The Oleaginous Rhodotorula mucilaginosa Isolated from Paddy Field Soils and the Profile of Fatty Acid in Its Intracellular Microbial Oils
DOI:
https://doi.org/10.48048/tis.2025.9470Keywords:
Fatty acid, Microbial oil, Oleaginous yeast, Rhodotorula mucilaginosa, Single-cell oil, Thailand, Paddy field soil, Soil-yeast oilAbstract
Oleaginous yeasts are a specific group of yeast species known for their ability to accumulate significant amounts of intracellular microbial oils, also referred to as single-cell oils, which constitute more than 20 % of their dry cell weight. These microbial oils represent a promising resource for various biotechnological and biofuel applications. However, only a few known yeast species have been classified as oleaginous. This study focused on isolating and identifying oleaginous yeasts from the soil in paddy fields across 3 provinces in Thailand. The yeasts that were isolated were assessed for their intracellular lipid accumulation using a quantitative Sudan IV staining and a qualitative weighting method. Among the isolated strains, the one with the highest lipid accumulation was considered for its fatty acid composition using gas chromatography. The results revealed that 6 strains of yeast were isolated from the paddy field soil samples, and these were designated as oleaginous, with intracellular lipid content ranging from 20.41 ± 2.24 to 44.11 ± 3.27 % (w/w). Molecular genetic evaluations indicated that the six strains belonged to the genera Debaryomyces, Meyerozyma, Rhodotorula, and Starmerella. Notably, the R. mucilaginosa strain RYA07 exhibited the highest lipid accumulation, with its intracellular lipids primarily consisting of palmitic acid (C16:0), stearic acid (C18:0), and oleic acid (C18:1n9c). These fatty acids have potential applications in various biotechnological fields, including biodiesel production, food industry, cosmetics, and pharmaceuticals.
HIGHLIGHTS
- Soils from paddy fields collected in 3 provinces of Thailand were used to isolate soil-derived oleaginous yeasts.
- Six strains of these soil yeasts were identified as oleaginous, each containing more than 20 % (w/w) of intracellular lipids.
- Some strains of these soil yeasts were newly recorded as oleaginous.
- Rhodotorula mucilaginosa strain RYA07 was found to accumulate 44.115 % (w/w) of intracellular lipids, which primarily consisted of palmitic acid (C16:0), stearic acid (C18:0), and oleic acid (C18:1n9c).
GRAPHICAL ABSTRACT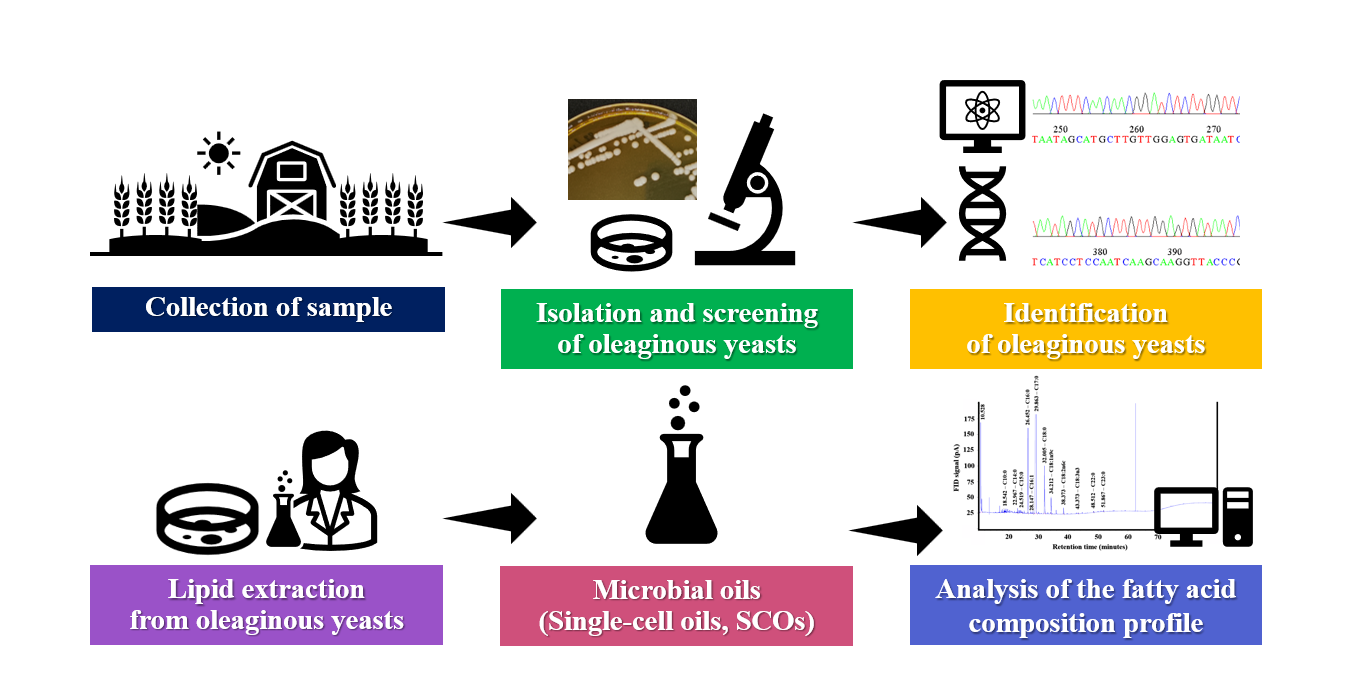
Downloads
References
S Maina, C Pateraki, N Kopsahelis, S Paramithiotis, EH Drosinos, S Papanikolaou and A Koutinas. Microbial oil production from various carbon sources by newly isolated oleaginous yeasts. Engineering in Life Sciences 2017; 17(3), 333-344.
S Planonth and A Chantarasiri. The oleaginous yeast Pichia manshurica isolated from Lansium domesticum fruit in Thailand and its fatty acid composition of single cell oil. Biodiversitas 2022; 23(2), 801-809.
S Ugur, B Zieniuk and A Fabiszewska. Nutritional and medicinal properties of microbial oil. Applied Sciences 2024; 14(10), 4232.
A Patel, D Karageorgou, E Rova, P Katapodis, U Rova, P Christakopoulos and L Matsakas. An overview of potential oleaginous microorganisms and their role in biodiesel and omega-3 fatty acid-based industries. Microorganisms 2020; 8(3), 434.
F Abeln and CJ Chuck. The history, state of the art and future prospects for oleaginous yeast research. Microbial Cell Factories 2021; 20, 221.
R Poontawee, W Lorliam, P Polburee and S Limtong. Oleaginous yeasts: Biodiversity and cultivation. Fungal Biology Reviews 2023; 44, 100295.
S Sapsirisuk, P Polburee, W Lorliam and S Limtong. Discovery of oleaginous yeast from mountain forest soil in Thailand. Journal of Fungi 2022; 8(10), 1100.
M Vincent, MC Hung, PRM Baran, AS Azahari and DSA Adeni. Isolation, identification and diversity of oleaginous yeasts from Kuching, Sarawak, Malaysia. Biodiversitas 2018; 19(4), 1266-1272.
S Wongchamrearn, P Boontanom, S Ungwiwatkul, N Emnin and A Chantarasiri. Short communication: The oleaginous Candida tropicalis isolated from mangrove soil in eastern Thailand and the fatty acid composition profile of its intracellular lipids. Biodiversitas 2023; 24(9), 5088-5095.
S Kunthiphun, P Chokreansukchai, P Hondee, S Tanasupawat and A Savarajara. Diversity and characterization of cultivable oleaginous yeasts isolated from mangrove forests. World Journal of Microbiology and Biotechnology 2018; 34, 125.
I Schulze, S Hansen, S Großhans, T Rudszuck, K Ochsenreither, C Syldatk and A Neumann. Characterization of newly isolated oleaginous yeasts-Cryptococcus podzolicus, Trichosporon porosum and Pichia segobiensis. AMB Express 2014; 4, 24.
PE Diaz, C Aranda, O Martínez, R Godoy, A Gonzales and E Valenzuela. Characterization of yeast in hapludands soil with biotechnological potential. Journal of Soil Science and Plant Nutrition 2017; 17(4), 948-965.
P Diaz-Navarrete, L Marileo, H Madrid, C Belezaca-Pinargote and P Dantagnan. Lipid production from native oleaginous yeasts isolated from southern Chilean soil cultivated in industrial vinasse residues. Microorganisms 2023; 11(10), 2516.
K Amprayn, MT Rose, M Kecskes, L Pereg, HT Nguyen and IR Kennedy. Plant growth promoting characteristics of soil yeast (Candida tropicalis HY) and its effectiveness for promoting rice growth. Applied Soil Ecology 2012; 61, 295-299.
AM Yurkov. Yeasts of the soil-obscure but precious. Yeast 2018; 35(5), 369-378.
C Sripodok, A Thammasittirong and SN Thammasittiron. Antifungal activity of soil yeast (Lachancea kluyveri sp132) against rice pathogenic fungi and its plant growth promoting activity. Journal of the International Society for Southeast Asian Agricultural Sciences 2019; 25(1), 55-65.
L Xu, H Zhang, C Wang, S Wei, B Zhang, F Wu and Y Tang. Paddy rice mapping in Thailand using time-series Sentinel-1 data and deep learning model. Remote Sensing 2021; 13(19), 3994.
A Chantarasiri and S Ungwiwatkul. Effects of CO2 aeration and light supply on the growth and lipid production of a locally isolated microalga, Chlorella variabilis RSM09. Applied Sciences 2024; 14(22), 10512.
A Caporusso, A Capece and ID Bari. Oleaginous yeasts as cell factories for the sustainable production of microbial lipids by the valorization of agri-food wastes. Fermentation 2021; 7(2), 50.
AM Kot, S Błażejak, A Kurcz, J Brys, I Gientka, A Bzducha-Wróbel, M Maliszewska and L Reczek. Effect of initial pH of medium with potato wastewater and glycerol on protein, lipid and carotenoid biosynthesis by Rhodotorula glutinis. Electronic Journal of Biotechnology 2017; 27, 25-31.
TM Jiru, D Abate, N Kiggundu, C Pohl and Ma Groenewald. Oleaginous yeasts from Ethiopia. AMB Express 2016; 6, 78.
SE Kantar, A Khelfa, E Vorobiev and M Koubaa. Strategies for increasing lipid accumulation and recovery from Y. lipolytica: A review. Oilseeds & Fats Crops and Lipids 2021; 28, 51.
MR Capoor, S Aggarwal, C Raghvan, DK Gupta, AK Jain and R Chaudhary. Clinical and microbiological characteristics of Rhodotorula mucilaginosa infections in a tertiary‑care facility. Indian Journal of Medical Microbiology 2014; 32(3), 304-309.
P Hu, J Mao, Y Zeng, Z Sun, H Deng, C Chen, W Sun and Z Tang. Isolation, identification, and function of Rhodotorula mucilaginosa TZR2014 and its effects on the growth and health of weaned piglets. Frontiers in Microbiology 2022; 13, 922136.
Z Li, C Li, P Cheng and G Yu. Rhodotorula mucilaginosa-alternative sources of natural carotenoids, lipids, and enzymes for industrial use. Heliyon 2022; 8(11), e11505.
P Bardhan, K Gupta, S Kishor, P Chattopadhyay, C Chaliha, E Kalita, VV Goud and M Mandal. Oleaginous yeasts isolated from traditional fermented foods and beverages of Manipur and Mizoram, India, as a potent source of microbial lipids for biodiesel production. Annals of Microbiology 2020; 70, 27.
S Tsai, H Yu and C Lin. The Potential of the oil-producing oleaginous yeast Rhodotorula mucilaginosa for sustainable production of bio-oil energy. Processes 2022; 10(2), 336.
M Desnos-Ollivier, M Ragon, V Robert, D Raoux, JC Gantier and F Dromer. Debaryomyces hansenii (Candida famata), a rare human fungal pathogen often misidentified as Pichia guilliermondii (Candida guilliermondii). Journal of Clinical Microbiology 2008; 46(10), 3237-3242.
L Ramos-Moreno, F Ruiz-Perez, E Rodriguez-Castro and J Ramos. Debaryomyces hansenii is a real tool to improve a diversity of characteristics in sausages and dry-meat products. Microorganisms 2021; 9(7), 1512.
H Kumdam, SN Murthy and SN Gummadi. Production of ethanol and arabitol by Debaryomyces nepalensis: Influence of process parameters. AMB Express 2013; 3, 23.
L De Marco, S Epis, A Capone, E Martin, J Bozic, E Crotti, I Ricci and D Sassera. The genomes of four Meyerozyma caribbica isolates and novel insights into the Meyerozyma guilliermondii species complex. Genes Genomes Genetics 2018; 8(3), 755-759.
BRA Alencar, RAA de Freitas, VEP Guimaaes, RK Silva, C Elsztein, SPD Silva, ED Dutra, MADM Junior and RBD Souza. Meyerozyma caribbica isolated from vinasse-irrigated sugarcane plantation soil: A promising yeast for ethanol and xylitol production in biorefineries. Journal of Fungi 2023; 9(8), 789.
P Polburee, W Yongmanitchai, N Lertwattanasakul, T Ohashi, K Fujiyama and S Limtong. Characterization of oleaginous yeasts accumulating high levels of lipid when cultivated in glycerol and their potential for lipid production from biodiesel-derived crude glycerol. Fungal Biology 2015; 119(12), 1194-1204.
H Chebbi, D Leiva-Candia, M Carmona-Cabello, A Jaouani and MP Dorado. Biodiesel production from microbial oil provided by oleaginous yeasts from olive oil mill wastewater growing on industrial glycerol. Industrial Crops and Products 2019; 139, 111535.
C Nadai, VDS Duarte, J Sica, S Vincenzi, M Carlot, A Giacomini and V Corich. Starmerella bacillaris released in vineyards at different concentrations influences wine glycerol content depending on the vinification protocols. Foods 2023; 12(1), 3.
MLR Eder and AL Rosa. Genetic, physiological, and industrial aspects of the fructophilic non-Saccharomyces yeast species, Starmerella bacillaris. Fermentation 2021; 7(2), 87.
JMS Lopez, S Jezierska, AE Kocabey, J Lee, R Schneiter and INAV Bogaert. The oleaginous yeast Starmerella bombicola reveals limitations of Saccharomyces cerevisiae as a model for fatty acid transport studies. FEMS Yeast Research 2022; 22(1), foac054.
HW Y, YT Liao and YX Liu. Cultivation of oleaginous Rhodotorula mucilaginosa in airlift bioreactor by using seawater. Journal of Bioscience and Bioengineering 2016; 121(2), 209-212.
D Dasgupta, T Sharma, A Bhatt, S Bandhu and D Ghosh. Cultivation of oleaginous yeast Rhodotorula mucilaginosa IIPL32 in split column airlift reactor and its influence on fuel properties. Biocatalysis and Agricultural Biotechnology 2017; 10, 308-316.
EG Giakoumis. Analysis of 22 vegetable oils’ physicochemical properties and fatty acid composition on a statistical basis, and correlation with the degree of unsaturation. Renewable Energy 2018; 126, 403-419.
A Areesirisuk, CH Chiu, TB Yen, CH Liu and JH Guo. A novel oleaginous yeast strain with high lipid productivity and its application to alternative biodiesel production. Applied Biochemistry and Microbiology 2015; 51, 411-418.
G Carta, E Murru, S Banni and C Manca. Palmitic acid: Physiological role, metabolism and nutritional implications. Frontiers in Physiology 2017; 8, 902.
P Hiremath, K Nuguru and V Agrahari. Chapter 8-Material attributes and their impact on wet granulation process performance. Academic Press, Massachusetts, 2019.
C Carrillo, MDM Cavia and S Alonso-Torre. Role of oleic acid in immune system; mechanism of action; a review. Nutricion Hospitalaria 2012; 27(4), 978-990.
J Whelan and K Fritsche. Linoleic acid. Advances in Nutrition 2013; 4(3), 311-312.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






