing and serum CA19-9 in a large cohort of patients with invasive cervical adenocarcinomas: Correlation with a new pathogenetic classification. Asian Pacific Journal of Cancer Prevention 2022; 23(8), 2599.
W Pornthanakasem, K Shotelersuk, W Termrungruanglert, N Voravud, S Niruthisard and A Mutirangura. Human papillomavirus DNA in plasma of patients with cervical cancer. BMC Cancer 2001; 1, 2.
S Sharmin, M Jamiruddin, MR Jamiruddin, ABMMK Islam, CR Ahsan and M Yasmin. Detection of protein markers from blood samples of cervical cancer patients. Cureus 2024; 16(10), e72365.
B Kotowicz, M Fuksiewicz, J Jonska-Gmyrek, M Bidzinski and M Kowalska. The assessment of the prognostic value of tumor markers and cytokines as SCCAg, CYFRA 21.1, IL-6, VEGF, and sTNF receptors in patients with squamous cell cervical cancer, particularly with early stage of the disease. Tumor Biology 2016; 37(1), 1271-1278.
X Zhou, Y Wang, M Bao, Y Chu, R Liu, Q Chen and Y Lin. Advanced detection of cervical cancer biomarkers using engineered filamentous phage nanofibers. Applied Microbiology and Biotechnology 2024; 108(1), 221.
I Sidorkiewicz, M Zbucka-Krętowska, K Zaręba, E Lubowicka, M Zajkowska, M Szmitkowski, E Gacuta and S Lawicki. Plasma levels of M-CSF and VEGF in laboratory diagnostics and differentiation of selected histological types of cervical cancers. BMC Cancer 2019; 19(1), 398.
I Piskur, Z Topolovec, M Bakula, I Zagorac, IM Vranjes and D Vidosavljevic. Expression of vascular endothelial growth factor-A (VEGF-A) in adenocarcinoma and squamous cell cervical cancer and its impact on disease progression: Single institution experience. Medicina 2023; 59(7), 1189.
KS Tewari, MW Sill, Hj Long, RT Penson, H Huang, LM Ramondetta, LM Landrum, A Oaknin, TJ Reid, MM Leitao, HE Michael and BJ Monk. Improved survival with bevacizumab in advanced cervical cancer. The New England Journal of Medicine 2014; 370(8), 734-743.
Y Kang, H Li, Y Liu and Z Li. Regulation of VEGF-A expression and VEGF-A-targeted therapy in malignant tumors. Journal of Cancer Research and Clinical Oncology 2024; 150, 221.
YY Lui, KW Chik, RW Chiu, CY Ho, CW Lam and YD Lo. Predominant hematopoietic origin of cell-free DNA in plasma and serum after sex-mismatched bone marrow transplantation. Clinical Chemistry 2002; 48(3), 421-427.
SA Leon, B Shapiro, D Sklaroff and MJ Yaros. Free DNA in the serum of cancer patients and the effect of therapy. Cancer Research 1977; 37(3), 646-650.
J Tian, Y Geng, D Lv, P Li, M Cordova, Y Liao, X Tian, X Zhang, Q Zhang, K Zou, Y Zhang, X Zhang, Y Li, J Zhang, Z Ma, Y Shao, L Song, GI Owen, T Li, R Liu, ..., Z Li. Using plasma cell-free DNA to monitor the chemoradiotherapy course of cervical cancer. International Journal of Cancer 2019; 145(9), 2547-2557.
PJ Vellanki, S Ghosh, A Pathak, MJ Fusco, EW Bloomquist, S Tang, H Singh, R Philip, R Pazdur and JA Beaver. Regulatory implications of ctDNA in immuno-oncology for solid tumors. Journal for ImmunoTherapy of Cancer 2023; 11(2), e005344.
RC Lee, RL Feinbaum and V Ambros. The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell 1993; 75(5), 843-854.
JR Lytle, TA Yario and JA Steitz. Target mRNAs are repressed as efficiently by microRNA-binding sites in the 5′UTR as in the 3′UTR. Proceedings of the National Academy of Sciences 2007; 104(25), 9667-9672.
N Yamamoto, T Kinoshita, N Nohata, T Itesako, H Yoshino, H Enokida, M Nakagawa, M Shozu and N Seki. Tumor suppressive microRNA-218 inhibits cancer cell migration and invasion by targeting focal adhesion pathways in cervical squamous cell carcinoma. International Journal of Oncology 2013; 42(5), 1523-1532.
MA Kepsha, AV Timofeeva, VS Chernyshev, DN Silachev, EA Mezhevitinova and GT Sukhikh. MicroRNA-based liquid biopsy for cervical cancer diagnostics and treatment monitoring. International Journal of Molecular Sciences 2024; 25(24), 13271.
D Patel, S Thankachan, S Sreeram, KP Kavitha, SP Kabekkodu and PS Suresh. LncRNA-miRNA-mRNA regulatory axes as potential biomarkers in cervical cancer: A comprehensive overview. Molecular Biology Reports 2025; 52, 110.
E Girardi, P Lopez and S Pfeffer. On the importance of host microRNAs during viral infection. Frontiers in Genetics 2018; 9, 439.
Z Zheng and X Wang. Regulation of cellular miRNA expression by human papillomaviruses. Biochimica et Biophysica Acta (BBA) - Gene Regulatory Mechanisms 2011; 1809(11-12), 668-677.
S Norouzi, A Farhadi, E Farzadfard, M Akbarzade-Jahromi, N Ahmadzadeh, M Nasiri and G Tamaddon. MicroRNAs expression changes coincide with low or high grade of squamous intraepithelial lesion infected by HPV-16. Gene Reports 2021; 23, 101186.
J Nunvar, L Pagacova, Z Vojtechova, NTD Azevedo, J Smahelova, M Salakova and R Tachezy. Lack of conserved miRNA deregulation in HPV-induced squamous cell carcinomas. Biomolecules 2021; 11(5), 764.
CJ Meijer, H Berkhof, DA Heideman, AT Hesselink and PJ Snijders. Validation of high-risk HPV tests for primary cervical screening. Journal of Clinical Virology 2009; 46(S3), S1-S4.
S Weng, K Huang, JT Weng, F Hung, T Chang and T Lee. Genome-wide discovery of viral microRNAs based on phylogenetic analysis and structural evolution of various human papillomavirus subtypes. Briefings in Bioinformatics 2017; 19(6), 1102-1114.
V Tony, A Sathyamurthy, JK Ramireddy, SJ Iswarya, SM Gowri, A Thomas, A Peedicayil and TS Ram. Role of squamous cell carcinoma antigen in prognostication, monitoring of treatment response, and surveillance of locally advanced cervical carcinoma. Journal of Cancer Research and Therapeutics 2023; 19(5), 1236-1240.
L Galati, J Combes, FL Calvez-Kelm, S McKay-Chopin, N Forey M Ratel, J McKay, T Waterboer, L Schroeder, G Clifford, M Tommasino and T Gheit. Detection of Circulating HPV16 DNA detection by a bead-based HPV assay. Microbiology Spectrum 2022; 10(2), e0148021.
S Du, Y Zhao, C Lv, M Wei, Z Gao and X Meng. Applying serum proteins and microRNA as novel biomarkers for early-stage cervical cancer detection. Scientific Reports 2020; 10, 9033.
G Ma, G Song, X Zou, X Shan, Q Liu, T Xia, X Zhou and W Zhu. Circulating plasma microRNA signature for the diagnosis of cervical cancer. Cancer Biomarkers 2019; 26(4), 491-500.
J Oh and JY Bae. Optimal cutoff level of serum squamous cell carcinoma antigen to detect recurrent cervical squamous cell carcinoma during post-treatment surveillance. Obstetrics & Gynecology Science 2018; 61(3), 337-343.
S Park, K Eom, J Kim, H Bang, HY Wang, S Ahn, G Kim, H Jang, S Kim, D Lee, KH Park and H Lee. MiR-9, miR-21, and miR-155 as potential biomarkers for HPV positive and negative cervical cancer. BMC Cancer 2017; 17(1), 658.
J Oh, HJ Lee, TS Lee, JH Kim, SB Koh and YS Choi. Clinical value of routine serum squamous cell carcinoma antigen in follow-up of patients with locally advanced cervical cancer treated with radiation or chemoradiation. Obstetrics & Gynecology Science 2016; 59, 269-278.
HK Ryu, JS Baek, WD Kang and SM Kim. The prognostic value of squamous cell carcinoma antigen for predicting tumor recurrence in cervical squamous cell carcinoma patients. Obstetrics & Gynecology Science 2015; 58, 368-376.
B Tang, S Liu, Y Zhan, L Wei, X Mao, J Wang, L Li and Z Lu. MicroRNA-218 expression and its association with the clinicopathological characteristics of patients with cervical cancer. Experimental and Therapeutic Medicine 2015; 10, 269-274.
Q Ma, G Wan, S Wang, W Yang, J Zhang and X Yao. Serum microRNA-205 as a novel biomarker for cervical cancer patients. Cancer Cell International 2014; 14, 81.
R Kawaguchi, N Furukawa, H Kobayashi and I Asakawa. Posttreatment cut-off levels of squamous cell carcinoma antigen as a prognostic factor in patients with locally advanced cervical cancer treated with radiotherapy. Journal of Gynecologic Oncology 2013; 24(4), 313-320.
S Zhao, D Yao, J Chen and N Ding. Circulating miRNA-20a and miRNA-203 for screening lymph node metastasis in early stage cervical cancer. Genetic Testing and Molecular Biomarkers 2013; 17, 631-636.
J Cox, M Hein, C Luber, I Paron, N Nagaraj and M Mann. Accurate proteome-wide label-free quantification by delayed normalization and maximal peptide ratio extraction, termed MaxLFQ. Molecular & Cellular Proteomics 2014; 13(9), 2513-2526.
JWH Wong and G Cagney. An overview of label-free quantitation methods in proteomics by mass spectrometry. Methods in Molecular Biology 2010; 604, 273-283.
JM An, J Suh, J Kim, Y Kim, JY Chung, HS Kim, SY Cho, JH Ku, C Kwak, HH Kim, CW Jeong and D kim. First-in-class: Cervical cancer diagnosis based on a urine test with fluorescent cysteine probe. Sensors and Actuators B: Chemical 2022; 360, 131646.
H Zhang, E Tian, Y Chen, H Deng and Q Wang. Proteomic analysis for finding serum pathogenic factors and potential biomarkers in multiple myeloma. Chinese Medical Journal 2015; 128(8), 1108-1113.
S Xu, H Xu, W Wang, S Li, H Li, T Li, W Zhang, X Yu and L Liu. The role of collagen in cancer: From bench to bedside. Journal of Translational Medicine 2019; 17(1), 309.
T Chan, A Poon, A Basu, N Addleman, J Chen, A Phong, PH Byers, TE Klein and P Kwok. Natural variation in four human collagen genes across an ethnically diverse population. Genomics 2008; 91(4), 307-314.
Y Zhao, H Wang, M Fang, Q Ji, Z Yang and C Gao. Study of the association between polymorphisms of the COL1A1 gene and HBV-related liver cirrhosis in Chinese patients. Digestive Diseases and Sciences 2009; 54(2), 369-376.
JC Marini, A Forlino, WA Cabral, AM Barnes, JDS Antonio, S Milgrom, JC Hyland, J Körkkö, DJ Prockop, AD Paepe, P Coucke, S Symoens, FH Glorieux, PJ Roughley, AM Lund, K Kuurila-Svahn, H Hartikka, DH Cohn, D Krakow, M Mottes, ..., PH Byers. Consortium for osteogenesis imperfecta mutations in the helical domain of type I collagen: Regions rich in lethal mutations align with collagen binding sites for integrins and proteoglycans. Human Mutation 2007; 28(3), 209-221.
A Li, J Si, Y Shang, L Gan, L Guo and T Zhou. Construction of COL1A1 short hairpin RNA vector and its effect on cell proliferation and migration of gastric cancer cells. Zhejiang Da Xue Xue Bao Yi Xue Ban 2010; 39(3), 257-263.
O Kitahara, T Katagiri, T Tsunoda, Y Harima and Y Nakamura. Classification of sensitivity or resistance of cervical cancers to ionizing radiation according to expression profiles of 62 genes selected by cDNA microarray analysis. Neoplasia 2002; 4(4), 295-303.
X Wang, N Cui, X Liu, J Ma, Q Zhu, S Guo, J Zhao and L Ming. Identification of DAPK1 promoter hypermethylation as a biomarker for intraepithelial lesion and cervical cancer: A meta-analysis of published studies, TCGA, and GEO datasets. Frontiers in Genetics 2018; 9, 258.
Q Feng, A Balasubramanian, SE Hawes, P Toure, PS Sow, A Dem, B Dembele, CW Critchlow, L Xi, H Lu, MW Mclntosh, AM Yong and NB Kiviat. Detection of hypermethylated genes in women with and without cervical neoplasia. Journal of the National Cancer Institute 2005; 97(4), 273-282.
BK Prusty, A Kumar, R Arora, S Batra and BC Das. Human papillomavirus (HPV) DNA detection in self-collected urine. International Journal of Gynecology & Obstetrics 2005; 90(3), 223-227.
S Liu, G Liao and G Li. Regulatory effects of COL1A1 on apoptosis induced by radiation in cervical cancer cells. Cancer Cell International 2017; 17, 73.
NC Garbett, ML Merchant, CW Helm, AB Jenson, JB Klein and JB Chaires. Detection of cervical cancer biomarker patterns in blood plasma and urine by differential scanning calorimetry and mass spectrometry. PLoS One 2014; 9(1), e84710.
Q Wang. Collagen type I alpha 1 contributes to radioresistance in cervical cancer by promoting DNA damage repair. Cancer Cell International 2017; 17, 44.
S Sharmin, M Jamiruddin, MR Jamiruddin, ABMMK Islam, CR Ahsan and M Yasmin. Detection of protein markers from blood samples of cervical cancer patients. Cureus 2024; 16(10), e72365.
J Li. Epigenetic biomarkers in cervical cancer: Sensitivity and specificity of promoter methylation. Clinical Epigenetics 2023; 15, 122.
W Chen. TLBA test in human papillomavirus genotyping and cancer risk assessment. Cancer Research Journal 2022; 18(4), 412-423.
X Zhang. ZNF516 and FKBP6: Novel biomarkers in cervical cancer methylation studies. BMC Genomics 2022; 23, 45.
C Gao, C Zhou, J Zhuang, L Liu, C Liu, H Li, G Liu, J Wei and C Sun. MicroRNA expression in cervical cancer: Novel diagnostic and prognostic biomarkers. Journal of Cellular Biochemistry 2018; 119(8), 7080-7090.
P Speiser, C Wanner, C Tempfer, M Mittelbock, E Hanzal, D Bancher-Todesca, G Gitsch, A Reinthaller and C Kainz. CD44 is an independent prognostic factor in early-stage cervical cancer. International Journal of Cancer 1997; 74(2), 185-188.
JM An, M Jeong, J Jung, SG Yeo, S Park and D Kim. Next-generation Femtech: Urine-based cervical cancer diagnosis using a fluorescent biothiol probe with controlled Smiles rearrangement. ACS Applied Materials & Interfaces. 2024; 16(4), 4493-4504.
M Aftab, SS Poojary, V Seshan, S Kumar, P Agarwal, S Tandon, V Zutshi and BC Das. Urine miRNA signature as a potential non-invasive diagnostic and prognostic biomarker in cervical cancer. Scientific Reports 2021; 11, 10323.
CM Oliveira, LW Musselwhite, NDP Pantano, FL Vazquez, JS Smith, J Schweizer, M Belmares, JC Possati-Resende, MDA Vieira, A Longatto-Fiho and JHTG Fregnani. Detection of HPV E6 oncoprotein from urine via a novel immunochromatographic assay. PLoS One 2020; 15(4), e0232105.
BC Snoek, APV Splunter, MCG Bleeker, MCV Ruiten, DAM Heideman, WF Rurup, W Varlaat, H Schotman, MV Gent, NEV Trommel and RDM Steenbergen. Cervical cancer detection by DNA methylation analysis in urine. Scientific Reports 2019; 9, 3088.
A Leeman, MD Pino, A Molijn, A Rodriguez, A Torne, MD Koning, J Ordi, FV Kemenade, D Jenkins and W Quint. HPV testing in first-void urine provides sensitivity for CIN2+ detection comparable with a smear taken by a clinician or a brush-based self-sample: Cross-sectional data from a triage population. BJOG: An International Journal of Obstetrics & Gynaecology 2017; 124(9), 1356-1363.
J Cuzick, L Cadman, AS Ahmad, L Ho, G Terry, M Kleeman, D Lyons, J Austin, MH Stoler, CRT Vibat, J Dockter, D Robbins, PR Bilings and MG Erlander. Performance and diagnostic accuracy of a urine-based human papillomavirus assay in a referral population. Cancer Epidemiology, Biomarkers & Prevention 2017; 26(7), 1053-1059.
VV Sahasrabuddhe, PE Gravitt, ST Dunn, D Robbins, D Brown, RA Allen, YJ Eby, KM Smith, RE Zuna, RR Zhang, MA Gold, M Schiffman, JL Walker, PE Catle and N Wentzensen. Evaluation of clinical performance of a novel urine-based HPV detection assay among women attending a colposcopy clinic. Journal of Clinical Virology 2014; 60(4), 414-417.
D Chokchaichamnankit, K Watcharatanyatip, P Subhasitanont, C Weeraphan, S Keeratichamroen, N Sritana, N Kantathavorn, P Diskul-Na-Ayudthaya, K Saharat, J Chantaraamporn, C Verathamjamras, N Phoolcharoen, K Wiriyaukaradecha, NM Paricharttanakul, W Udomchaiprasertkul, T Sricharunrat, C Auewarakul, J Svasti and C Srisomsap. Urinary biomarkers for the diagnosis of cervical cancer by quantitative label-free mass spectrometry analysis. Oncology Letters 2019; 17(6), 5453-5468.
V Senkomago, ACD Marais, L Rahangdale, CR Vibat, MG Erlander and JS Smith. Comparison of urine specimen collection times and testing fractions for the detection of high-risk human papillomavirus and high-grade cervical precancer. Journal of Clinical Virology 2016; 74, 26-31.
A Ferencz, D Szatmári and D Lőrinczy. Thermodynamic sensitivity of blood plasma components in patients afflicted with skin, breast, and pancreatic forms of cancer. Cancers 2022; 14(24), 6147.
A Derbie, D Mekonnen, Y Woldeamanuel, XV Ostade and T Abebe. HPV E6/E7 mRNA test for the detection of high grade cervical intraepithelial neoplasia (CIN2+): A systematic review. Infect Agent Cancer 2020; 15, 9.
C Ren, Y Zhu, L Yang, X Zhang, L Liu, Z Wang and D Jiang. Prognostic and diagnostic validity of p16/Ki-67, HPV E6/E7 mRNA, and HPV DNA in women with ASCUS: A follow-up study. Virology Journal 2019; 16, 143.
Y Zhu, C Ren, L Yang, X Zhang, L Liu and Z Wang. Performance of p16/Ki67 immunostaining, HPV E6/E7 mRNA testing, and HPV DNA assay to detect high-grade cervical dysplasia in women with ASCUS. BMC Cancer 2019; 19(1), 271.
S Singh, A Sarin, M Jain, A Purkayastha, D Shelly and N Bisht. Role of squamous cell carcinoma antigen (SCC-Ag) in the management of carcinoma cervix in an Indian population: A pilot study. Elsevier Science, Amsterdam, Netherland, 2024.
H Zhu. Squamous cell carcinoma antigen: Clinical application and research status. Diagnostics 2022; 12(5), 1065.
M Farzanehpour, S Mozhgani, S Jalilvand, E Faghihloo, S Akhavan, V Salimi, TM Azad. Serum and tissue miRNAs: Potential biomarkers for the diagnosis of cervical cancer. Virology Journal 2019; 16, 116.
S Park, K Eom, J Kim, H Bang, H Wang, S Ahn, G Kim, H Jang, S Kim, D Lee, KH Park and H Lee. MiR-9, miR-21, and miR-155 as potential biomarkers for HPV positive and negative cervical cancer. BMC Cancer 2017; 17, 658.
ML Tornesello, R Faraonio, L Buonaguro, C Annunziata, N Starita, A Cerasuolo, F Pezzuto, AL Tornesello and FM Buonaguro. The role of microRNAs, long non-coding RNAs, and circular RNAs in cervical cancer. Frontiers in Oncology 2020; 10, 150.
Y Jiang, Z Hu, Z Zuo, Y Li, F Pu, B Wang, Y Tang, Y Guo and H Tao. Identification of circulating microRNAs as a promising diagnostic biomarker for cervical intraepithelial neoplasia and early cancer: A meta-analysis. BioMed Research International 2020; 2020, 4947381.
CE Condrat, DC Thompson, MG Barbu, OL Bugnar, A Boboc, D Cretoiu, N Suciu, SM Cretoiu and SC Voinea. miRNAs as biomarkers in disease: Latest findings regarding their role in diagnosis and prognosis. Cells 2020; 9(2), 276.
M Schmitz, K Eichelkraut, D Schmidt, I Zeiser, Z Hilal, Z Tettenborn, A Hansel and H Ikenberg. Performance of a DNA methylation marker panel using liquid-based cervical scrapes to detect cervical cancer and its precancerous stages. BMC Cancer 2018; 18(1), 1197.
L Kong, L Wang, Z Wang, X Xia, Y You, H Wu, M Wu, P Liu and L Li. DNA methylation for cervical cancer screening: A training set in China. Clinical Epigenetics 2020; 12, 91.
L Dong, L Zhang, S Hu, R Feng, X Zha, Q Zhang, Q Pan, X Zhang, Y Qiao and F Zhao. Risk stratification of HPV 16 DNA methylation combined with E6 oncoprotein in cervical cancer screening: A 10-year prospective cohort study. Clinical Epigenetics 2020; 12, 62.
RWV Leeuwen, A Ostrbenk, M Poljak, AGVD Zee, E Schuuring and GBA Wisman. DNA methylation markers as a triage test for identification of cervical lesions in a high risk human papillomavirus positive screening cohort. International Journal of Cancer 2019; 144(4), 746-754.
LMAD Strooper, VMJ Verhoef, J Berkhof, AT Hesselink, HMED Bruin, FJV Kemenade, RP Bosgraaf, RLM Bekkers, LFAG Massuger, WJG Melchers, RDM Steenbergen, PJF Snijders, CJLM Meijer and DAM Heideman. Validation of the FAM19A4/mir124–2 DNA methylation test for both lavage- and brush-based self-samples to detect cervical (pre) cancer in HPV-positive women. Gynecology Oncology 2016; 141(2), 341-347.
S Chujan, N Kitkumthorn, S Siriangku and A Mutirangura. CCNA1 promoter methylation: A potential marker for grading Papanicolaou smear cervical squamous intraepithelial lesions. Asian Pacific Journal of Cancer Prevention 2014; 15(8), 7971-7975.
C Kottaridi, M Kyrgiou, A Pouliakis, M Magkana, E Aga, A Spathis, A Mitra, G Makris, C Chrelias, V Mpakou, E Paraskevaidis, JG Panayiotides and P Karakitsos. Quantitative measurement of L1 human papillomavirus type 16 methylation for the prediction of Preinvasive and invasive cervical disease. The Journal of Infectious Diseases 2017; 215(5), 764-771.
A Leeman, MD Pino, L Marimon, A Torne, J Ordi, BT Harmsel, CJLM Meijer, D Jenkins, FJVV Kemenade and WGV Quint. Reliable identification of women with CIN3+ using hrHPV genotyping and methylation markers in a cytology-screened referral population. International Journal of Cancer 2019; 144(1), 160-168.
Z Piškur, M Topolovec, I Bakula, I Zagorac, M Vranješ and D Vidosavljević. Expression of vascular endothelial growth factor-A (VEGF-A) in adenocarcinoma and squamous cell cervical cancer and its impact on disease progression: single institution experience. Medicina 2023; 59(7), 1189.
S Lawicki, M Zajkowska, EK Glazewska, GE Będkowska and M Szmitkowski. Plasma levels and diagnostic utility of VEG F, MMP-9, and TIMP-1 in the diagnosis of patients with breast cancer. Onco Targets and Therapy 2016; 9, 911-919.
D Cheng, B Liang and Y Li. Serum vascular endothelial growth factor (VEGF-C) as a diagnostic and prognostic marker in patients with ovarian cancer. PLoS One 2013; 8(2), e55309.
V Urquidi, S Goodison, J Kim, M Chang, Y Daib and CJ Rosser. VEGF, CA9 and angiogenin as a urinary biomarker for bladder cancer detection. Urology 2012; 79(5), 1185.e1-1185.e6.
C Ceci, MG Atzori, PM Lacal and G Graziani. Role of VEGFs/VEGFR-1 signaling and its inhibition in modulating tumor invasion: Experimental evidence in different metastatic cancer models. International Journal of Molecular Sciences 2020; 21(4), 1388.
PLM Zusterzeel, PN Span, MGK Dijksterhuis, CMG Thomas, FCGJ Sweep and LFAG Massuger. Serum vascular endothelial growth factor: A prognostic factor in cervical cancer. Journal of Cancer Research and Clinical Oncology 2009; 135(2), 283-290.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






