Erythroxylum Cuneatum Enhanced Receptor Internalisation on Morphine-Desensitised Neuroblastoma Cells Line
DOI:
https://doi.org/10.48048/tis.2025.9453Keywords:
Erythroxylum cuneatum, G protein-coupled receptor kinase 2 (GRK 2), β-arrestin 1/2, Clathrin heavy chain, Chronic morphine, Methadone, Addiction, Morphine desensitisation, Receptor internalisationAbstract
Chronic morphine exposure disrupts receptor signalling by inhibiting receptor internalisation, leading to receptor desensitisation and reduced availability of active receptors. This process significantly contributes to morphine addiction, a condition with limited effective and accessible treatments. Current pharmacological interventions, such as methadone, face challenges including restricted availability, risk of misuse, and adverse effects, leaving a substantial treatment gap for individuals with opioid use disorder (OUD). To address this, alkaloid extract of Erythroxylum cuneatum (E. cuneatum) was investigated as a potential alternative treatment. Using the human neuroblastoma cell line (SK-N-SH), 2 experimental designs were employed: Antagonistic treatment and pre-treatment with morphine. The antagonistic design evaluated the direct interaction between morphine and E. cuneatum, while the pre-treatment model assessed its anti-addictive properties during morphine withdrawal. Methadone served as a positive control. Cytosolic protein fractions were analysed to determine the expression of G protein-coupled receptor kinase 2 (GRK2), β-arrestin 1/2, and clathrin heavy chain, essential for receptor internalisation and resensitisation. Morphine exposure significantly downregulated these proteins, indicating impaired receptor function. Treatment with the alkaloid extract of E. cuneatum reversed these effects, restoring protein levels to those comparable with methadone. These findings highlight the alkaloid extract of E. cuneatum as a promising candidate for mitigating morphine addiction by targeting the molecular mechanisms underlying receptor desensitisation and resensitisation. E. cuneatum addresses a critical gap in addiction management by providing a potential alternative therapy that overcomes some limitations of existing treatments.
HIGHLIGHTS
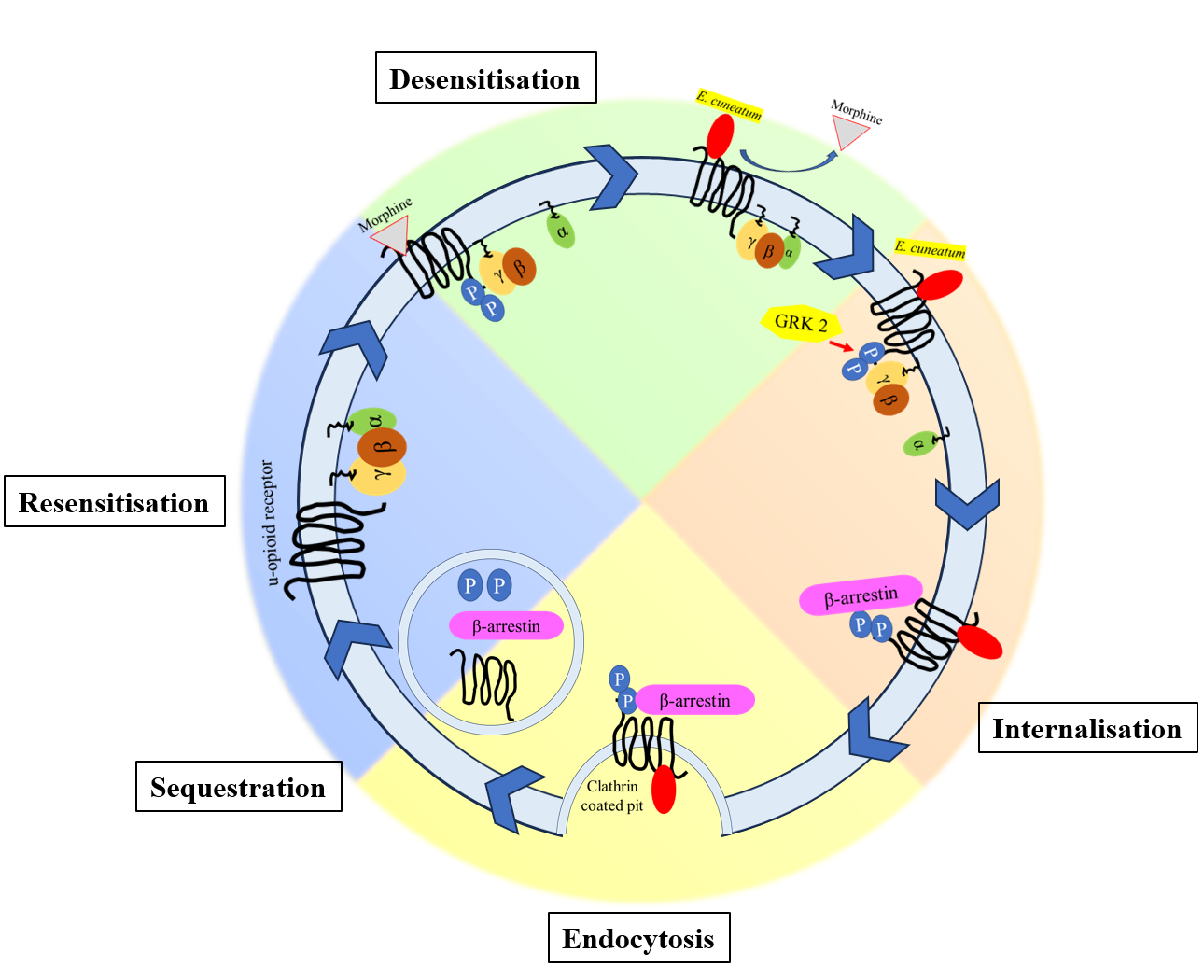
- Erythroxylum cuneatum alkaloid extract effectively counteracts morphine-induced receptor desensitisation and dysregulation in neuroblastoma cell lines.
- Dose-dependent restoration of GRK2, β-arrestin 1/2, and clathrin heavy chain by cuneatum highlights its role in promoting receptor internalisation and desensitisation.
- The antagonist effects of cuneatum are comparable to methadone, suggesting its potential as a natural alternative for opioid addiction treatment.
- The study proposes a novel mechanism of action for cuneatum, targeting the receptor trafficking machinery to restore μ-opioid receptor (MOR) function.
- cuneatum represents a promising plant-based therapeutic agent with lower dependency risks, offering a safer alternative to conventional opioid replacement therapies.
GRAPHICAL ABSTRACT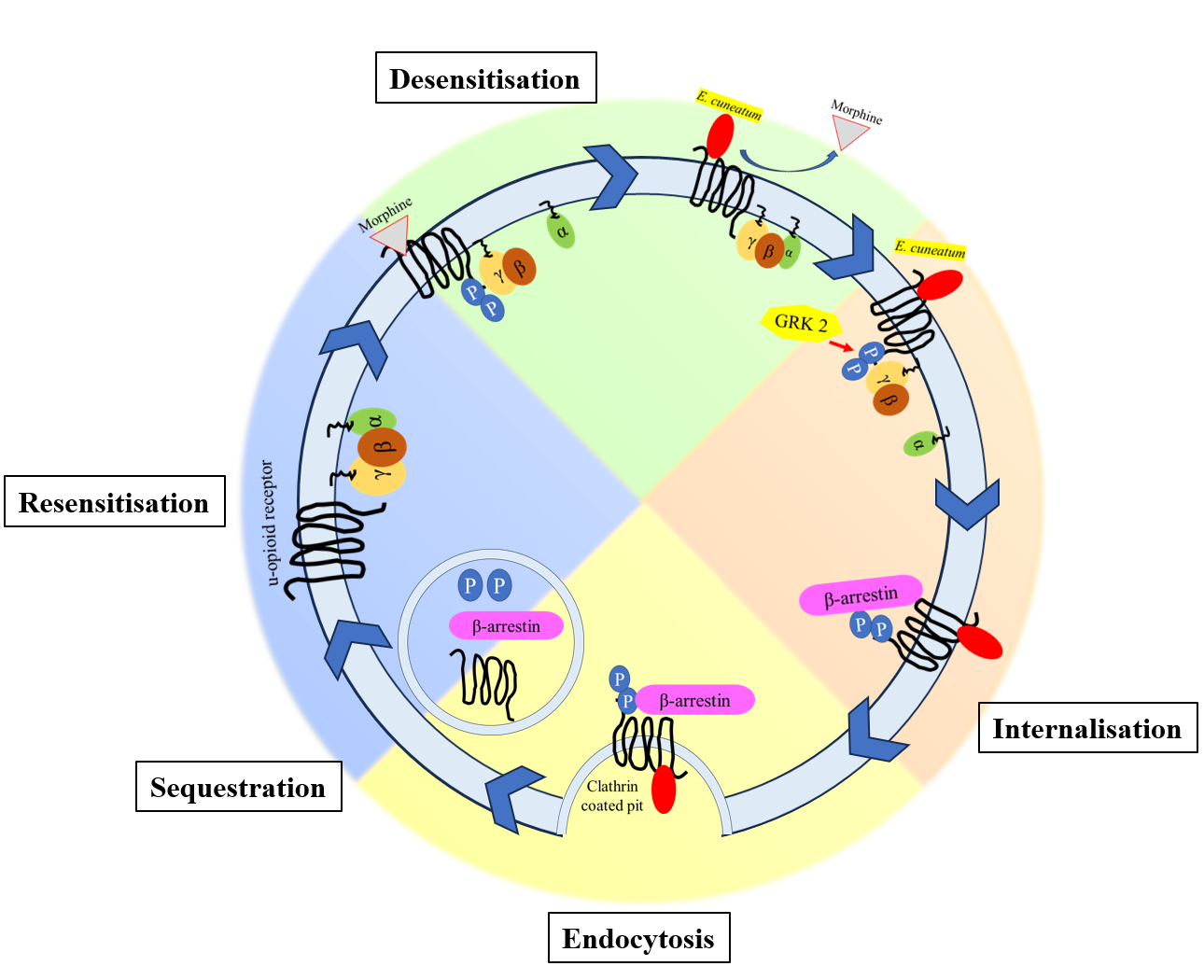
Downloads
References
SM Murphy. The cost of opioid use disorder and the value of aversion. Drug Alcohol Dependence 2020; 217(3), 108382.
E Gibula-Tarlowska and JH Kotlinska. Crosstalk between opioid and anti-opioid systems: An overview and its possible therapeutic significance. Biomolecules 2020; 10(10), 1376.
EM Lefevre, EA Gauthier, LL Bystrom, J Scheunemann and PE Rothwell. Differential patterns of synaptic plasticity in the nucleus accumbens caused by continuous and interrupted morphine exposure. Journal of Neuroscience 2023; 43(2), 308-318.
L Gopalakrishnan, O Chatterjee, N Ravishankar, S Suresh, R Raju, A Mahadevan and TK Prasad. Opioid receptors signaling network. Journal of Cell Communication and Signaling 2022; 16(3), 475-483.
ER Jaeckel, YN Herrera, S Schulz and WT Birdsong. Chronic morphine induces adaptations in opioid receptor signaling in a Thalamostriatal circuit that are location dependent, sex specific, and regulated by μ-opioid receptor phosphorylation. Journal of Neuroscience 2024; 44(3), e0293232023.
S Ide, T Hirai, T Muto, T Yamakawa and K Ikeda. Effects of the novel selective κ-opioid receptor agonist NP-5497-KA on morphine-induced reward-related behaviors. Scientific Reports 2023; 13(1), 18164.
D Jin, H Chen, Y Huang, SR Chen and HL Pan. δ-Opioid receptors in primary sensory neurons tonically restrain nociceptive input in chronic pain but do not enhance morphine analgesic tolerance. Neuropharmacology 2022; 217, 109202.
GA Sanchez, AV Smrcka and EM Jutkiewicz. Biasing Gβγ downstream signaling with Gallein inhibits development of morphine tolerance and potentiates morphine-induced nociception in a tolerant state. Molecular Pharmacology 2024; 106(1), 47-55.
J Zhou, R Ma, Y Jin, J Fang, J Du, X Shao, Y Liang and J Fang. Molecular mechanisms of opioid tolerance: From opioid receptors to inflammatory mediators. Experimental and Therapeutic Medicine 2021; 22(3), 1004.
CB Harper and KJ Smillie. Current molecular approaches to investigate pre‐synaptic dysfunction. Journal of Neurochemistry 2021; 157(2), 107-129.
JJ Zhang, CG Song, M Wang, GQ Zhang, B Wang, X Chen, P Lin, YM Zhu, ZC Sun, YZ Wang, JL Jiang, L Li, XM Yang and ZN Chen. Monoclonal antibody targeting mu-opioid receptor attenuates morphine tolerance via enhancing morphine-induced receptor endocytosis. Journal of Pharmaceutical Analysis 2023; 13(10), 1135-1152.
A Shiraki and S Shimizu. The molecular associations in clathrin-coated pit regulate β-arrestin-mediated MAPK signaling downstream of μ-opioid receptor. Biochemical and Biophysical Research Communications 2023; 640, 64-72.
TC Møller, MF Pedersen, JRV Senten, SD Seiersen, JM Mathiesen, M Bouvier and H Bräuner-Osborne. Dissecting the roles of GRK2 and GRK3 in μ-opioid receptor internalization and β-arrestin2 recruitment using CRISPR/Cas9-edited HEK293 cells. Scientific Reports 2020; 10(1), 17395.
O Underwood, S Fritzwanker, J Glenn, NK Blum, A Batista-Gondin, J Drube, C Hoffmann, SJ Briddon, S Schulz and M Canals. Key phosphorylation sites for robust β-arrestin2 binding at the MOR revisited. Communications Biology 2024; 7(1), 933.
Y Zhang, P Zhou, F Lu, R Su and Z Gong. A20-binding inhibitor of nuclear factor-κB targets β-arrestin2 to attenuate opioid tolerance. Molecular Pharmacology 2021; 100(2), 170-180.
JJ Manning, HM Green, M Glass and DB Finlay. Pharmacological selection of cannabinoid receptor effectors: Signalling, allosteric modulation and bias. Neuropharmacology 2021; 193(176), 108611.
LS Li, SM Chiroma, T Hashim, SK Adam, MAM Moklas, Z Yusuf and SA Rahman. Antioxidant and anti-inflammatory properties of Erythroxylum cuneatum alkaloid leaf extract. Heliyon 2020; 6(6), e04141.
Y Lv, T Tian, YJ Wang, JP Huang and SX Huang. Advances in chemistry and bioactivity of the genus Erythroxylum. Natural Products and Bioprospecting 2022; 12(1), 15.
SK Adam, N Shanmugam, S Mohamad, SA Rahman and MAM Moklas. In vitro antioxidant and anti-inflammatory effects of Erythroxylum Cuneatum leaf extract on oxidized low-density lipoprotein-stimulated human aortic endothelial cells. Pharmacognosy Magazine 2023; 19(2), 203-213.
NA Suliman, MAM Moklas, CNM Taib, MT Hidayat Baharuldin and MS Chiroma. Erythroxylum cuneatum prevented cellular adaptation in morphine induced neuroblastoma cells. Central Nervous System Agents in Medicinal Chemistry 2022; 22(2), 108-117.
NA Suliman, NAA Bakar, MAM Moklas and CNM Taib. Establishment of human neuroblastoma cell line (SK-N-SH) as an in vitro model of morphine addiction. Malaysian Applied Biology 2021; 50(2), 1-7.
L Wanka, V Behr and AG Beck-Sickinger. Arrestin-dependent internalization of rhodopsin-like G protein-coupled receptors. Biological Chemistry 2022; 403(2), 133-149.
S Singleton, C Sneddon, A Bakina, JJ Lambert and TG Hales. Early-life adversity increases morphine tolerance and persistent inflammatory hypersensitivity through upregulation of δ opioid receptors in mice. Pain 2023; 164(10), 2253-2264
S Adhikary. 2021, Adaptations following chronic opioid treatment in the presynaptic and postsynaptic compartments of neuron. Ph. D. Dissertation. Oregon Health & Science University, Oregon.
M Makvand, SD Mirtorabi, G Hassanzadeh, A Campbell and G Ahangari. The effectiveness of methadone maintenance therapy (MMT) on drug tolerance, mediated by receptor signaling, signal transduction and intracellular transport. Europe PMC 2022. https://doi.org/10.21203/rs.3.rs-1562550/v1
V Markova, L Hejnova, A Benda, J Novotny and B Melkes. β-Arrestin 1 and 2 similarly influence μ-opioid receptor mobility and distinctly modulate adenylyl cyclase activity. Cellular Signalling 2021; 87, 110124.
JJ Zhang, CG Song, JM Dai, L Li, XM Yang and ZN Chen. Mechanism of opioid addiction and its intervention therapy: Focusing on the reward circuitry and mu‐opioid receptor. MedComm 2022; 3(3), e148.
AN Edinoff, NW Wu, CA Nix, B Bonin, R Mouhaffel, S Vining and AD Kaye. Historical pathways for opioid addiction, withdrawal with traditional and alternative treatment options with ketamine, cannabinoids, and noribogaine: A narrative review. Health Psychology Research 2022; 10(4), 38672.
KL Prichard, NS O’Brien, SR Murcia, JR Baker and A McCluskey. Role of Clathrin and dynamin in clathrin mediated endocytosis/synaptic vesicle recycling and implications in neurological diseases. Front Cell Neurosci 2022; 15, 754110.
HS Kizilkaya, KV Sørensen, CJ Kibsgaard, LS Gasbjerg, AS Hauser, AH Sparre-Ulrich, N Grarup and MM Rosenkilde. Loss of function glucose-dependent insulinotropic polypeptide receptor variants are associated with alterations in BMI, bone strength and cardiovascular outcomes. Frontiers in Cell and Developmental Biology 2021; 9, 749607.
I Badshah, M Anwar, B Murtaza and MI Khan. Molecular mechanisms of morphine tolerance and dependence; novel insights and future perspectives. Molecular and Cellular Biochemistry 2024; 479(6), 1457-1485.
SH Syed, S Unavane, P Taru, A Thakur, N Rai, L Sajimon, S Deore and M Shah. Ethnobotanical, phytochemical and pharmacological activities of genus erythroxylum. Journal of Natural Remedies 2024; 24(8), 1653-1671.
B Aryal, BK Raut, S Bhattarai, S Bhandari, P Tandan, K Gyawali, K Sharma, D Ranabhat, R Thapa, D Aryal, A Ojha, HP Devkota and N Parajuli. Potential therapeutic applications of plant‐derived alkaloids against inflammatory and neurodegenerative diseases. Evidence-Based Complementary and Alternative Medicine 2022; 2022(1), 7299778.
MV Zastrow and A Sorkin. Mechanisms for regulating and organizing receptor signaling by endocytosis. Annual Review of Biochemistry 2021; 90(1), 709-737.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






