Enhancement of Arachidonic Acid Production by A Newly Isolated Diplosphaera sp. Associated with Bryophyte
DOI:
https://doi.org/10.48048/tis.2025.9219Keywords:
Arachidonic acid, Polyunsaturated fatty acids, Microalgae, Diplosphaera sp., OptimizationAbstract
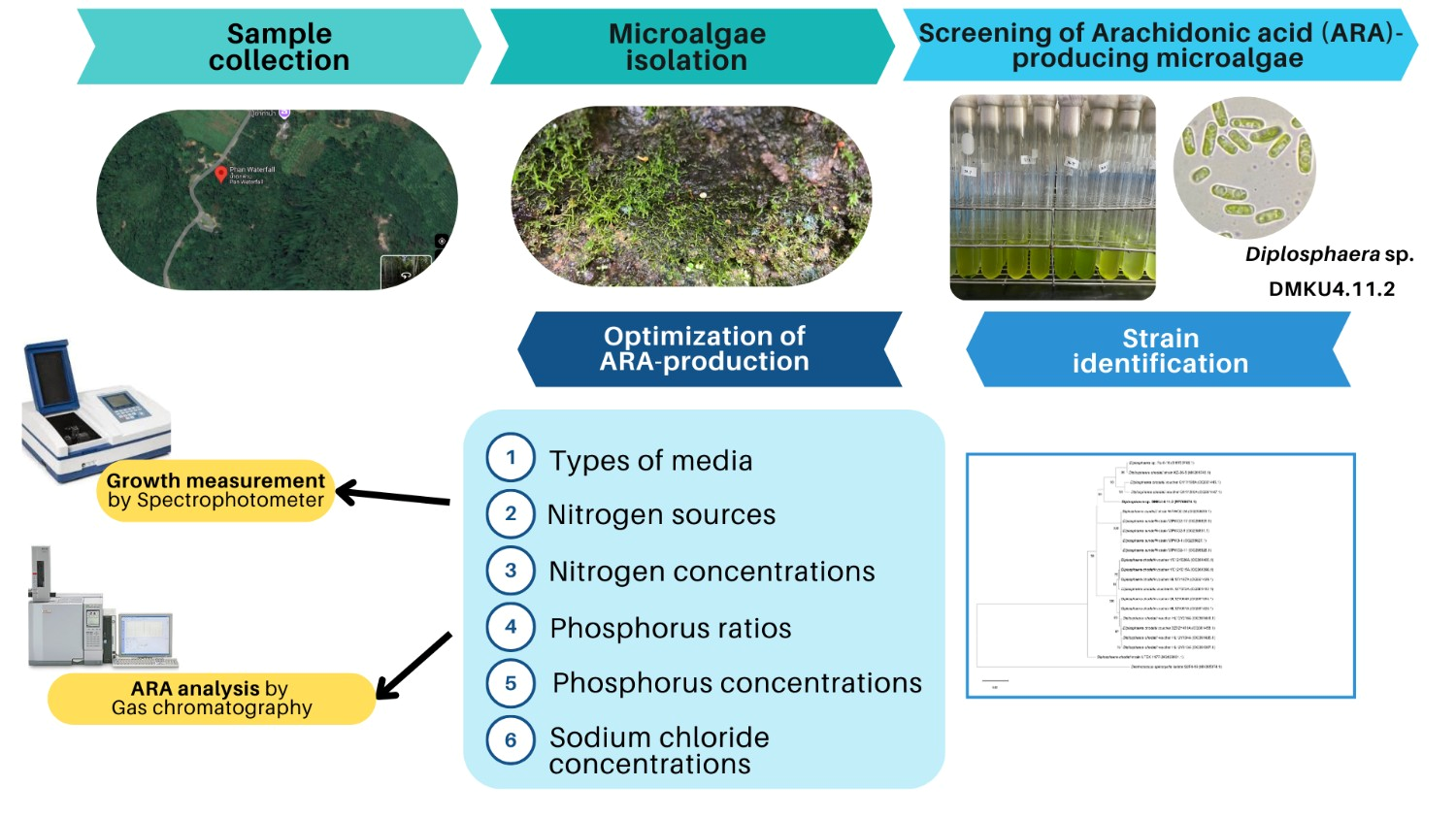
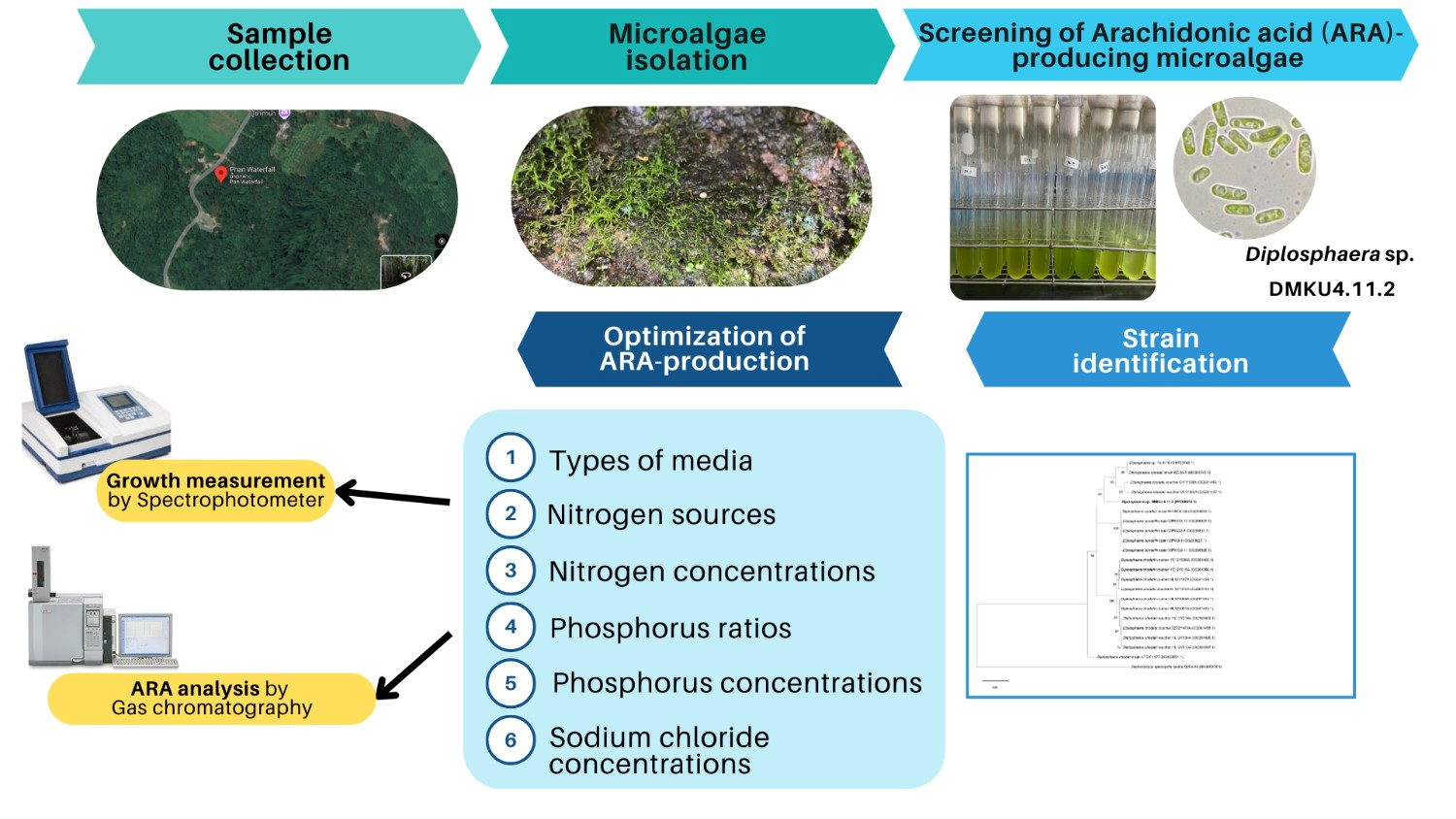
Arachidonic acid (ARA) is an essential omega-6 polyunsaturated fatty acid with significant roles in nervous system development and various physiological processes. While ARA production from freshwater microalgae has been limited, this study aimed to optimize cultivation conditions for ARA production in Diplosphaera sp. DMKU4.11.2, a newly isolated strain from a bryophyte in Thailand. Among 6 tested media, BG11 demonstrated the highest ARA production. Further optimization of the BG11 medium involved adjusting factors such as the nitrogen source, nitrogen concentration, phosphorus ratio, phosphorus level and sodium chloride supplementation. The optimal conditions were determined to be BG11 medium containing 0.75 g/L sodium nitrate and potassium phosphate at a 1:1 ratio, with 3.50 mg P/L. Under these conditions, Diplosphaera sp. DMKU4.11.2 produced the highest ARA yield of 115.31 ± 2.03 mg/g dry cell weight (DCW), with an ARA production rate of 3.23 ± 0.12 mg/L/day. Supplementing the medium with 50 mM sodium chloride further enhanced ARA content to 9.57 ± 1.67 % of total fatty acids (TFAs) and increased biomass production to 0.36 ± 0.00 g/L. Culturing in the optimized BG11 medium resulted in a 1.24-fold increase in growth and a 3.95-fold increase in ARA content compared to the standard BG11 medium. In conclusion, this study highlights the potential of Diplosphaera sp. DMKU4.11.2 as a sustainable source for ARA production, with opportunities to enhance productivity for industrial and health-related applications.
HIGHLIGHTS
- The freshwater microalga Diplosphaera DMKU4.11.2, newly isolated from a bryophyte in Thailand, was identified for its potential in producing arachidonic acid (ARA).
- BG11 medium demonstrated the highest ARA production among tested media.
- Cultivation in modified BG11 resulted in a 1.24-fold increase in biomass and a 3.95-fold increase in ARA content, compared to cultivation in standard BG11.
GRAPHICAL ABSTRACT
Downloads
References
JG Bell and JR Sargent. Arachidonic acid in aquaculture feeds: Current status and future opportunities. Aquaculture 2003; 218(1-4), 491-499.
B Koletzko and M Braun. Arachidonic acid and early human growth: Is there a relation? Annals of Nutrition and Metabolism 1991; 35(3), 128-131.
A Ahmad, SW Hassan and F Banat. An overview of microalgae biomass as a sustainable aquaculture feed ingredient: Food security and circular economy. Bioengineered 2022; 13(4), 9521-9547.
H Qi, Y Liu, F Jian, X Xing, J Wang and C Li. Effects of dietary arachidonic acid (ARA) on immunity, growth and fatty acids of Apostichopus japonicus. Fish & Shellfish Immunology 2022; 127, 901-909.
L Chang, H Lu, H Chen, X Tang, J Zhao, H Zhang, YQ Chen and W Chen. Lipid metabolism research in oleaginous fungus Mortierella alpina: Current progress and future prospects. Biotechnology Advances 2022; 54, 107794.
LL Ouyang, H Li, F Liu, M Tong, SY Yu and ZG Zhou. Accumulation of arachidonic acid in a green microalga, Myrmecia incisa H4301, enhanced by nitrogen starvation and its molecular regulation mechanisms. In: Arachidonic acid: Dietary sources and general functions. In: GG Dumancas, BS Murdianti and EA Lucas (Eds.). Arachidonic acid: Dietary sources and gneral functions. Nova Science Publishers, New York, 2013.
B Gao, L Huang, X Lei, G Meng, J Liu and C Zhang. Evaluation and transcriptome analysis of the novel oleaginous microalga Lobosphaera bisecta (Trebouxiophyceae, Chlorophyta) for arachidonic acid production. Marine Drugs 2020; 18(5), 229.
SM Shanab, RM Hafez and AS Fouad. A review on algae and plants as potential source of arachidonic acid. Journal of Advanced Research 2018; 11, 3-13.
V Dolganyuk, D Belova, O Babich, A Prosekov, S Ivanova, D Katserov, N Patyukov and S Sukhikh. Microalgae: A promising source of valuable bioproducts. Biomolecules 2020; 10(8), 1153.
W Klinthong, YH Yang, CH Huang and CS Tan. A review: Microalgae and their applications in CO2 capture and renewable energy. Aerosol and Air Quality Research 2015; 15(2), 712-742.
N Politaeva, I Ilin, K Velmozhina and P Shinkevich. Carbon dioxide utilization using Chlorella microalgae. Environments 2023; 10(7), 109.
IA Matter, OM Darwesh and FK El-baz. Using the natural polymer chitosan in harvesting Scenedesmus species under different concentrations and cultural pH values. International Journal of Pharma and Bio Sciences 2016; 7(4), 254-260.
A Juneja, RM Ceballos and GS Murthy. Effects of environmental factors and nutrient availability on the biochemical composition of algae for biofuels production: A review. Energies 2013; 6(9), 4607-4638.
G Su, K Jiao, J Chang, Z Li, X Guo, Y Sun, X Zeng, Y Lu and L Lin. Enhancing total fatty acids and arachidonic acid production by the red microalgae Porphyridium purpureum. Bioresources and Bioprocessing 2016; 3, 33.
CJ Hulatt, RH Wijffels, S Bolla and V Kiron. Production of fatty acids and protein by Nannochloropsis in flat-plate photobioreactors. PLoS One 2017; 12(1), e0170440.
S Celewicz-Gołdyn and N Kuczyńska-Kippen. Ecological value of macrophyte cover in creating habitat for microalgae (diatoms) and zooplankton (rotifers and crustaceans) in small field and forest water bodies. PLoS One 2017; 12(5), e0177317.
JA Ress and RL Lowe. Bryophytes and associated algal communities from an exposed cliff face on O’ahu (Hawai’i, USA). Algological Studies 2014; 144(1), 45-72.
C Bigogno, G Khozin and Z Cohen. Accumulation of arachidonic acid-rich triacylglycerols in the microalga Parietochloris incisa (Trebuxiophyceae, Chlorophyta). Phytochemistry 2002; 60, 135-143.
S Watanabe, S Hirabayashi, S Boussiba, Z Cohen, A Vonshak and A Richmond. Parietochloris incisa comb. nov.(Trebouxiophyceae, Chlorophyta). Phycological Research 1996; 44(2), 107-108.
T Ichimura. Sexual cell division and conjugation-papilla formation in sexual reproduction of Closterium strigosum. In: Proceedings of the 7th International Seaweed Symposium, Tokyo, Japan. 1971, p. 208-214.
JJ Doyle and JL Doyle. A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochemical Bulletin 1987; 19(1), 11-15.
TJ White, T Bruns, S Lee and J Taylor. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: MA Innis, DH Gelfand, JJ Sninsky and TJ White (Eds.). PCR protocols: A guide to methods and applications. Academic Press, Massachusetts, 1990, p. 315-322.
SJ Kato. Laboratory culture and morphology of Colacium vesiculosum Ehrb. (Euglenophyceae). The Japanese Journal of Phycology (Sôrui) 1982; 30(1), 63-67.
PT Kantz and HC Bold. Morphological and taxonomic investigations of Nostoc and Anabaena in culture. University of Texas, Texas, 1969.
A Vonshak and G Torzillo. Environmental stress physiology. In: A Richmond (Ed.). Handbook of microalgal culture: Biotechnology and applied phycology. Blackwell science, Oxford, 2003, p. 57-82.
C Largeau, E Casadevall and C Berkaloff. The biosynthesis of long-chain hydrocarbons in the green alga Botryococcus braunii. Phytochemistry 1980; 19(6), 1081-1085.
HD Payer. Algae project. Institute of Food Research and Product Development (IFRPD), Bangkok, Thailand, 1971.
EG Bligh and WJ Dyer. A rapid method of total lipid extraction and purification. Canadian Journal of Biochemistry and Physiology 1959; 37(8), 911-917.
BJ Holub and CM Skeaff. Nutritional regulation of cellular phosphatidylinositol. Methods in Enzymology 1987; 141, 234-244.
HI Ayad, IA Matter, MM Gharieb and OM Darwesh. Bioflocculation harvesting of oleaginous microalga Chlorella sp. using novel lipid-rich cellulolytic fungus Aspergillus terreus (MD1) for biodiesel production. Biomass Conversion and Biorefinery 2023; 14, 30315-30327.
KN Thermozier, MW Fawley and KP Fawley. A new terrestrial species of Diplosphaera (Trebouxiophyceae), Diplosphaera sundellii, from a sodic-saline slick in Warren Prairie Natural Area, Arkansas, USA. Fottea 2024; 24(1), 109-117.
R Muñoz and B Guieysse. Algal-bacterial processes for the treatment of hazardous contaminants: A review. Water Research 2006; 40(15), 2799-2815.
B George, I Pancha, C Desai, K Chokshi, C Paliwal, T Ghosh and S Mishra. Effects of different media composition, light intensity and photoperiod on morphology and physiology of freshwater microalgae Ankistrodesmus falcatus - a potential strain for biofuel production. Bioresource Technology 2014; 171, 367-374.
NW Rasdi and JG Qin. Effect of N:P ratio on growth and chemical composition of Nannochloropsis oculata and Tisochrysis lutea. Journal of Applied Phycology 2015; 27, 2221-2230.
WG Sunda, NM Price and FMM Morel. Trace metal ion buffers and their use in culture studies. In: RA Andersen (Ed.). Algal culturing techniques. Academic Press, New York, 2005, p. 35-63.
HA Aratboni, N Rafiei, R Garcia-Granados, A Alemzadeh and JR Morones-Ramírez. Biomass and lipid induction strategies in microalgae for biofuel production and other applications. Microbial Cell Factories 2019; 18, 178.
A Karemore, R Pal and R Sen. Strategic enhancement of algal biomass and lipid in Chlorococcum infusionum as bioenergy feedstock. Algal Research 2013; 2(2), 113-121.
M Nayak, WI Suh, YK Chang and B Lee. Exploration of two-stage cultivation strategies using nitrogen starvation to maximize the lipid productivity in Chlorella sp. HS2. Bioresource Technology 2019; 276, 110-118.
SO Lourenco, E Barbarino, FJ Mancini, KP Schinke and E Aidar. Effects of different nitrogen sources on the growth and biochemical profile of 10 marine microalgae in batch culture: An evaluation for aquaculture. Phycologia 2002; 41(2), 158-168.
M Arumugam, A Agarwal, MC Arya and Z Ahmed. Influence of nitrogen sources on biomass productivity of microalgae Scenedesmus bijugatus. Bioresource Technology 2013; 131, 246-249.
J Zhan, Y Hong and H Hu. Effects of nitrogen sources and C/N ratios on the lipid-producing potential of Chlorella sp. HQ. Journal of Microbiology and Biotechnology 2016; 26(7), 1290-1302.
H Campos, WJ Boeing, BN Dungan and T Schaub. Cultivating the marine microalga Nannochloropsis salina under various nitrogen sources: Effect on biovolume yields, lipid content and composition, and invasive organisms. Biomass and Bioenergy 2014; 66, 301-307.
N Xu, X Zhang, X Fan, L Han and C Zeng. Effects of nitrogen source and concentration on growth rate and fatty acid composition of Ellipsoidion sp. (Eustigmatophyta). Journal of Applied Phycology 2001; 13(6), 463-469.
S Ho, X Ye, T Hasunuma, JS Chang and A Kondo. Perspectives on engineering strategies for improving biofuel production from microalgae - a critical review. Biotechnology Advances 2014; 32(8), 1448-1459.
Y Zhang, Q Wang, X Liu, H Zheng and A Li. Two types of growth pattern of the five microalgal species under different nitrogen supplies. Biomass and Bioenergy 2023; 171, 106720.
SK Lenka, N Carbonaro, R Park, SM Miller, I Thorpe and Y Li. Current advances in molecular, biochemical, and computational modeling analysis of microalgal triacylglycerol biosynthesis. Biotechnology Advances 2016; 34(5), 1046-1063.
I Pancha, K Chokshi, B George, T Ghosh, C Paliwal, R Maurya and S Mishra. Nitrogen stress triggered biochemical and morphological changes in the microalgae Scenedesmus sp. CCNM 1077. Bioresource Technology 2014; 156, 146-154.
Z Yu, H Pei, L Jiang, Q Hou, C Nie and L Zhang. Phytohormone addition coupled with nitrogen depletion almost tripled the lipid productivities in two algae. Bioresource Technology 2018; 247, 904-914.
K Jiao, W Xiao, X Shi, SH Ho, JS Chang, IS Ng, X Tang, Y Sun, X Zeng and L Lin. Molecular mechanism of arachidonic acid biosynthesis in Porphyridium purpureum promoted by nitrogen limitation. Bioprocess and Biosystems Engineering 2021; 44, 1491-1499.
MD Kavitha, S Kathiresan, S Bhattacharya and R Sarada. Culture media optimization of Porphyridium purpureum: Production potential of biomass, total lipids, arachidonic and eicosapentaenoic acid. Journal of Food Science and Technology 2016; 53, 2270-2278.
BJ Krieg, SM Taghavi, GL Amidon and GE Amidon. In vivo predictive dissolution: Comparing the effect of bicarbonate and phosphate buffer on the dissolution of weak acids and weak bases. Journal of Pharmaceutical Sciences 2015; 104(9), 2894-2904.
YY Choi, JM Joun, J Lee, ME Hong, HM Pham, WS Chang and SJ Sim. Development of large-scale and economic pH control system for outdoor cultivation of microalgae Haematococcus pluvialis using industrial flue gas. Bioresource Technology 2017; 244, 1235-1244.
G Su, K Jiao, Z Li, X Guo, J Chang, T Ndikubwimana, Y Sun, X Zeng, Y Lu and L Lin. Phosphate limitation promotes unsaturated fatty acids and arachidonic acid biosynthesis by microalgae Porphyridium purpureum. Bioprocess and Biosystems Engineering 2016; 39, 1129-1136.
K Muylaert, A Beuckels, O Depraetere, I Foubert, G Markou and D Vandamme. Wastewater as a source of nutrients for microalgae biomass production. Biomass and Biofuels from Microalgae. Springer Nature, Germany, 2015, p. 75-94.
JC de Carvalho, DT Molina-Aulestia, WJ Martinez-Burgos, SG Karp, MC Manzoki, ABP Medeiros, C Rodrigues, T Scapini, LPDS Vandenberghe, S Vieira, AL Woiciechowski, VT Soccol and CR Soccol. Agro-industrial wastewaters for algal biomass production, bio-based products, and biofuels in a circular bioeconomy. Fermentation 2022; 8(12), 728.
MA Ahanger, MN Alyemeni, L Wijaya, SA Alamri, P Alam, M Ashraf and P Ahmad. Potential of exogenously sourced kinetin in protecting Solanum lycopersicum from NaCl-induced oxidative stress through up-regulation of the antioxidant system, ascorbate-glutathione cycle and glyoxalase system. PLoS One 2018; 13(9), e0202175.
M Hagemann. Molecular biology of cyanobacterial salt acclimation. FEMS Microbiology Reviews 2011; 35(1), 87-123.
D Remias, C Nicoletti, K Krennhuber, B Möderndorfer, L Nedbalová and L Procházková. Growth, fatty, and amino acid profiles of the soil alga Vischeria sp. E71. 10 (Eustigmatophyceae) under different cultivation conditions. Folia Microbiologica 2020; 65, 1017-1023.
I Pancha, K Chokshi, R Maurya, K Trivedi, SK Patidar, A Ghosh and S Mishra. Salinity induced oxidative stress enhanced biofuel production potential of microalgae Scenedesmus sp. CCNM 1077. Bioresource Technology 2015; 189, 341-348.
MA Sinetova, RA Sidorov, AA Medvedeva, AY Starikov, AG Markelova, SI Allakhverdiev and DA Los. Effect of salt stress on physiological parameters of microalgae Vischeria punctata strain IPPAS H-242, a superproducer of eicosapentaenoic acid. Journal of Biotechnology 2021; 331, 63-73.
Z Cohen, A Vonshak and A Richmond. Effect of environmental conditions on fatty acid composition of the red alga Porphyridium cruentum: Correlation to growth rate. Journal of Phycology 1988; 24(3), 328-332.
H Suzuki, CJ Hulatt, RH Wijffels and V Kiron. Growth and LC-PUFA production of the cold-adapted microalga Koliella antarctica in photobioreactors. Journal of Applied Phycology 2019; 31(2), 981-997.
C Bigogno, I Khozin-Goldberg, S Boussiba, A Vonshak and Z Cohen. Lipid and fatty acid composition of the green oleaginous alga Parietochloris incisa, the richest plant source of arachidonic acid. Phytochemistry 2002; 60(5), 497-503.
AE Solovchenko, I Khozin-Goldberg, S Didi-Cohen, Z Cohen and MN Merzlyak. Effects of light intensity and nitrogen starvation on growth, total fatty acids and arachidonic acid in the green microalga Parietochloris incisa. Journal of Applied Phycology 2008; 20, 245-251.
WZ Cheng, Z Cohen, GI Khozin and A Richmond. Characterization of growth and arachidonic acid production of Parietochloris incisa comb. nov (Trebouxiophyceae, Chlorophyta). Journal of Applied Phycology 2002; 14, 453-460.
B Zorin, D Pal-Nath, A Lukyanov, S Smolskaya, S Kolusheva, S Didi-Cohen, S Boussiba, Z Cohen, I Khozin-Goldberg and A Solovchenko. Arachidonic acid is important for efficient use of light by the microalga Lobosphaera incisa under chilling stress. Biochimica et Biophysica Acta (BBA) - Molecular and Cell Biology of Lipids 2017; 1862(9), 853-868.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






