Effect of Cold Shock on Larval Viability and Tetraploid Percentage of Asian Redtail Catfish, Hemibagrus nemurus (Valenciennes, 1840)
DOI:
https://doi.org/10.48048/tis.2025.9204Keywords:
Cold shock, Hemibagrus nemurus, Induced polyploid, TetraploidAbstract
This study aimed to evaluate the effects of cold shock time and temperature on larval viability and the formation of tetraploid Asian redtail catfish, Hemibagrus nemurus (Valenciennes, 1840). A completely randomized factorial design was used with 2 factors, shock time after fertilization (AF) which consisted of 5 levels (26th, 27th, 28th, 29th, and 30th min AF) and shock temperature with 3 levels (6, 8, and 10 °C). Furthermore, each group carried out in 3 replicates, and the observed parameters including the percentage of tetraploid fish, fertilization rate, hatching, larval survival, and percentage of abnormal larvae recorded. The identification of tetraploid individuals was based on the analysis of the total nucleoli, erythrocyte size, and DNA content by flow cytometry. The results showed that cold temperature shock had an effect on hatching rate, survival rate, and the number of abnormal larvae (p < 0.05) but had no effect on egg fertilization rate (p > 0.05). The highest hatching rate was obtained in temperature shock treatment at 10 °C (60.06 ± 0.85 %), while the highest survival was obtained in temperature shock treatment at 10 °C (68.33 ± 1.38 %). Moreover, the shock time AF and temperature shock treatments influenced the formation of tetraploid the Asian redtail catfish (p < 0.05). Shock treatment at 28 min AF with a shock temperature of 6 °C for 3 min produced the highest tetraploid percentage (78.33 ± 1.67 %, p < 0.05). Tetraploid the Asian redtail catfish had a maximum number of 4 nucleoli per cell, while diploid catfish had a maximum of 2. The erythrocyte and nucleus volumes were 98.26 and 99.51 %, respectively, which were larger than those of the diploid the Asian redtail catfish. Furthermore, the DNA content in the tetraploid individuals was twice that in the diploid individuals. Thus, cold temperature shock at 6 °C for 3 min to the 28-minute-old embryos to produce a tetraploid the Asian redtail catfish is recommended.
HIGHLIGHTS
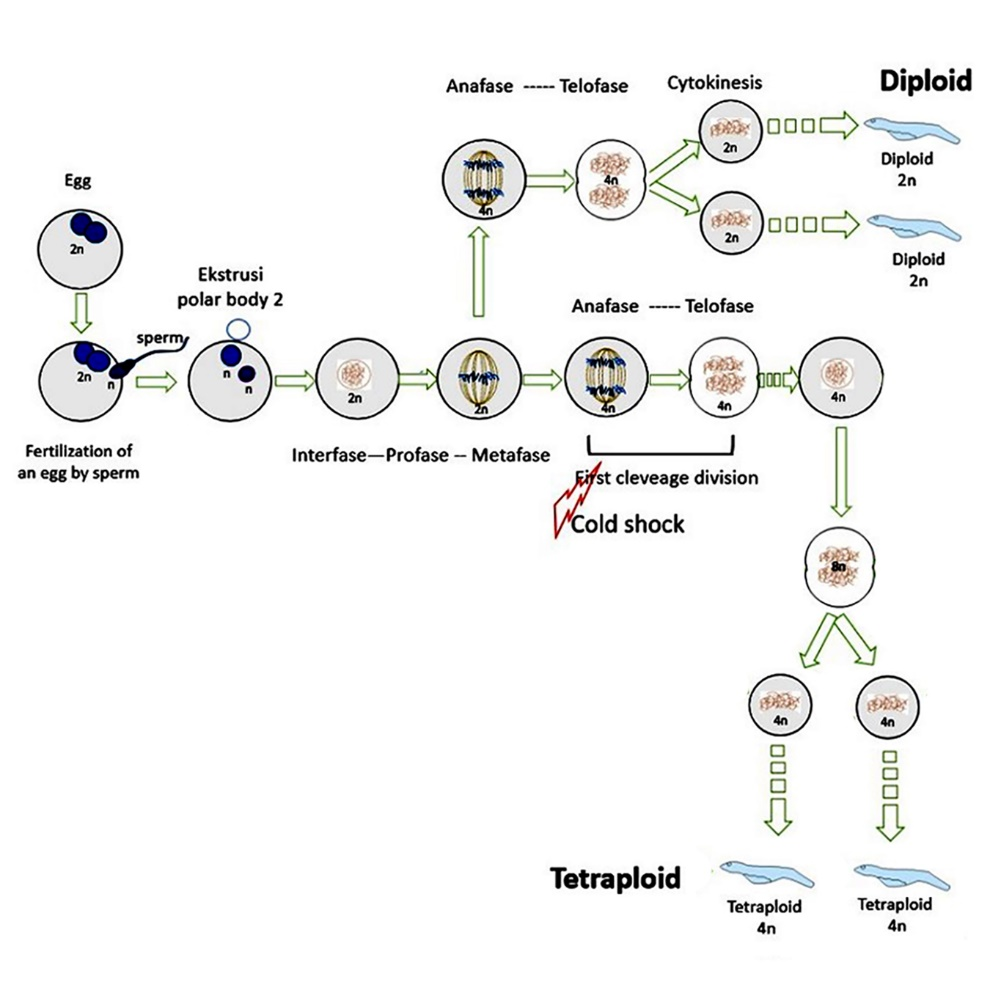
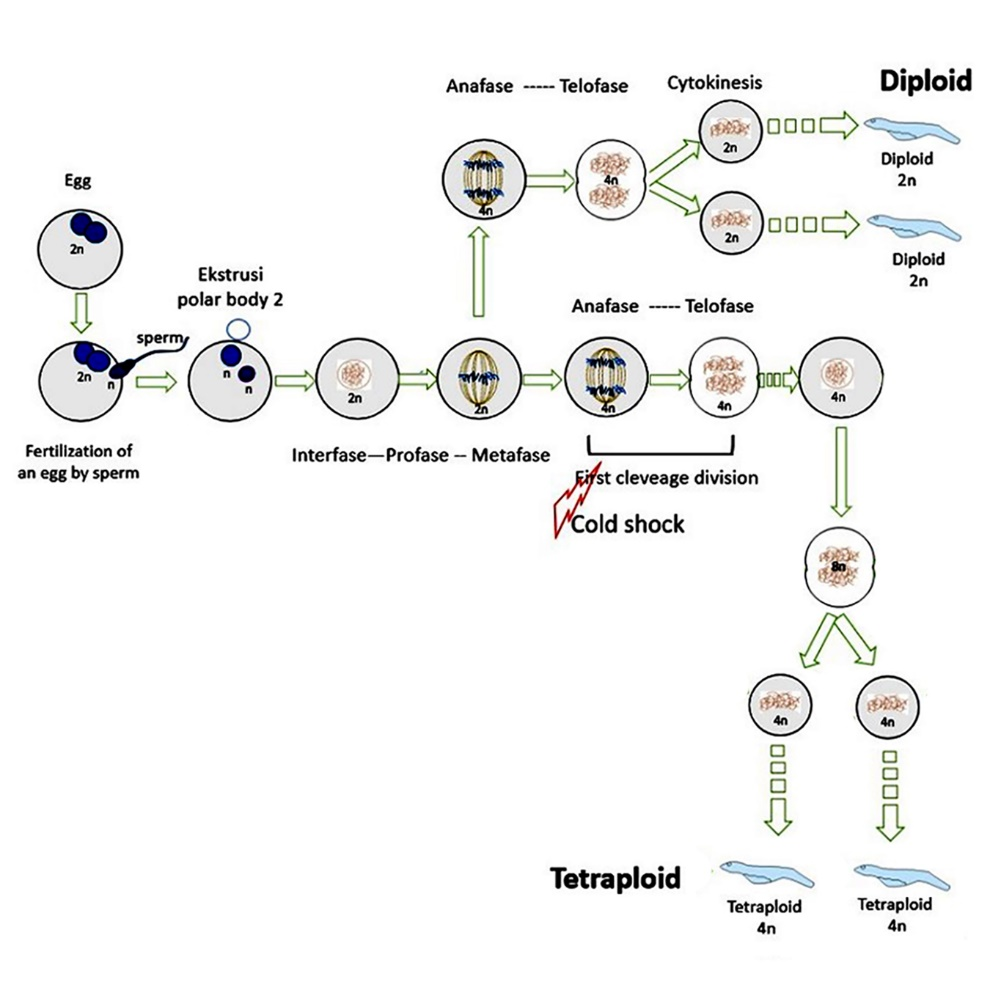
- This article focuses on the application of cold shock to the formation of tetraploid Asian Redtail Catfish
- Cold shock in embryos can produce tetraploid Asian Redtail Catfish
- Tetraploid in Asian Redtail Catfish is an intermediate process to produce triploid Asian Redtail Catfish
- The success of tetraploid induction of Asian Redtail Catfish is affected by shock time and shock temperature
GRAPHICAL ABSTRACT
Downloads
References
A Nuryanto, N Komalawati and Sugiharto. Genetic diversity assessment of Hemibagrus nemurus from rivers in Java Island, Indonesia using COI gene. Biodiversitas 2019; 20(9), 2707-2717.
II Kusmini, KA Hari, A Widiyati and FP Putri. Growth and development of Bagrid catfish gonads (Hemibagrus nemurus) offspring G-1, G-2 dan G-3. Jurnal Riset Akuakultur 2019; 14(4), 201-211.
K Arai and T Fujimoto. Chromosome manipulation techniques and applications to aquaculture. In: HP Wang, F Piferrer, SL Chen and ZG Shen (Eds.). Sex control in aquaculture. John Wiley & Sons, New Jersey, 2019, p. 137-162.
A Hassan, VT Okomoda and MN Nurhayati. Embryonic development of diploid and triploid eggs of Clarias gariepinus (Burchell, 1822). Caryologia 2018; 71(4), 372-379.
A Hassan, VT Okomoda and PJ Pradeep. Triploidy induction by electric shock in red hybrid tilapia. Aquaculture 2018; 495, 823-830.
J Normala, AA Mohd, MAB Abol, AA Nur, W Khor, TV Okomoda and MS Shahreza. Morphometric variations between triploid and diploid Clarias gariepinus (Burchell, 1822). Croatian Journal of Fisheries 2017; 75, 113-121.
L Zhou and J Gui. Natural and artificial polyploids in aquaculture. Aquaculture and Fisheries 2017; 2(3), 103-111.
DT Fox, DE Soltis, PS Soltis, TL Ashman and YVD Peer. Polyploidy: A biological force from cells to ecosystems. Trends in Cell Biology 2020; 30(9), 688-694.
VT Okomoda, JP Pradeep, AS Oladimeji, AB Abol-Munafi, KI Alabi, M Ikhwanuddin, C Martins, JA Umaru and A Hassan. Effect of electric induced triploidization on sex ratio, growth and gonad histology of red hybrid tilapia. Aquaculture 2020; 520, 734991.
M Ruiz, J Oustric, J Santini and R Morillon. Synthetic polyploidy in grafted crops. Frontiers in Plant Science 2020; 11, 540894.
K Xu, W Duan, J Xiao, M Tao, C Zhang, Y Liu and S Liu. Development and application of biological technologies in fish genetic breeding. Science China Life Sciences 2015; 58(2), 187-201.
IS Park, HW Gil, TH Lee, YK Nam and DS Kim. Comparative study of growth and gonad maturation in diploid and triploid marine medaka, Oryzias dancena. Development & Reproduction 2016; 20(4), 305-314.
B Gomelsky, KJ Schneider, A Anil and TA Delomas. Gonad development in triploid ornamental koi carp and results of crossing triploid females with diploid males. North American Journal of Aquaculture 2015; 77(1), 96-101.
S Chatchaiphan, P Srisapoome and U Na-Nakorn. Effects of strains on growth performances of triploid bighead catfish, Clarias macrocephalus Günther, 1864. Agriculture and Natural Resources 2016; 50(4), 299-305.
H Liu, Z Wu, X Zhu, Z Song, J Hu, L Wang, J Li and F You. Comparative performance of growth, vertebral structure and muscle composition in diploid and triploid Paralichthys olivaceus. Journal of Fish Diseases 2018; 41(10), 1495-1504.
LS Sato, PH Jorge, VA Mastrochirico-Filho, ME Hata, SR Batlouni, RY Kuradomi, F Porto-Foresti, F Foresti and DT Hashimoto. Triploidy in tambaqui Colossoma macropomum identified by chromosomes of fish larvae. Journal of Aquaculture & Marine Biology 2020; 9(3), 65-69.
H Otterå. A Thorsen, Ø Karlsen, PG Fjelldal, HC Morton and GL Taranger. Performance of triploid Atlantic cod (Gadus morhua L.) in commercial aquaculture. Aquaculture 2016; 464, 699-709.
KA Glover, JB Bos, K Urdal, AS Madhun, AGE Sørvik, L Unneland, BB Seliussen, Ø Skaala, OT Skilbrei, Y Tang and V Wennevik. Genetic screening of farmed Atlantic salmon escapees demonstrates that triploid fish display reduced migration to freshwater. Biological Invasions 2016; 18, 1287-1294.
Z Wu, L Wang, Y Lu, X Zhu, X Yue and F You. Artificial induction and genetic structure analysis of tetraploid turbot Scophthalmus maximus. Frontiers in Marine Science 2019; 6, 637.
LP Oliver, J Ma, TJ Bruce, JT Evavold, DB Korbel and KD Cain. Triploid induction in cultured burbot (Lota lota) using thermal and hydrostatic shock. Aquaculture 2020; 515, 734582.
BL Peachey and SKJ Allen. Evaluation of cytochalasin B and 6-dimethylaminopurine for tetraploidy induction in the Eastern oyster, Crassostrea virginica. Aquaculture 2016; 450, 199-205.
Y Jiang, W Xu, H Luo, Hao Luo, C Ji, B Tao, D Luo, C Zhu, Z Zhu, Y Song and W Hu. Hydrostatic pressure shock induced tetraploids in rice field eel (Monopterus albus). Aquaculture 2024; 584, 740636.
Z Zhang, H Yi, Y Su, C Huang, X Wei, Q Chen, J Chen, H Li, S Bi, H Lai, X Liu, S Liu, Z Zeng, L Han, Y Zhang and G Li. Hydrostatic pressure shock induced diploid/tetraploid mosaic in mandarin fish (Siniperca chuatsi), with the observation of embryo development and change in body spots. Aquaculture 2023; 563, 738989.
X Zhu, Z Lin, Z Wu, J Li and F You. Effect of initiation time of hydrostatic pressure shock on chromosome set doubling of tetraploidization in turbot Scophthalmus maximus. Marine Biotechnology 2017; 19(5), 528-540.
AI Bazaz, I Ahmad, Nafath-Ul-Arab, TH Shah, OA Asimi, BA Bhat, Z Yousuf, SH Baba, N Razak and A Fatima. A review on induction of triploidy in fish using heat, pressure and cold shock treatments. Journal of Entomology and Zoology Studies 2020; 8(2), 381-385.
F Hu, J Fan, Q Qin, Y Huo, Y Wang, C Wu, Q Liu, W Li, X Chen, C Liu, M Tao, S Wang, R Zhao, K Luo and S Liu. The sterility of allotriploid fish and fertility of female autotriploid fish. Frontiers in Genetics 2019; 10, 377.
F Hu, H Zhong, J Fan, C Wu, S Wang, Y Wang, K Luo, R Zhao, J Liu, Q Qin, C Tang and S Liu. A new type of tetraploid fish derived via female autotetraploid × male allotetraploid hybridization. Aquaculture 2020; 524, 735244.
GM Weber, MA Hostuttler, KJ Semmens and BA Beers. Induction and viability of tetraploids in brook trout (Salvelinus fontinalis). Canadian Journal of Fisheries and Aquatic Sciences 2015; 72(10), 1443-1449.
WK Hershberger and MA Hostuttler. Protocols for more effective induction of tetraploid rainbow trout. North American Journal of Aquaculture 2007; 69(4), 367-372.
O Carman, AT Mukti, MJ Zairin and Alimudin. Reproductive performances of triploid male and female Nile tilapia Oreochromis niloticus (Linnaeus, 1758) at different ages. Biodiversitas 2023; 24(8), 4235-4242.
AT Mukti, O Carman, A Alimuddin, MJ Zairin and MA Suprayudi. Growth performance, survival rate, flesh, and proximate composition of sex-grouped triploid and diploid Nile tilapia, Oreochromis niloticus. Turkish Journal of Veterinary & Animal Sciences 2020; 44(2), 290-298.
JG Christopher, R Siva and P Ezilrani. The effect of heat shock and timing on the induction of tetraploidy in Catfish, Heteropnestus fossilis. Journal of Applied Aquaculture 2019; 32(2), 186-192.
GM Weber, MA Hostuttler, BM Cleveland and TD Leeds. Growth performance comparison of intercross-triploid, induced triploid, and diploid rainbow trout. Aquaculture 2014; 433, 85-93.
P Hartami, O Carman, A Alimuddin, MJ Zairin, R Rahman and Hermawan. Performance analysis of tetraploid striped catfish (Pangasianodon hypophthalmus) resulting from heat shock induction. Acta Aquatica: Aquatic Sciences Journal 2021; 8(1), 43-48.
DP Hartono, P Witoko and N Purbosari. The effect of heat shock on the tetraploidy of catfish, Pangasius hypophthalmus. AACL Bioflux 2016; 9, 597-603.
F Ali and R Othman. The effect of heat shock on the embryonic development of tetraploid Clarias gariepinus. ASEAN Journal of Life Sciences 2022; 2(2), 22-31.
VT Okomoda, SO Olufeagba, GA Ataguba, SG Solomon, SA Oladimeji, A Hassan and AB Abol-Munafi. Tetraploid induction in Clarias gariepinus using cold shock protocol. Aquaculture 2021; 545, 737178.
NFD Nascimento, M Pereira-Santos, N Levy-Pereira, PS Monzani, D Niedzielski, T Fujimoto, JA Senhorini, LSO Nakaghi and GS Yasui. High percentages of larval tetraploids in the yellowtail tetra Astyanax altiparanae induced by heat-shock: The first case in Neotropical characins. Aquaculture 2020; 520, 734938.
SHA Tan, CP Teh, GO Chang and Z Yasin. Tetraploid induction in tropical oysters, Crassostrea belcheri (Sowerby) and Crassostrea iredalei (Faustino). Aquaculture Research 2017; 48, 1406-1412.
D Fopp-Bayat, G Chandra and A Nitkiewicz. How cold shock affects ploidy level and early ontogenetic development of the sterlet, A. Ruthenus L. International Journal of Molecular Sciences 2022; 23(1), 494.
A Syahril, O Carman and D Soelistyowati. Cold temperature shock tetraploidization of striped catfish Pangasianodon hypophtalmus. Jurnal Iktiologi Indonesia 2020; 20(1), 13-22.
RA Dunham. Aquaculture and fisheries biotechnology: Genetic approaches. CABI International, Wallingford, 2023.
S Ferosekhan, SK Sahoo, SS Giri, BK Das, BR Pillai and PC Das. Broodstock development, captive breeding and seed production of bagrid catfish, Mahanadi rita, Rita chrysea (Day, 1877). Aquaculture 2019; 503, 339-346.
J Normala, AA Mohd, MAB Abol. It is all in the blood: Erythrocyte characterization of triploid and diploid African catfish, Clarias gariepinus. Journal of Fisheries and Aquatic Science 2016; 11(6), 425-431.
SA Okunsebor, PC Ofojekwu, DG Kakwi and BS Audu. Effect of temperature on fertilization, hatching and survival rates of Heterobranchus bidorsalis eggs and hatchlings. British Journal of Applied Science & Technology 2015; 7(4), 372-376.
W Widyatmoko, H Effendi and NT Pratiwi. The growth and survival rate of Nile tilapia, Oreochromis niloticus (Linnaeus, 1758) in the aquaponic system with different vetiver (Vetiveria zizanioides L. Nash) plant density. Jurnal Iktiologi Indonesia 2019; 19(1), 157-166.
AT Mukti, M Ahmadi, Widjiat and EM Luqman. The different effects of heat shock duration and initial period on hatching rate, abnormality rate, egg yolk absorption, and survival rate of spotted barb (Puntius binotatus) larvae. IOP Conference Series: Earth and Environmental Science 2020; 441(1), 12085.
CA Bidwell, LC Chrisman and GS Libey. Polyploidy induced by heat shock in channel catfish. Aquaculture 1985; 51(1), 25-32.
N Jalil, MA Alim, AB Abol-Munafi, NA Ariffin, K Waiho and SM Sheriff. It is all in the blood: Erythrocyte characterization of triploid and diploid African catfish, Clarias gariepinus. Journal of Fisheries and Aquatic Science 2016; 11, 425-431.
J Normala, VT Okomoda, AA Mohd, AA Nur, AB Abol-Munafi and SM Sheriff. Genetic variation between triploid and diploid Clarias gariepinus (Burchell, 1822) using RAPD markers. Veterinary Sciences 2021; 8(5), 75.
A Adan, G Alizada, Y Kiraz, Y Baran and A Nalbant. Flow cytometry: Basic principles and applications. Critical Reviews in Biotechnology 2017; 37(2), 163-176.
H Yang and X Guo. Tetraploid induction by inhibiting mitosis I with heat shock, cold shock, and nocodazole in the hard clam Mercenaria mercenaria (Linnaeus, 1758). Marine Biotechnology 2006; 8(5), 501-510.
LF Martins, CC Hilbig, GS Yasui, PS Monzani, JA Senhorini, LSO Nakaghi and NFD Nascimento. Return temperature after heat shock affects the production of tetraploids in the yellowtail tetra Astyanax altiparanae. Zygote 2021; 29(1), 82-86.
ÉRD Alvarenga, AFA Fernandes, LR Lopes, TE Soares, GFDO Alves, FFBD Costa, SCMD Sales, GK Lima and EM Turra. Attempt to produce a Nile tilapia tetraploid line by heat shock induction. Aquaculture 2020; 529, 735647.
AR Foote, GC Mair, AT Wood and MJ Sellars. Tetraploid inductions of Penaeus monodon using cold shock. Aquaculture International 2012; 20(5), 1003-1007.
JP Alcántar-Vázquez, S Dumas, R Peña and AM Ibarra. Effects of cold-shock triploidy induction treatment on cell cleavage, hatching rate, and survival of triploid larvae of spotted sand bass (Paralabrax maculatofasciatus). Ciencias Marinas 2016; 42(2), 111-122.
F Piferrer, A Beaumont, JC Falguière, M Flajšhans, P Haffray and L Colombo. Polyploid fish and shellfish: Production, biology and applications to aquaculture for performance improvement and genetic containment. Aquaculture 2009; 293(3-4), 125-156.
MR Donaldson, SJ Cooke, DA Patterson and JS Macdonald. Cold shock and fish. Journal of Fish Biology 2008; 73(7), 1491-1530.
T Yabu, S Todoriki and M Yamashita. Stress-induced apoptosis by heat shock, UV and g-ray irradiation in zebrafish embryos detected by increased caspase activity and whole-mount TUNEL staining. Fisheries Science 2021; 67, 333-340.
JM Myers, WK Hershberger and RN Iwamoto. The induction of tetraploidy in salmonids. Journal of the World Aquaculture Society 1986; 17(1-4), 1-7.
SB Babaheydari, S Keyvanshokooh, S Dorafshan and SA Johari. Effects of tetraploidy induction on rainbow trout (Oncorhynchus mykiss, Walbaum, 1792) proteome at early stages of development. Comparative Biochemistry and Physiology Part D: Genomics and Proteomics 2016; 20, 57-64.
DDA Aloise, FDA Maia-Lima, RMD Oliveira, TDM Cabral and WF Molina. Ploidy manipulation and polyploid detection in the white shrimp Litopenaeus vannamei (Boone 1931) (Decapoda, Penaeidae). Marine Biotechnology 2011; 13(1), 41-47.
P Hartami, O Carman, MJ Zairin and Alimuddin. Heat shock and its consequences on early life performance of stripped catfis (Pangasianodon hypophthalmus). Omni-Akuatika 2018; 14, 52-58.
TJ Pandian and R Koteeswaran. Ploidy induction and sex control in fish. Hydrobiologia 1998; 384, 167-243.
DC Hardie and PDN Hebert. The nucleotypic effects of cellular DNA content in cartilaginous and ray-finned fishes. Genome 2003; 46(4), 683-706.
M Jackson, L Marks, GHW May and JB Wilson. The genetic basis of disease. Essays in Biochemistry 2018; 62(5), 643-723.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






