Polycarboxylic Acid-Based Deep Eutectic Solvents for Critical Metal Recovery from Lithium-Ion Batteries: Kinetic and Efficiency Analysis
DOI:
https://doi.org/10.48048/tis.2025.9244Keywords:
Lithium-ion batteries, DES, ChCl, Succinic acid, Malonic acid, Maleic acidAbstract
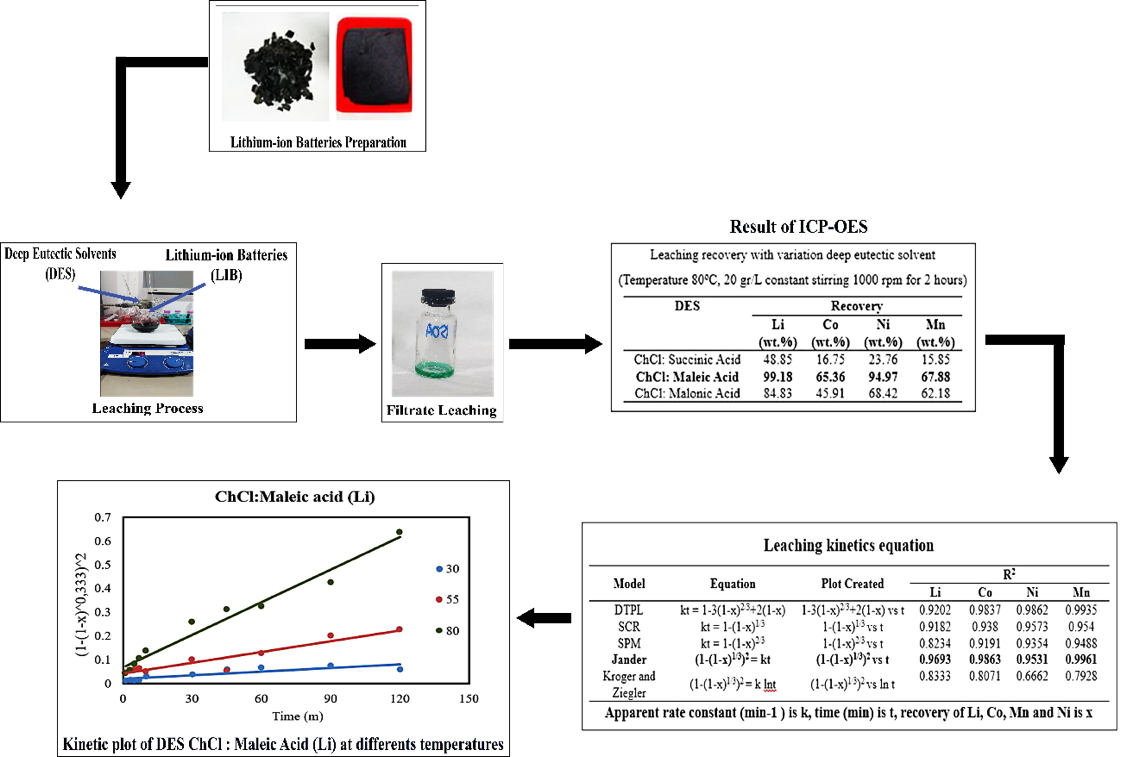
To meet the high demand for lithium-ion batteries (LIBs) and their corresponding challenges, effective and sustainable recycling methods are necessary. This research explores the use of deep eutectic solvents (DES) based on polycarboxylic acid to recover critical metals, such as lithium (Li), cobalt (Co), nickel (Ni), and manganese (Mn) from spent LIBs. The leaching efficiency of 3 DES was evaluated under various conditions by synthesizing them with succinic acid, malonic acid, and maleic acid using choline chloride. Optimal recovery was achieved with ChCl: Maleic acid, yielding 99.18 wt. % Li, 65.36 wt. % Co, 94.97 wt. % Ni, and 67.88 wt. % Mn at a S/L ratio of 20 g/L at 80 °C with constant stirring. Higher S/L ratios led to reduced recovery rates due to mass transfer limitations and solution saturation. Kinetic modeling revealed that the Jander model best described the leaching mechanism, suggesting a diffusion-controlled process. The activation energy calculation on DES ChCl: Maleic acid produces Li 38.57 kJ/mol, Co 63.09 kJ/mol, Ni 64.87 kJ/mol, and Mn 52.64 kJ/mol. The use of DES derived from eco-friendly polycarboxylic acids (succinic, malonic, and maleic acids) in this study represents an innovative way to recover critical metals from spent lithium-ion batteries (LIBs) in a sustainable and effective manner. Future studies are advised to examine the DES composition and investigate how agitation influences the recovery of specific metals.
HIGHLIGHTS
- Lithium-ion battery leaching using deep eutectic solvent as leaching agent
- The results obtained from the leaching of lithium-ion batteries were the recovery of 99.18 % Li, 65.36 % Co, 67.88 % Mn, and 94.97 % Ni with the optimum leaching condition used was a solid/liquid ratio of 20gr/L with a temperature of 80 °C for 2 h.
- The kinetic mechanism of the leaching process follows Jander kinetics.
GRAPHICAL ABSTRACT
Downloads
References
A Zentani, A Almaktoof and MT Kahn. A comprehensive review of developments in electric vehicles fast charging technology. Applied Sciences 2024; 14(11), 4728.
M Jafari, SZ Shafaie, H Abdollahi and A Entezari-Zarandi. Green recycling of spent Li-ion batteries by deep eutectic solvents (DESs): Leaching mechanism and effect of ternary DES. Journal of Environmental Chemical Engineering 2022; 10(6), 109014.
A Zanoletti, E Carena, C Ferrara and E Bontempi. A review of lithium-ion battery recycling: Technologies, sustainability, and open issues. Batteries 2024; 10(1), 38.
AF Yi, ZW Zhu, YH Liu, J Zhang, H Su and T Qi. Using highly concentrated chloride solutions to leach valuable metals from cathode-active materials in spent lithium-ion batteries. Rare Metals 2020; 40, 1971-1978.
T Raj, K Chandrasekhar, AN Kumar, P Sharma, A Pandey, M jang, B Jeon, S Varjani and S Kim. Recycling of cathode material from spent lithium-ion batteries: Challenges and future perspectives. Journal of Hazardous Materials 2022; 429, 128312.
B Musariri, G Akdogan, C Dorfling and S Bradshaw. Evaluating organic acids as alternative leaching reagents for metal recovery from lithium ion batteries. Minerals Engineering 2019; 137, 108-117.
L Yurramendi, J Hidalgo and A Siriwardana. A Sustainable process for the recovery of valuable metals from spent lithium ion batteries by deep eutectic solvents leaching. Materials Proceedings 2021; 5(1), 100.
A Zhu, X Bian, W Han, D Cao, Y Wen, K Zhu and S Wang. The application of deep eutectic solvents in lithium-ion battery recycling: A comprehensive review. Resources, Conservation and Recycling 2023; 188, 106690.
Y Hua, Y Sun, F Yan, S Wang, Z Xu, B Zhao and Z Zhang. Ionization potential-based design of deep eutectic solvent for recycling of spent lithium ion batteries. Chemical Engineering Journal 2022; 436, 133200.
S Wang, Z Zhang, Z Lu and Z Xu. A novel method for screening deep eutectic solvent to recycle the cathode of Li-ion batteries. Green Chemistry 2020; 22(14), 4473-4482.
B Lu, R Du, G Wang, Y Wang, S Dong, D Zhou, S Wang and C Li. High-efficiency leaching of valuable metals from waste Li-ion batteries using deep eutectic solvents. Environmental Research 2022; 212(B), 113286.
E Prasetyo, WA Muryanta, AG Anggraini, S Sudibyo, M Amin and MA Muttaqii. Tannic acid as a novel and green leaching reagent for cobalt and lithium recycling from spent lithium-ion batteries. Journal of Material Cycles and Waste Management 2022; 24, 927-938.
J Liu, X Li and KH Row. Development of deep eutectic solvents for sustainable chemistry. Journal of Molecular Liquids 2022; 362, 119654.
MA Ali, MA Kaium, SN Uddin, MJ Uddin, O Olawuyi, AD Campbell, CJ Saint-Louis and MA Halim. Elucidating the structure, dynamics, and interaction of a choline chloride and citric acid based eutectic system by spectroscopic and molecular modeling investigations. ACS Omega 2023; 8(41), 38243-38251.
Y Wen, X He, S Di, K Liu, D Li and J Du. Comparative of malonic acid aqueous solution and malonic acid-based deep eutectic solvent for LiCoO2 cathode materials recovery: Leaching efficiency and mechanism. Journal of Environmental Chemical Engineering 2023; 11(5), 110979.
J Coates. Interpretation of infrared spectra, a practical approach. Wiley Online Library, New Jersey, 2006.
R Ninayan, AS Levshakava, EM Khairullina, OS Vezo, II Tumkin, A Ostendorf, LS Logunov, AA Manshina and AY Shishov. Water-induced changes in choline chloride-carboxylic acid deep eutectic solvents properties. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2023; 679, 1325243.
X Yu, W Li, V Gupta, H Gao, D Tran, S Sarwar and Z Chen. Current challenges in efficient lithium-ion batteries’ recycling: A perspective. Global Challenges 2022; 6(12), 2200099.
T Zhang, J Dao, J Wang, R Wang, C Li, D Wang, C Bao, Y Zhang, Z Zhang and P Dong. Efficient leaching of valuable metals from NCM cathode materials by green deep eutectic solvent. Journal of Cleaner Production 2024; 438, 140636.
TR Somo, TE Mabokela, DM Teffu, TK Sekgobela, B Ramogayana, MJ Hato and KD Modibane. A comparative review of metal oxide surface coatings on three families of cathode materials for lithium ion batteries. Coatings 2021; 11(7), 744.
C Padwal, HD Pham, S Jadhav, TT Do, J Nerkar, LTM Hoang, AK Nanjundan, SG Mundree and DP Dubal. Deep eutectic solvents: Green approach for cathode recycling of Li‐Ion batteries. Advanced Energy and Sustainability Research 2021; 3(1), 2100133.
Q Peng, S Qi, LP Lv, W Sun, Y Wang and S Chen. A cobalt-based coordination compound with high capacity as the anode of lithium-ion battery and its stepwise reaction mechanism. Journal of Alloys and Compounds 2022; 927, 167064.
Y Zhang, P Cui, G Lua, L Chen, X Li, Y Chao and W Zhu. One-step selective separation and efficient recovery of valuable metals from spent lithium batteries by phosphoric acid-based deep eutectic solvent. Green Chemical Engineering 2024; 5(3), 390-398.
C Liu, J Yu, J Hu, J Xu, A Yu, T Liu, Z Wang, X Luo, C Deng, F Luo, J He and G Zeng. Ultra-low viscosity betaine hydrochloride-formic acid deep eutectic solvent for leaching critical metals from spent NCM lithium-ion batteries. Journal of Environmental Chemical Engineering 2024; 12(3), 112586.
SC Chelgani, M Rudolph, T Leistner, J Gutzmer and UA Peuker. A review of rare earth minerals flotation: Monazite and xenotime. International Journal of Mining Science Technology 2015; 25(6), 877-883.
X He, Y Wen, X Wang, Y Cui, L Li and H Ma. Leaching NCM cathode materials of spent lithium-ion batteries with phosphate acid-based deep eutectic solvent. Waste Management 2023; 157, 8-16.
PG Schiavi, P Altimari, E Sturabotti, AG Marrani, G Simonetti and F Pagnanelli. Decomposition of deep eutectic solvent aids metals extraction in lithium-ion batteries recycling. ChemSusChem 2022; 15(18), e202200966.
L Chen, Y Chao, X Li, G Zhou, Q Lu, M Hua, H Li, X Ni, P Wu and W Zhu. Engineering a tandem leaching system for the highly selective recycling of valuable metals from spent Li-ion batteries. Green Chemistry 2021; 23(5), 2177-2184.
R Morina, D Callegari, D Merli, G Alberti, P Mustarelli and E Quartarone. Cathode active material recycling from spent lithium batteries: A green (circular) approach based on deep eutectic solvents. ChemSusChem 2021; 15(2), e202102080.
E Prasetyo, E Purwaningsih and W Astuti. Selective-reductive leaching of manganese from low-grade manganese ore using tannic acid as reductant. Mining, Metallurgy & Exploration 2019; 36, 1003-1012.
W Aryani, AG Anggraini, F Bahfie, U Herlina, MA Muttaqii and E Prasetyo. A kinetic study of manganese leaching from low-grade psilomelane ore by acetic-tannic acid lixiviant. ASEAN Journal of Chemical Engineering 2021; 21(2), 188-200.
C Cui, Y Dang, C Luo, L Wang and H Peng. Mechanical properties and reaction kinetics of alkali-activated metakaolin. Materials 2024; 17(2), 367.
B Zhu, Y Zhang, Y Zou, Z yang, B Zhang, Y Zhao, M Zhang, Q Meng and P Dong. Leaching kinetics and interface reaction of LiNi0.6Co0.2Mn0.2O2 materials from spent LIBs using GKB as reductant. Journal of Environmental Management 2021; 300, 113710.
X Cao, H Wang, T Liu, Y Shi and X Xue. Electrodeposition of Bi from choline chloride-malonic acid deep eutectic solvent. Materials 2023; 16(1), 415.

Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






