Enhanced Fluoride Removal from Groundwater Using Napier Grass-Derived Adsorbent: Experimental and DFT Study
DOI:
https://doi.org/10.48048/tis.2025.9217Keywords:
Adsorption, Fluoride, Fluorosis, Napier grass, Alkaline modification, Biochar, DFTAbstract
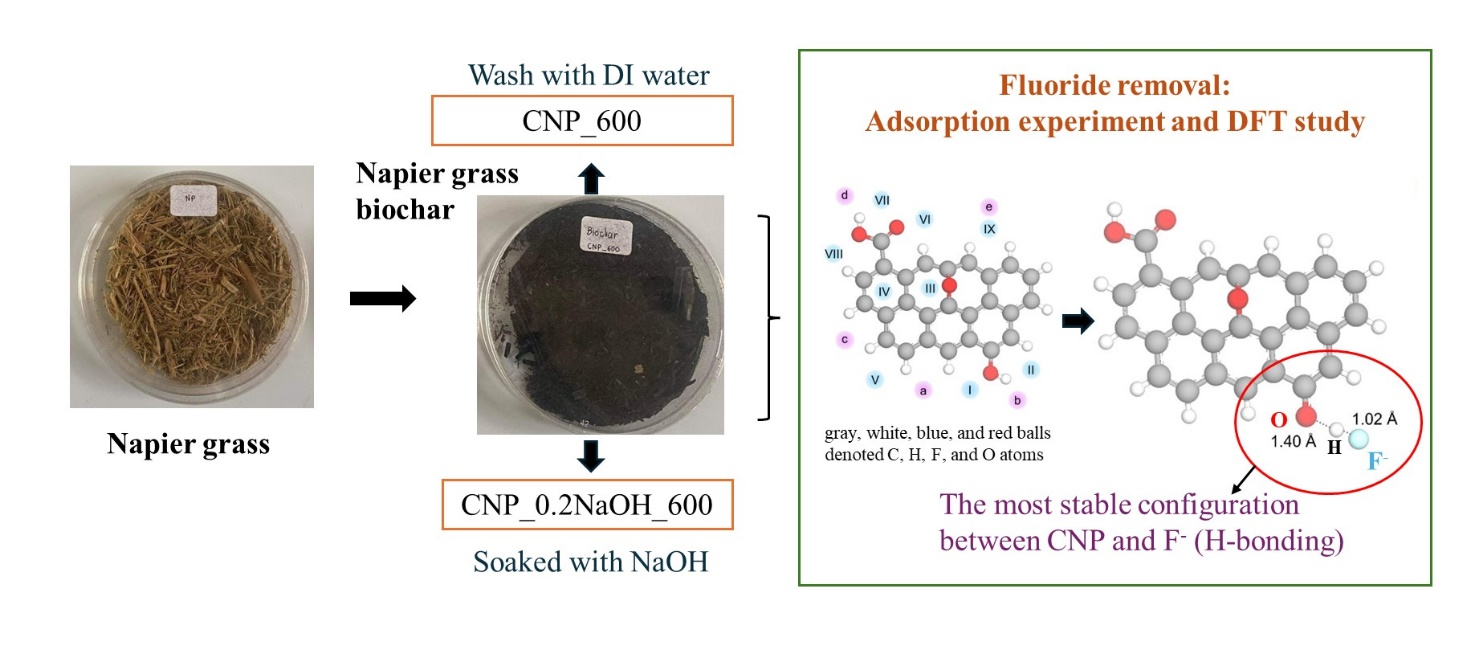
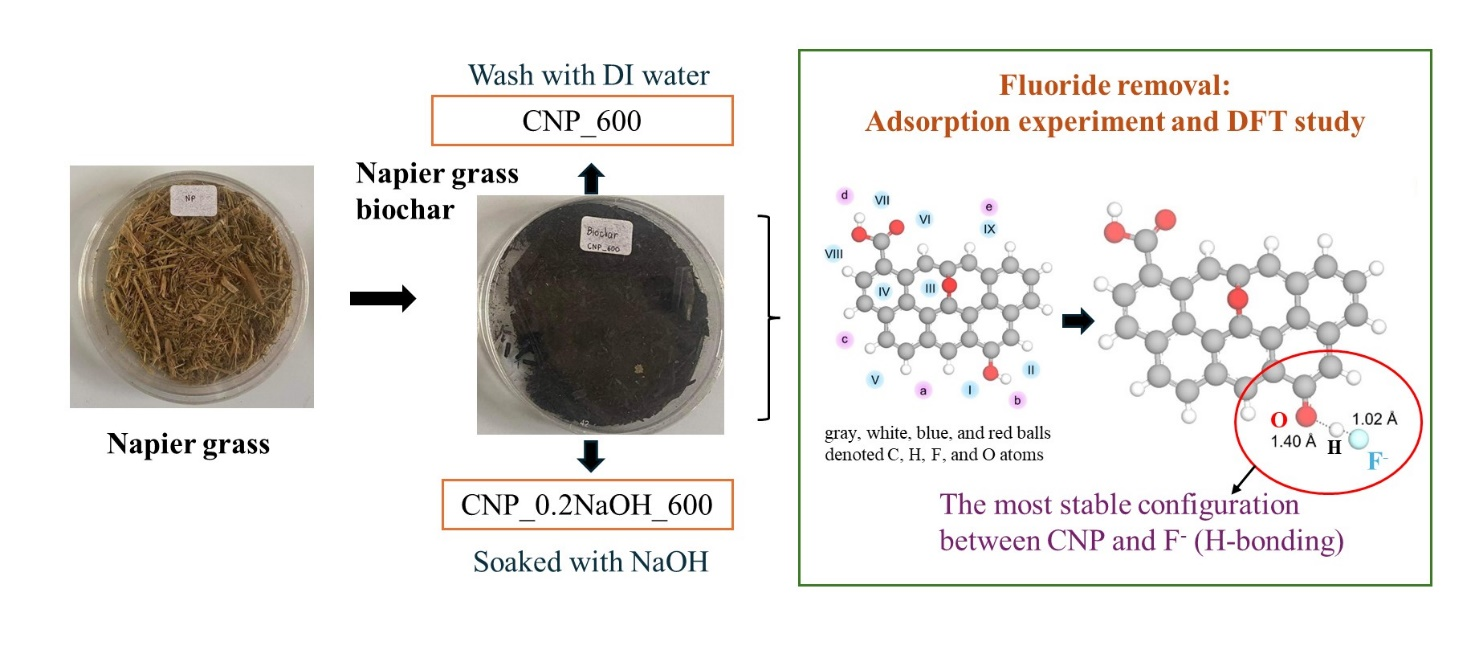
Lab-scale experiments and Density Functional Theory (DFT) studies were carried out to investigate the efficiency of Napier grass-derived adsorbent (CNP_600) and alkaline modification (CNP_0.2NaOH_600) as low-cost adsorption materials for fluoride removal. CNP that had been treated with an alkaline solution cloud contained more fluoride than the CNP that had been used initially. The NaOH modification of prepared materials promoted a change in surface characteristics that could enhance fluoride removal efficiency. Those adsorbents fitted well with the pseudo-2nd-order kinetic model and declared that the adsorption process was chemisorption. The isotherm study exhibited the best fit with the Langmuir model, resulting in mono-layer adsorption. The experimental adsorption behavior of fluoride ions (F–) onto CNPs was investigated. The proposed adsorption process involved electrostatic attraction and hydrogen bonding, which were found to be consistent with the results of a Density Functional Theory (DFT) study. The results indicated that the -OH and -COOH groups on the CNPs displayed electron acceptors from the F– ions. Additionally, the fluoride removal of actual groundwater from the prepared adsorbents was 17 %. The small percentage of adsorption efficiency in groundwater was because of pH and co-exiting ions. Finally, the use of biomass waste as a biochar adsorbent to control the fluoride excess in groundwater in the problematic area could promote the sustainability of water safety for the community, especially in the northern part of Thailand.
HIGHLIGHTS
- The adsorption process removes fluoride using Napier grass biochar (CNP_600) and NaOH functionalization (CNP_0.2NaOH_600).
- Based on the function groups of the materials, we can use the DFT study results to confirm the behavior of F–ions in the biomass samples.
- The experimental study's adsorption mechanism followed a similar pattern to the DFT studies. This confirms that CNPs help F–ions form strong H-bonds.
- The O-H bond lengthened from 0.97 Å to 1.40 Å, confirming the strong interaction between CNP and fluoride ions.
GRAPHICAL ABSTRACT
Downloads
References
P Loganathan, S Vigneswaran, J Kandasamy and R Naidu. Defluoridation of drinking water using adsorption processes. Journal of Hazardous Materials 2013; 248-249, 1-19.
M Jiménez-Reyes and M Solache-Ríos. Sorption behavior of fluoride ions from aqueous solutions by hydroxyapatite. Journal of Hazardous Materials 2010; 180(1-3), 297-302.
A Bhatnagar, E Kumar and M Sillanpää. Fluoride removal from water by adsorption-a review. Chemical Engineering Journal 2011; 171(3), 811-840.
WHO. Guidelines for drinking-water quality: Fourth edition incorporating the first and second addenda. World Health Organization, Geneva, Switzerland, 2017.
C Rojanaworarit, L Claudio, N Howteerakul, A Siramahamongkol, P Ngernthong, P Kongtip and S Woskie. Hydrogeogenic fluoride in groundwater and dental fluorosis in Thai agrarian communities: A prevalence survey and case-control study. BMC Oral Health 2021; 21(1), 545.
N Theerawasttanasiri, S Taneepanichskul, W Pingchai, Y Nimchareon and S Sriwichai. Implementing a geographical information system to assess endemic fluoride areas in Lamphun, Thailand. Risk Management and Healthcare Policy 2018; 11, 15-24.
R Kumar, P Sharma, PK Rose, PK Sahoo, P Bhattacharya, A Pandey and M Kumar. Co-transport and deposition of fluoride using rice husk-derived biochar in saturated porous media: Effect of solution chemistry and surface properties. Environmental Technology & Innovation 2023; 30, 103056.
KK Shimabuku, ME Baumgardner, RB Bahr, NR Frojelin, AM Kennedy, KT Nolan and NE Stanton. Fluoride removal in batch and column systems using bone char produced in a top-lit updraft drum gasifier and furnace. Water Research 2023; 244, 120332.
NA Tajuddin, EFB Sokeri, NA Kamal and M Dib. Fluoride removal in drinking water using layered double hydroxide materials: Preparation, characterization and the current perspective on IR4.0 technologies. Journal of Environmental Chemical Engineering, 2023; 11(3), 110305.
MM Damtie, YC Woo, B Kim, RH Hailemariam, KD Park, HK Shon, C Park and JS Choi. Removal of fluoride in membrane-based water and wastewater treatment technologies: Performance review. Journal of Environmental Management 2019; 251, 109524.
J He, Y Yang, Z Wu, C Xie, K Zhang, L Kong and J Liu. Review of fluoride removal from water environment by adsorption. Journal of Environmental Chemical Engineering 2020; 8(6), 104516.
JL Marco-Brown, M Melotta, M Fernández and A Iriel. Arsenic and fluoride adsorption from multielement solutions onto aluminum-modified montmorillonite. Groundwater for Sustainable Development 2024; 26, 101205.
J Shen and A Schäfer. Removal of fluoride and uranium by nanofiltration and reverse osmosis: A review. Chemosphere 2014; 117, 679-691.
F Aziz, I Din, F Khan, P Manan, A Sher and S Hakim. Treatment of fluoride-contaminated water using mango (Mangifera indica) leaves powder as an adsorbent. Current Research in Green and Sustainable Chemistry 2023; 6(25), 100359.
R Kumar, P Sharma, W Yang, M Sillanpää, J Shang, P Bhattacharya, M Vithanage and JP Maity. State-of-the-art research progress on adsorptive removal of fluoride-contaminated water using biochar-based materials: Practical feasibility through reusability and column transport studies. Environmental Research 2022; 214(4), 114043.
Q Wang, Y Li, Z Yu, X Li, S Yin, W Ji, Y Hu, W Cai and X Wang. Highly porous carbon derived from hydrothermal-pyrolysis synergistic carbonization of biomass for enhanced CO2 capture. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2023; 673, 131787.
AH Jawad, AS Waheeb, RA Rashid, WI Nawawi and E Yousif. Equilibrium isotherms, kinetics, and thermodynamics studies of methylene blue adsorption on pomegranate (Punica granatum) peels as a natural low-cost biosorbent. Desalination and Water Treatment 2018; 105, 322-331.
H Shao, Y Li, L Zheng, T Chen and J Liu. Removal of methylene blue by chemically modified defatted brown algae Laminaria japonica. Journal of the Taiwan Institute of-Chemical Engineers 2017; 80, 525-532.
AD Becke. Density-functional exchange-energy approximation with correct asymptotic behavior. Physical Review A 1988; 38(6), 3098-3100.
S Grimme, J Antony, S Ehrlich and H Krieg. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. The Journal of Chemical Physics 2010; 132(15), 154104.
M Odelius, M Bernasconi and M Parrinello. Two-dimensional ice adsorbed on mica surface. Physical Review Letters 1997; 78(14), 2855-2858.
SH Vosko, L Wilk and M Nusair. Accurate spin-dependent electron liquid correlation energies for local spin density calculations: A critical analysis1. Canadian Journal of Physics 1980; 58, 1-12.
Y Qi, Z Zhou, R Xu, Y Dong, Z Zhang and M Liu. Effect of NaOH pretreatment on permeability and surface properties of three wood species. ACS Omega 2023; 8(43), 40362-40374.
P Induvesa, R Rattanakom, S Sriboonnak., C Pumas, K Duangjan, P Rakruam, S Nitayavardhana, P Sittisom and A Wongrueng. Adsorption of fluoride onto acid-modified low-cost pyrolusite ore: Adsorption characteristics and efficiencies. International Journal of Environmental Research and Public Health 2022; 19(24), 17103.
AK Yadav, AK Chaubey, S Kapoor, T Pratap, B Preetiva, V Vimal and D Mohan. Sustainable Napier grass (Pennisetum purpureum) biochar for the Sorptive removal of acid orange 7 (AO7) from water. Processes 2024; 12(6), 1115.
J Qin, J Wang, J Long, J Huang, S Tang, H Hou and P Peng. Recycling of heavy metals and modification of biochar derived from Napier grass using HNO3. Journal of Environmental Management 2022; 318(62-68), 115556.
R Qadeer, J Hanif, M Saleem and M Afzal. Characterization of activated-charcoal. Journal of the Chemical Society of Pakistan 1994; 16(4) 229-235.
A Shaaban, SM Se, NMM Mitan and MF Dimin. Characterization of biochar derived from rubber wood sawdust through slow pyrolysis on surface porosities and functional groups. Procedia Engineering 2013; 68, 365-371.
W Tongpoothorn, M Sriuttha, P Homchan, S Chanthai and C Ruangviriyachai. Preparation of activated carbon derived from Jatropha curcas fruit shell by simple thermo-chemical activation and characterization of their physico-chemical properties. Chemical Engineering Research and Design 2011; 89(3), 335-340.
R Khezri, WAWAK Ghani, DRA Biak, R Yunus and K Silas. Experimental evaluation of Napier grass gasification in an auto thermal bubbling fluidized bed reactor. Energies 2019; 12(8), 1517.
HVRD Santos, PS Scalize, FJC Teran and RMF Cuba. Fluoride removal from aqueous medium using biochar produced from coffee ground. Resources 2023; 12(7), 84.
S Guilhen, T Watanabe, T Silva, S Rovani, J Marumo, J Tenório, O Masek and LD Araujo. Role of point of zero charge in the adsorption of cationic textile dye on standard Biochars from aqueous solutions: Selection criteria and performance assessment. Recent Progress in Materials 2022; 4(2), 1.
Y Ren, M He, G Qu, N Ren, P Ning, Y Yang, X Chen, Z Wang and Y Hu. Study on the mechanism of removing fluoride from wastewater by oxalic acid modified aluminum ash-carbon slag-carbon black doped composite. Arabian Journal of Chemistry 2023; 16(5), 104668.
DSGD Senewirathna, S Thuraisingam, S Prabagar and J Prabagar. Fluoride removal in drinking water using activated carbon prepared from palmyra (Borassus flabellifer) nut shells. Current Research in Green and Sustainable Chemistry 2022; 5(6), 100304.
NT Hue and TN Hoang. Study about doping ion La3+ onto the surface of pyrolusite ore for removing simultaneously both fluoride and phosphate from wastewater. Journal of Chemistry 2017; 2017(2), 11.
TL Tan, PA Krusnamurthy, H Nakajima and SA Rashid. Adsorptive, kinetics and regeneration studies of fluoride removal from water using zirconium-based metal-organic frameworks. RSC Advances 2020; 10(32), 18740-18752.
M Yousefi, S Arami, H Takallo, M Hosseini, M Radfarad, H Soleimani and A Mohammadi. Modification of pumice with HCl and NaOH enhancing its fluoride adsorption capacity: Kinetic and isotherm studies. Human and Ecological Risk Assessment: An International Journal 2018; 25(6), 1-13.
M Regassa, F Melak, W Birke and E Alemayehu. Defluoridation of water using natural and activated coal. International Advanced Research Journal in Science, Engineering and Technology 2016; 3(1), 1-7.
HVRD Santos, PS Scalize, FJC Teran and RMF Cuba. Fluoride removal from aqueous medium using biochar produced from coffee ground. Resources 2023; 12(7), 84.
S Bibi, A Farooqi, A Yasmin, MA Kamran and NK Niazi. Arsenic and fluoride removal by potato peel and rice husk (PPRH) ash in aqueous environments. International Journal of Phytoremediation 2017; 19(11), 1029-1036.
D Mohan, R Sharma, VK Singh, P Steele and CU Pittman. Fluoride removal from water using bio-char, a green waste, low-cost adsorbent: Equilibrium uptake and sorption dynamics modeling. Industrial & Engineering Chemistry Research 2012; 51(2), 900-914.
Y Wei, L Wang, H Li, W Yan and J Feng. Synergistic fluoride adsorption by composite adsorbents synthesized from different types of materials-a review. Frontiers in Chemistry 2022; 10, 900660.
J Hoslett, H Ghazal, N Mohamad and H Jouhara. Removal of methylene blue from aqueous solutions by biochar prepared from the pyrolysis of mixed municipal discarded material. Science of The Total Environment 2020; 714, 136832.
H Yu, Y Zhang, L Wang, Y Tuo, S Yan, J Ma, X Zhang, Y Shen, H Guo and L Han. Experimental and DFT insights into the adsorption mechanism of methylene blue by alkali-modified corn straw biochar. RSC Advances 2024; 14(3), 1854-1865.
Z Zhao, T Nie and W Zhou. Enhanced biochar stabilities and adsorption properties for tetracycline by synthesizing silica-composited biochar. Environmental Pollution 2019; 254, 113015.
B Sawangjang, P Induvesa, A Wongrueng, C Pumas, S Wattanachira, P Rakruam, P Punyapalakul, S Takizawa and E Khan. Evaluation of fluoride adsorption mechanism and capacity of different types of bone char. International Journal of Environmental Research and Public Health 2021; 18(13), 6878.
A Wongrueng, B Sookwong, P Rakruam and S Wattanachira. Kinetic adsorption of fluoride from an aqueous solution onto a dolomite sorbent. Engineering Journal 2016; 20(3), 1-9.
N Nabbou, M Belhachemi, M Boumelik, T Merzougui, D Lahcene, Y Harek, AA Zorpas and M Jeguirim. Removal of fluoride from groundwater using natural clay (kaolinite): Optimization of adsorption conditions. Comptes Rendus Chimie 2019; 22(2-3), 105-112.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






