Effect of Size, Charge, and Surface Functionalization of Gadolinium Nanoparticles on Biocompatibility and Cellular Uptake as Magnetic Resonance Imaging Contrast Agents
DOI:
https://doi.org/10.48048/tis.2025.9330Keywords:
Biocompatibility, Cellular uptake, Contrast agents, Functionalization, Gadolinium nanoparticleAbstract
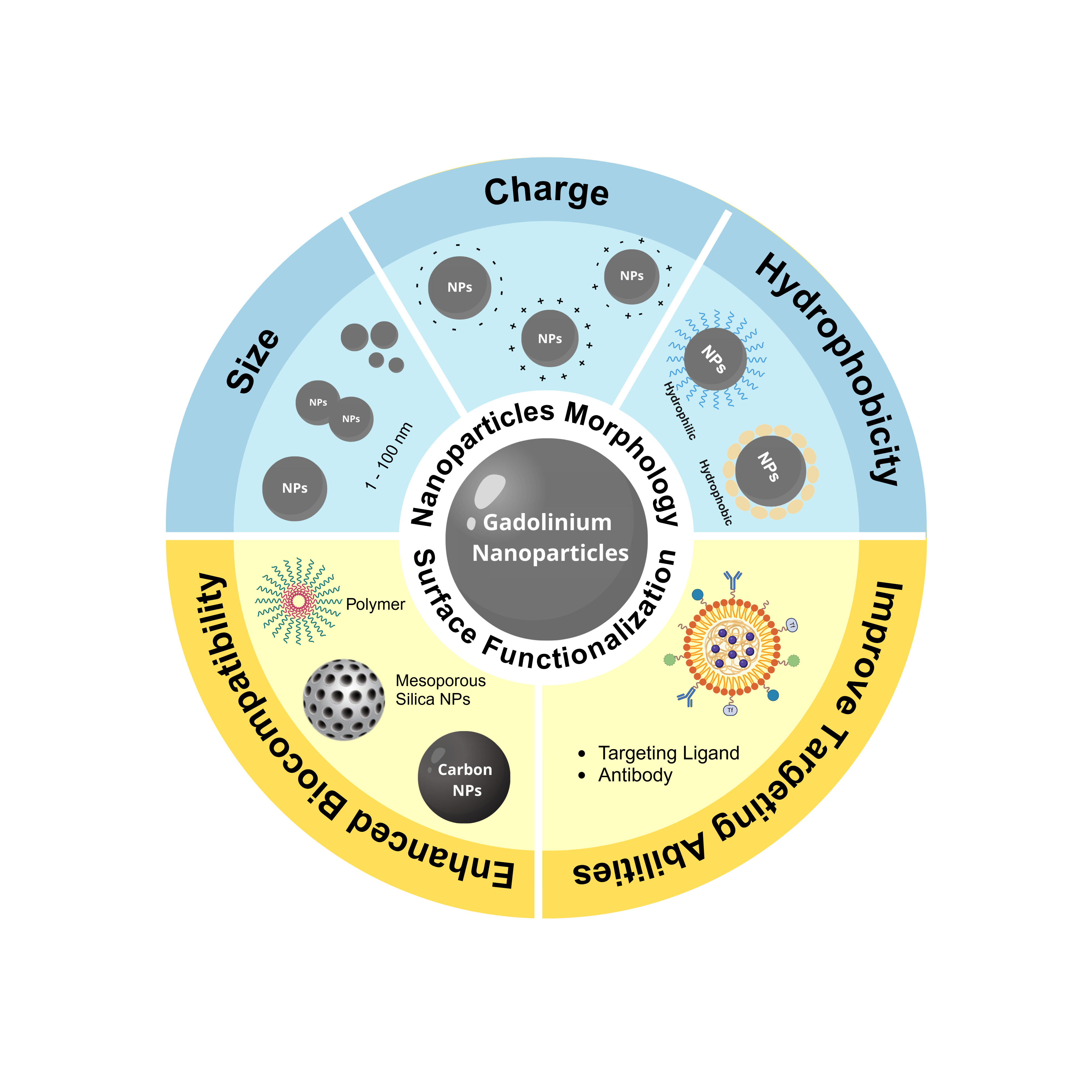
Gadolinium nanoparticles (GdNPs) are nanomaterials with great potential to be applied as MRI contrast agents. GdNPs have been widely explored in recent years as a T1 contrast agent with high performance due to the large relaxivity value (r1) compared to commercial Gd-based contrast agents. However, the major limitations of using these materials for biomedical applications are their cytotoxic effects and cellular uptake efficiency. The morphology of GdNPs, such as size, charge, and surface play an important role in affecting biocompatibility and cellular uptake. For instance, by modifying the surface of nanoparticles, the physical-chemical properties can be altered, leading to improvement in biocompatibility and cellular uptake. Various molecules have the potential to be functionalized on GdNPs, but it is essential to select those that can effectively enhance their abilities. Therefore, this review aims to discuss several studies on the effect of morphology and surface modification of GdNPs to improve biocompatibility and cellular uptake.
HIGHLIGHTS
- Biocompatibility and uptake challenges: Size, surface charge, and functionalization of gadolinium nanoparticles (GdNPs) directly affect cellular uptake and biocompatibility, which are essential for safe and effective Magnetic Resonance Imaging (MRI) contrast.
- Surface modification: The review emphasizes surface modification strategies to enhance biocompatibility and cellular uptake. Functionalization with various molecules tailors GdNPs' physicochemical properties for optimized performance.
- Implications for design: A detailed examination of studies on morphology and modification approaches reveals pathways to improve GdNPs efficacy and safety in medical imaging.
GRAPHICAL ABSTRACT
Downloads
References
S Lacerda. Targeted contrast agents for molecular MRI. Inorganics 2018; 6(4), 129.
AA Rahmani, Q Jia, HH Bahti and RP Fauzia. Recent advances in lanthanide-based nanoparticle contrast agents for magnetic resonance imaging: Synthesis, characterization, and applications. OpenNano 2025; 21, 100226.
RP Fauzia, AG Denkova and K Djanashvili. Potential of MRI in radiotherapy mediated by small conjugates and nanosystems. Inorganics 2019; 7(5), 59.
L Smith, HL Byrne, D Waddington and Z Kuncic. Nanoparticles for MRI-guided radiation therapy: A review. Cancer Nanotechnology 2022; 13, 38.
TA Rose and JW Choi. Intravenous imaging contrast media complications: The basics that every clinician needs to know. American Journal of Medicine 2015; 128(9), 943-949.
H Setiawan, F Triyatna, A Nurmanjaya, M Subechi, DA Sarwono, AA Billah and F Rindiyantono. Synthesis and characterization of gadolinium nanoparticles using polyol method as a candidate for MRI contrast agent. In: Proceedings of the 3rd International Conference on Sciences and Technology Applications, Medan, Indonesia. 2022, p. 12010.
M Yon, C Billotey and JD Marty. Gadolinium-based contrast agents: From gadolinium complexes to colloidal systems. International Journal of Pharmaceutics 2019; 569, 118577.
MF Attia, N Anton, J Wallyn, Z Omran and TF Vandamme. An overview of active and passive targeting strategies to improve the nanocarriers efficiency to tumour sites. Journal of Pharmacy and Pharmacology 2019; 71(8), 1185-1198.
G Sanità, B Carrese and A Lamberti. Nanoparticle surface functionalization: How to improve biocompatibility and cellular internalization. Frontiers in Molecular Biosciences 2020; 7, 587012.
M Ma, Y Huang, H Chen, X Jia, S Wang, Z Wang and J Shi. Bi2S3-embedded mesoporous silica nanoparticles for efficient drug delivery and interstitial radiotherapy sensitization. Biomaterials 2015; 37, 447-455.
P Shi, Z Cheng, K Zhao, Y Chen, A Zhang, W Gan and Y Zhang. Active targeting schemes for nano-drug delivery systems in osteosarcoma therapeutics. Journal of Nanobiotechnology 2023; 21, 103.
RP Fauzia, AJ Sinambela, Z Afriani, Q Jia, HH Bahti and S Wyantuti. Synthesis and characterization of folic acid-modified polyethylene glycol-coated holmium nanoparticles as targeted magnetic resonance imaging agent candidate. Chinese Journal of Analytical Chemistry 2025; 53(1), 100478.
N Hoshyar, S Gray, H Han and G Bao. The effect of nanoparticle size on in vivo pharmacokinetics and cellular interaction. Nanomedicine 2016; 11(6), 673-692.
C He, Y Hu and L Yin. Effects of particle size and surface charge on cellular uptake and biodistribution of polymeric nanoparticles. Biomaterials 2010; 31(13), 3657-3666.
SMA Sadat, ST Jahan and A Haddadi. Effects of size and surface charge of polymeric nanoparticles on in vitro and in vivo applications. Journal of Biomaterials and Nanobiotechnology 2016; 7, 91-108.
F Whba, F Mohamed and MI Idris. Evaluation of physicochemical and biocompatibility characteristics of gadolinium oxide nanoparticles as magnetic resonance imaging contrast agents. Radiation Physics and Chemistry 2023; 213, 111189.
P Vahdatkhah, HRM Hosseini, A Khodaei, AR Montazerabadi, R Irajirad, MA Oghabian and H Delavari. Rapid microwave-assisted synthesis of PVP-coated ultrasmall gadolinium oxide nanoparticles for magnetic resonance imaging. Chemical Physics 2015; 453-454, 35-41.
Y Dai, C Wu, S Wang, Q Li, M Zhang, J Li and K Xu. Comparative study on in vivo behavior of PEGylated gadolinium oxide nanoparticles and Magnevist as MRI contrast agent. Nanomedicine 2018; 14(2), 547-555.
A Guleria, P Pranjali, MK Meher, A Chaturvedi, S Chakraborti, R Raj, KM Poluri and D Kumar. Effect of polyol chain length on proton relaxivity of gadolinium oxide nanoparticles for enhanced magnetic resonance imaging contrast. Journal of Physical Chemistry C 2019; 123(29), 18061-18070.
T Mortezazadeh, E Gholibegloo, NR Alam, S Dehghani, S Haghgoo, H Ghanaati and M Khoobi. Gadolinium (III) oxide nanoparticles coated with folic acid-functionalized poly(β-cyclodextrin-co-pentetic acid) as a biocompatible targeted nano-contrast agent for cancer diagnostic: In vitro and in vivo studies. MAGMA 2019; 32(4), 487-500.
YJ Jang, S Liu, H Yue, JA Park, H Cha, S Ho, S Marasini, A Ghazanfari, MY Ahmad, X Miao, T Tegafaw, KS Chae, Y Chang and GH Lee. Hydrophilic biocompatible poly(acrylic acid-co-maleic acid) polymer as a surface-coating ligand of ultrasmall Gd2O3 nanoparticles to obtain a high R1 value and T1 Mr images. Diagnostics 2021; 11(1), 2.
MY Ahmad, MW Ahmad, H Yue, SL Ho, JA Park, KH Jung, H Cha, S Marasini, A Ghazanfari, S Liu, T Tegafaw, KS Chae, Y Chang and GH Lee. In vivo positive magnetic resonance imaging applications of poly(methyl vinyl ether-alt-maleic acid)-coated ultra-small paramagnetic gadolinium oxide nanoparticles. Molecules 2020; 25(5), 1159.
W Jiang, H Fang, F Liu, X Zhou, H Zhao, X He and D Guo. PEG-coated and Gd-loaded fluorescent silica nanoparticles for targeted prostate cancer magnetic resonance imaging and fluorescence imaging. International Journal of Nanomedicine 2019; 14, 5611-5622.
J Zhang, Z Lin, M Zhao, W Xu and F Nian. Preparation and MRI performances of core-shell structural PEG salicylic acid-gadolinium composite nanoparticles. Journal of Rare Earths 2022; 40(7), 1098-1105.
Y Sui, Y Li, Y Li, H Jin, Y Zheng, W Huang and S Chen. Tumor-specific design of PEGylated gadolinium-based nanoscale particles: Facile synthesis, characterization, and improved magnetic resonance imaging of metastasis lung cancer. Journal of Photochemistry and Photobiology B: Biology 2020; 202, 111669.
S Huang, Q Han, L Wang, T Gong and Q Yuan. One-pot synthesis of PEGylated Gd-based nanoparticles as high-performance and biocompatibility contrast agents for T1-weighted magnetic resonance imaging in vivo. Chemical Research in Chinese Universities 2019; 35(3), 537-541.
S Wyantuti, B Fadhilatunnisa, RP Fauzia, AA Ramani, Irkham and HH Bahti. Response surface methodology box-behnken design to optimise the hydrothermal synthesis of gadolinium nanoparticles. Chinese Journal of Analytical Chemistry 2023; 51(10), 100316.
K He, J Li, Y Shen and Y Yu. pH-Responsive polyelectrolyte coated gadolinium oxide-doped mesoporous silica nanoparticles (Gd2O3@MSNs) for synergistic drug delivery and magnetic resonance imaging enhancement. Journal of Materials Chemistry B 2019; 7(43), 6840-6854.
J Yin, X Wang, H Zheng, J Zhang, H Qu, L Tian, F Zhao and Y Shao. Silica nanoparticles decorated with gadolinium oxide nanoparticles for magnetic resonance and optical imaging of tumors. ACS Applied Nano Materials 2021; 4(4), 3767-3779.
H Yue, S Marasini, MY Ahmad, SL Ho, H Cha, S Liu, YJ Jang, T Tegafaw, A Ghazanfari, X Miao, KS Chae, Y Chang and GH Lee. Carbon-coated ultrasmall gadolinium oxide (Gd2O3@C) nanoparticles: Application to magnetic resonance imaging and fluorescence properties. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2020; 586, 124261.
C Zhang, C Shi, P Chang, S Bian, B Li, J Li and P Hou. MRI directed magnevist effective to study toxicity of Gd-doped mesoporous carbon nanoparticles in mice model. International Journal of Nanomedicine 2023; 18, 6119-6136.
F Maghsoudinia, MB Tavakoli, RK Samani, SH Hejazi, T Sobhani, F Mehradnia and MA Mehrgardi. Folic acid-functionalized gadolinium-loaded phase transition nanodroplets for dual-modal ultrasound/magnetic resonance imaging of hepatocellular carcinoma. Talanta 2021; 228, 122245.
LA Helmy, M Abdel-Halim, R Hassan, A Sebak, HAM Farghali, S Mansour and SN Tammam. The other side to the use of active targeting ligands; the case of folic acid in the targeting of breast cancer. Colloids and Surfaces B: Biointerfaces 2022; 211, 112289.
Y Cai, Y Wang, T Zhang and Y Pan. Gadolinium-labeled ferritin nanoparticles as T1 contrast agents for magnetic resonance imaging of tumors. ACS Applied Nano Materials 2020; 3(9), 8771-8783.
MQ Rodrigues, PM Alves and A Roldão. Functionalizing ferritin nanoparticles for vaccine development. Pharmaceutics 2021; 13(10), 1621.
J He, K Fan and X Yan. Ferritin drug carrier (FDC) for tumor targeting therapy. Journal of Controlled Release 2019; 311-312, 288-300.
K Kim, H Choi, ES Choi, MH Park and JH Ryu. Hyaluronic acid-coated nanomedicine for targeted cancer therapy. Pharmaceutics 2019; 11(7), 301.
Z Luo, Y Dai and H Gao. Development and application of hyaluronic acid in tumor targeting drug delivery. Acta Pharmaceutica Sinica B 2019; 9(6), 1099-1112.
C Wu, R Cai, T Zhao, L Wu, L Zhang, J Jin, L Xu, P Li, T Li, M Zhang and F Du. Hyaluronic acid-functionalized gadolinium oxide nanoparticles for magnetic resonance imaging-guided radiotherapy of tumors. Nanoscale Research Letters 2020; 15(1), 94.
A Fatima, MW Ahmad, AKAA Saidi, A Choudhury, Y Chang and GH Lee. Recent advances in gadolinium based contrast agents for bioimaging applications. Nanomaterials 2021; 11(9), 2449.
SD Serai, ML Ho, M Artunduaga, SS Chan and GB Chavhan. Components of a magnetic resonance imaging system and their relationship to safety and image quality. Pediatric Radiology 2021; 51(5), 716-723.
S Khairnar, N More, C Mounika and G Kapusetti. Advances in contrast agents for contrast-enhanced magnetic resonance imaging. Journal of Medical Imaging and Radiation Sciences 2019; 50(4), 575-589.
YD Xiao, R Paudel, J Liu, C Ma, ZS Zhang and SK Zhou. MRI contrast agents: Classification and application. International Journal of Molecular Medicine 2016; 38(5), 1319-1326.
S Caspani, R Magalhães, JP Araújo and CT Sousa. Magnetic nanomaterials as contrast agents for MRI. Materials 2020; 13(11), 2586.
Y Jeong, HS Hwang and K Na. Theranostics and contrast agents for magnetic resonance imaging. Biomaterials Research 2018; 22, 20.
SM Vithanarachchi and MJ Allen. Strategies for target-specific contrast agents for magnetic resonance imaging. Current Molecular Imaging 2012; 1(1), 12-25.
T Curry, R Kopelman, M Shilo and R Popovtzer. Multifunctional theranostic gold nanoparticles for targeted CT imaging and photothermal therapy. Contrast Media & Molecular Imaging 2014; 9(1), 53-61.
Y Shi, RVD Meel, X Chen and T Lammers. The EPR effect and beyond: Strategies to improve tumor targeting and cancer nanomedicine treatment efficacy. Theranostics 2020; 10(17), 7921-7924.
R Augustine, A Hasan, R Primavera, RJ Wilson, AS Thakor and BD Kevadiya. Cellular uptake and retention of nanoparticles: Insights on particle properties and interaction with cellular components. Materials Today Communications 2020; 25, 101692.
E Fröhlich. The role of surface charge in cellular uptake and cytotoxicity of medical nanoparticles. International Journal of Nanomedicine 2012; 7, 5577-5591.
K Kettler, K Veltman, DVD Meent, AV Wezel and AJ Hendriks. Cellular uptake of nanoparticles as determined by particle properties, experimental conditions, and cell type. Environment Toxicology Chemistry 2014; 33(3), 481-492.
S Majeed and SA Shivashankar. Rapid microwave-assisted synthesis of Gd2O3 and Eu:Gd2O3 nanocrystals: Characterization, magnetic, optical and biological studies. Journal of Materials Chemistry B 2014; 2(34), 5585-5593.
L Zhou, T Yang, J Wang, Q Wang, X Lv, H Ke, Z Guo, J Shen, Y Wang, C Xing and H Chen. Size-tunable Gd2O3@Albumin nanoparticles conjugating chlorin e6 for magnetic resonance imaging-guided photo-induced therapy. Theranostics 2017; 7(3), 764-774.
A Chaturvedi, P Pranjali, MK Meher, R Raj, M Basak, RK Singh, KM Poluri, D Kumar and A Guleria. In vitro and ex vivo relaxometric properties of ethylene glycol coated gadolinium oxide nanoparticles for potential use as contrast agents in magnetic resonance imaging. Journal of Applied Physics 2020; 128, 34903.
M Awashra and P Młynarz. The toxicity of nanoparticles and their interaction with cells: An in vitro metabolomic perspective. Nanoscale Advances 2023; 5(10), 2674-2723.
P Sabourian, G Yazdani, SS Ashraf, M Frounchi, S Mashayekhan, S Kiani and A Kakkar. Effect of physico-chemical properties of nanoparticles on their intracellular uptake. International Journal of Molecular Sciences 2020; 21(21), 8019.
L Kou J, Sun, Y Zhai and Z He. The endocytosis and intracellular fate of nanomedicines: Implication for rational design. Asian Journal of Pharmaceutical Sciences 2013; 8(1), 1-10.
T Mortezazadeh, E Gholibegloo, NR Alam, S Haghgoo and A Mesbahi. In vitro and in vivo characteristics of doxorubicin-loaded cyclodextrine-based polyester modified gadolinium oxide nanoparticles: A versatile targeted theranostics system for tumour chemoteraphy and molecular resonance imaging. Journal of Drug Targeting 2019; 28(5), 533-546.
E Wysokińska, J Cichos, E Zioło, A Bednarkiewicz, L Strządała, M Karbowiak, D Hreniak and W Kałas. Cytotoxic interactions of bare and coated NaGdF4:Yb3+:Er3+ nanoparticles with macrophage and fibroblast cells. Toxicology in Vitro 2016; 32, 16-25.
FS Mozar and EH Chowdhury. Impact of PEGylated nanoparticles on tumor targeted drug delivery. Current Pharmaceutical Design 2018; 24(28), 3283-3296.
Z Li, J Guo, M Zhang, G Li and L Hao. Gadolinium-coated mesoporous silica nanoparticle for magnetic resonance imaging. Frontiers in Chemistry 2022; 10, 837032.
D Kudaibergen, HS Park, J Park, GB Im, JR Lee, YK Joung, SH Bhang and JH Kim. Silica-based advanced nanoparticles for treating ischemic disease. Tissue Engineering and Regenerative Medicine 2023; 20(2), 177-198.
H Chen, GD Wang, W Tang, T Todd, Z Zhen, C Tsang, K Hekmatyar, T Cowger, R Hubbard, W Zhang, J Stickney, B Shen and J Xie. Gd-encapsulated carbonaceous dots with efficient renal clearance for magnetic resonance imaging. Advanced Materials 2014; 26(39), 6761-6766.
RP Fauzia and HH Bahti. Sintesis dan karakterisasi (3-aminopropil)-trimetoksisilan terkonjugasi asam folat untuk fungsionalisasi nanopartikel (in Indonesian). Chimica et Natura Acta 2023; 11(1), 36-40.
C Martín-Sabroso, AI Torres-Suárez, M Alonso-González, A Fernández-Carballido and AI Fraguas-Sánchez. Active targeted nanoformulations via folate receptors: State of the art and future perspectives. Pharmaceutics 2022; 14(1), 14.
SL Ho, H Yue, S Lee, T Tegafaw, MY Ahmad, S Liu, AKAA Saidi, D Zhao, Y Liu, SW Nam, KS Chae, Y Chang and GH Lee. Mono and multiple tumor-targeting ligand-coated ultrasmall gadolinium oxide nanoparticles: Enhanced tumor imaging and blood circulation. Pharmaceutics 2022; 14(7), 1458.
G Huang and H Huang. Hyaluronic acid-based biopharmaceutical delivery and tumor-targeted drug delivery system. Journal of Controlled Release 2018; 278, 122-126.
E López-Ruiz, G Jiménez, LÁD Cienfuegos, C Antic, R Sabata, JA Marchal and P Gálvez-Martín. Advances of hyaluronic acid in stem cell therapy and tissue engineering, including current clinical trials. European Cells & Materials 2019; 37, 186-213.
H Nosrati, M Salehiabar, J Charmi, K Yaray, M Ghaffarlou, E Balcioglu and YN Ertas. Enhanced in vivo radiotherapy of breast cancer using gadolinium oxide and gold hybrid nanoparticles. ACS Applied Bio Materials 2023; 6(2), 784-792.
Z Liu, M Zhao, H Wang, Z Fu, H Gao, W Peng, D Ni, W Tang and Y Gu. High relaxivity Gd3+ based organic nanoparticles for efficient magnetic resonance angiography. Journal of Nanobiotechnology 2022; 20, 170.
Y Liu, Y Tang, Y Tian, J Wu, J Sun, Z Teng, S Wang and G Lu. Gadolinium-doped hydroxyapatite nanorods as T1 contrast agents and drug carriers for breast cancer therapy. ACS Applied Nano Materials 2019; 2(3), 1194-1201.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






