The Evaluation of α-amyrin from Callistemon Citrinus: A Study on Distribution and Cytotoxic Properties
DOI:
https://doi.org/10.48048/tis.2025.9436Keywords:
C. citrinus, Cytotoxic, α-amyrin, Screening content, Aromatic herbAbstract
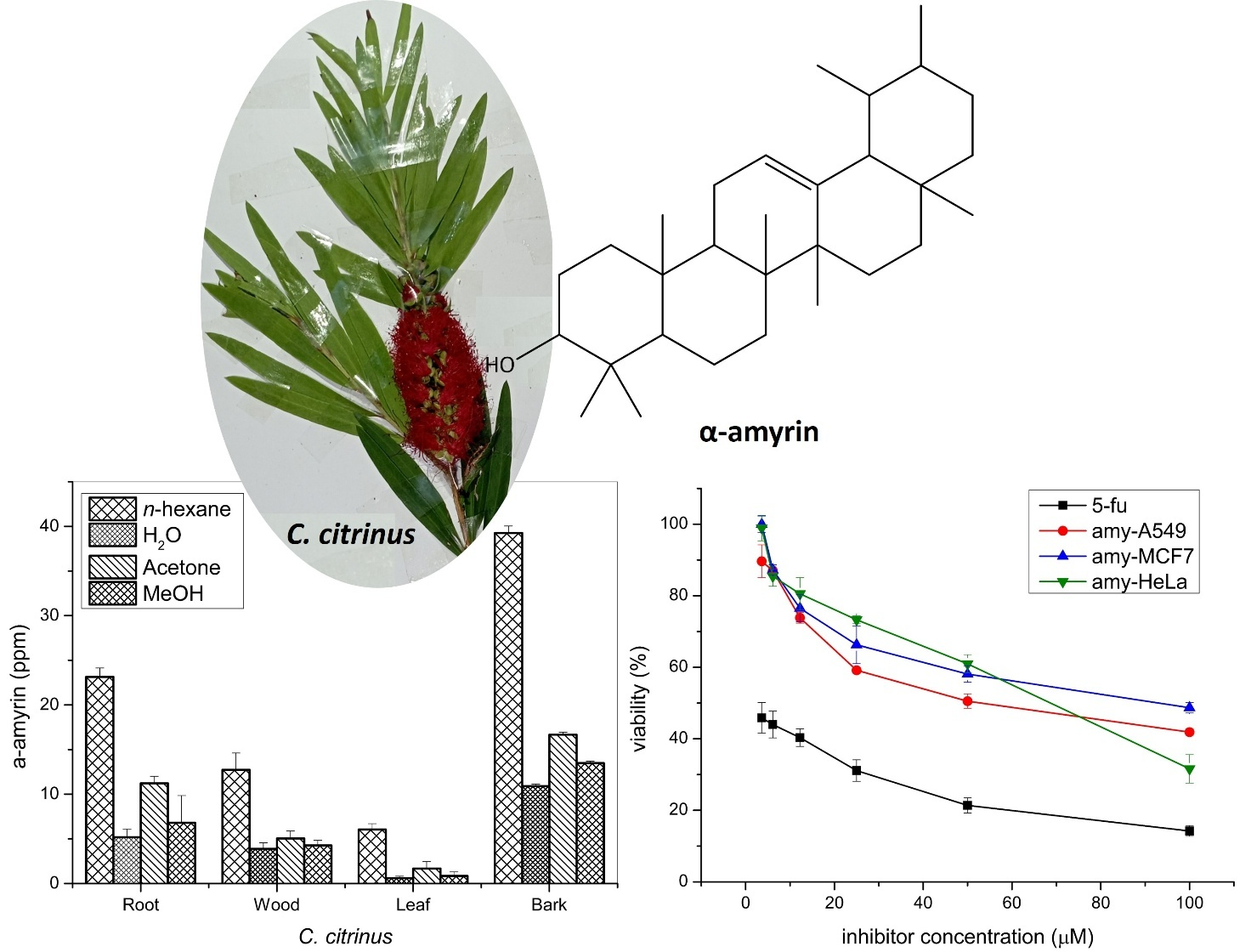
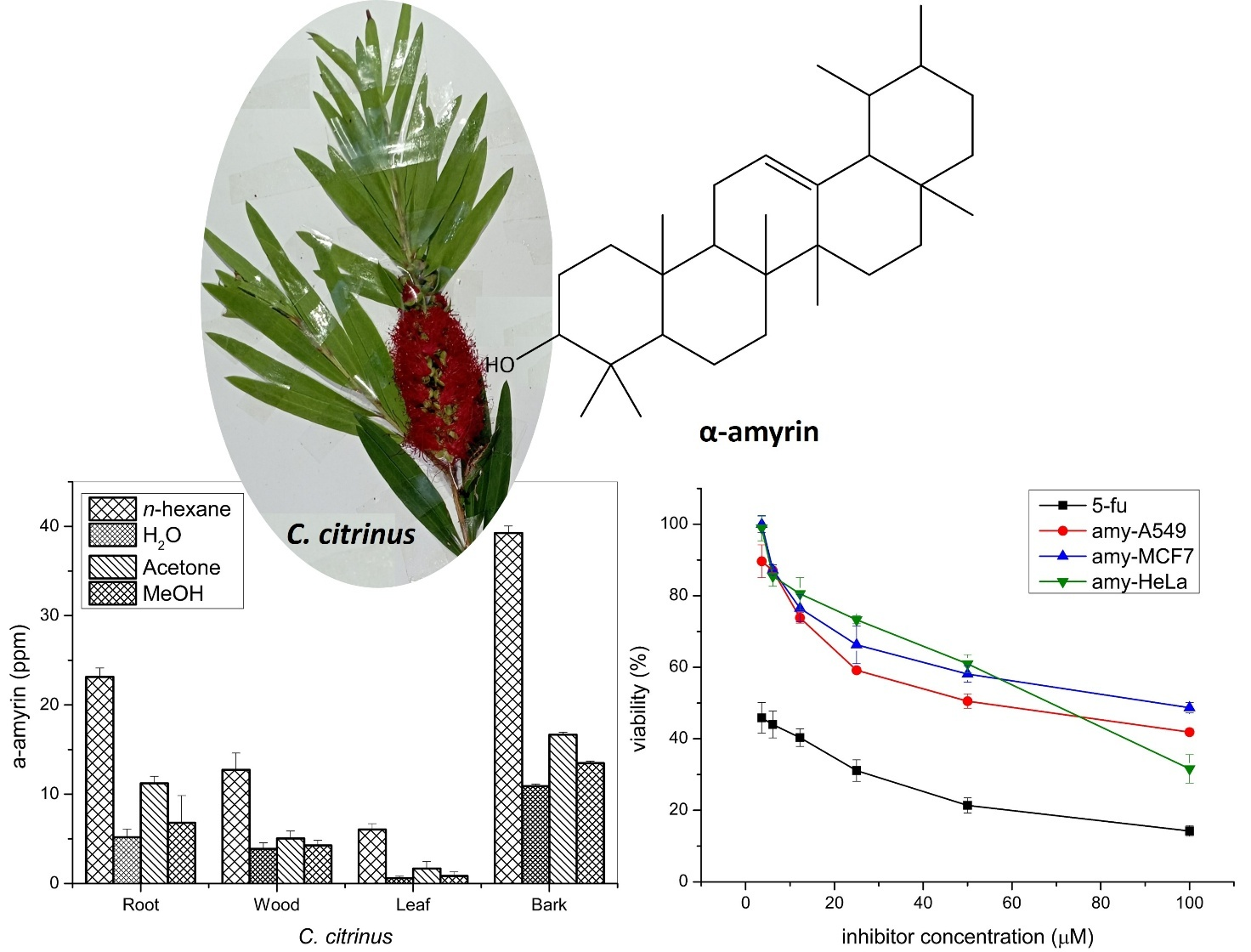
Callistemon citrinus is an aromatic herb, its lemon-scented leaves and bioactive potential propose that it could find a niche as a flavoring spice, intensely in herbal teas and natural medicine formulations. The C. citrinus from ITERA Botanical Gardens is reported to contain α-amyrin, a triterpene with promising bioactivity, prevalent in various segments of C. citrinus. The α-amyrin has cytotoxic properties, which are of interest in both medicinal and food science topics. This study objects to isolate and measure the α-amyrin content from various parts of C. citrinus. The α-amyrin exhibited high concentrations in n-hexane bark extract by 39.27 ± 0.774 ppm and showed moderate-weak activity against A549, MCF7, and HeLa, with IC50 values of 54.14 ± 1.41, 82.79 ± 0.86, 69.35 ± 2.37 µM respectively. This research intends to support the Botanical Garden’s greening campaign by investigating bioactive compounds against malignant human cancer cells.
HIGHLIGHTS
This research supports the Botanical Garden’s greening program by exploring bioactive compounds derived from reforested plants. The C. citrinus contains essential oils and terpenoids, commonly used in aromatherapy, holistic medicine, and culinary applications. Bioactive compounds are found in various parts of the C. citrinus plant, with the bark’s n-hexane extract exhibiting the highest content. The compound exhibits moderate to weak activity against A549, MCF7, and HeLa cancer cell lines. This research promotes the exploration of synergistic treatments using multiple bioactive compounds for cancer therapy.
GRAPHICAL ABSTRACT
Downloads
References
S Chauhan, SVS Chauhan and L Galetto. Floral and pollination biology, breeding system and nectar traits of callistemon citrinus (myrtaceae) cultivated in India. South African Journal Botany 2017; 111, 319-325.
P Rios-Chavez, J Perez-Gonzalez, R Salgado-Garciglia, E Ramirez-Chavez, J Molina-Torres, M Martinez-Trujillo and Y Carreon-Abud. Antibacterial and cytotoxicity activities and phytochemical analysis of three ornamental plants grown in Mexico. Annals of the Brazilian Academy of Sciences 2019; 91(2), e20180468.
C Armijos, W Tapia and K Alexandrino. Assessment of airborne metal pollution in urban parks and industrial areas using callistemon citrinus and acacia melanoxylon. Applied Geochemistry 2022; 139, 105263.
R Rathore and N Rai. Pharmacological action and underlying molecular mechanism of Callistemon: A genus of promising medicinal herbs. Phytomedicine 2022; 99, 154013.
O Rabie, HAS El-Nashar, MY George, TA Majrashi, T Al-Warhi, FE Hassan, WM Eldehna and NM Mostafa. Phytochemical profiling and neuroprotective activity of Callistemon subulatus leaves against cyclophosphamide-induced chemobrain. Biomedicine & Pharmacotherary 2023; 167, 115596.
LA Ayala-Ruiz, LG Ortega-Perez, JS Pinon-Simental, OR Magana-Rodriguez, E Melendez-Herrera and P Rios-Chavez. Role of the major terpenes of Callistemon citrinus against the oxidative stress during a hypercaloric diet in rats. Biomedicine & Pharmacotherary 2022; 153, 113505.
A Lopez-Mejia, LG Ortega-Perez, OR Magana-Rodriguez, LA Ayala-Ruiz, JS Pinon-Simental, DG Hernandez and P Rios-Chavez. Protective effect of Callistemon citrinus on oxidative stress in rats with 1,2-dimethylhydrazine-induced colon cancer. Biomedicine & Pharmacotherary 2021; 142,112070.
MM Sourestani, J Mottaghipisheh, S Vitalini and M Iriti. Monthly changes in contents and compositions of oil of Callistemon citrinus: A comparison study. Natural Product Research 2020; 35(21), 4115-4121.
S Petronilho, SM Rocha, E Ramirez-Chavez, J Molina-Torres and P Rios-Chavez. Assessment of the terpenic profile of callistemon citrinus (curtis) skeels from Mexico. Industrial Crops and Products 2013; 46, 369-379.
LH Vazquez, J Palazon, A Navarro-ocana, A Metropolitana, U Xochimilco and DS Biologicos. A review of sources and biological activities. Intechopen 2011; 426, 487-502.
KMD Melo, FTBD Oliveira, RAC Silva, ALG Quinder, JDBM Filho, AJ Araujo, EDB Pereira, AA Carvalho, MH Chaves, VS Rao and FA Santos. α, β-Amyrin, a pentacyclic triterpenoid from Protium heptaphyllum suppresses adipocyte differentiation accompanied by down regulation of PPARγ and C/EBPα in 3T3-L1 cells. Biomedicine & Pharmacotherary 2018; 109, 1860-1866.
S Haldar, BS Kale, DD Jadhav and HV Thulasiram. Lipase mediated separation of triterpene structural isomers, α- and β-amyrin. Tetrahedron Letters 2014; 55(19), 3122-3125.
BK Cardoso, HLMD Oliveira, UZ Melo, CMM Fernandez, CFDAA Campo, JE Goncalves, AL Jr, MB Romagnolo, GA Linde and ZC Gazim. Antioxidant activity of α and β-amyrin isolated from Myrcianthes pungens leaves. Natural Product Research 2018; 34(12), 1777-1781.
SS Ipav, JO Igoli, TA Tor-Anyiin and JV Anyam. Isolation and characterisation of alpha and beta amyrins from propolis obtained from benue state. Journal of Chemical Society of Nigeria 2022; 47(2), 250-261.
SF Neto, AL Prada, LDR Achod, HFV Torquato, CS Lima, EJ Paredes-Gamero, MOSD Moraes, ES Lima, EH Sosa, TPD Souza and JRR Amado. Α-Amyrin-Loaded nanocapsules produce selective cytotoxic activity in leukemic cells. Biomedicine & Pharmacotherapy 2021; 139, 111656.
MM Victor, JM David, MASD Santos, ALBS Barreiros, ML Barreiros, FS Andrade, AA Carvalho, MCS Luciano, MOD Moraes, FWA Barros-Nepomuceno and C Pessoa. Synthesis and evaluation of cytotoxic effects of amino-ester derivatives of natural α,β-amyrin mixture. Journal of Brazilian Chemical Society 2017; 28(11), 2155-2162.
NN Okoye, DL Ajaghaku, HN Okeke, EE Ilodigwe, CS Nworu and FBC Okoye. Beta-Amyrin and alpha-Amyrin acetate isolated from the stem bark of Alstonia boonei display profound anti-inflammatory activity. Pharmaceutical Biology 2014; 52(11), 1478-1486.
PY Chung, PLN Loh, H Neoh and R Ramli. Alpha-amyrin as an anti-biofilm agent against methicillin-resistant and vancomycin-intermediate staphylococcus aureus. Heliyon 2023; 9(7), e17892.
R Kurniawan, S Sukrasno, A Ashari and T Suhartati. Diving into paclitaxel: Isolation and screening content from Taxus sumatrana at singgalang conservation center, West Sumatra. Natural Product Research 2024; 1(1), 1-5.
A Szakiel, C Paczkowski and S Huttunen. Triterpenoid content of berries and leaves of bilberry vaccinium myrtillus from finland and poland. Journal of Agricultural and Food Chemistry 2012; 48, 11839-11849.
S Wu, F Zhang, W Xiong, I Molnar, J Liang, A Ji, C Wang, S Wang, Z Liu, R Wu and L Duan. An unexpected oxidosqualene cyclase active site architecture in the iris tectorum multifunctional α‑amyrin synthase. ACS Catalysis 2020; 16, 9515-9520.
Y Zhao and C Li. Biosynthesis of plant triterpenoid saponins in microbial cell factories. Journal of Agricultural and Food Chemistry 2018; 46, 12155-12165.
BC Poirier, DA Buchanan, DR Rudell and JP Mattheis. Differential partitioning of triterpenes and triterpene esters in apple peel. Journal of Agricultural and Food Chemistry 2018; 8, 1800-1806.
AM Tawila, S Sun, MJ Kim, AM Omar, DF Dibwe, J Ueda, N Toyooka and S wale. Highly potent antiausterity agents from callistemon citrinus and their mechanism of action against the PANC ‑ 1 human pancreatic cancer cell line. Journal of Natural Products 2020; 7, 2221-2232.
R Kurniawan, S Azis, S Maulana, A Ashari, BA Prasetyo, T Suhartati and S Sukrasno. The cytotoxicity studies of phytosterol discovered from rhizophora apiculata against three human cancer cell lines. Journal of Applied Pharmaceutical Science 2023; 13(1), 156-162.
I Serbian and R Csuk. An improved scalable synthesis of α- and β-amyrin. Molecules 2018; 23(7), 1552.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






