Early Protective Effect of Live Influenza Vaccine against Homologous and Heterologous Influenza Infection at Different Times after Immunization
DOI:
https://doi.org/10.48048/tis.2025.9166Keywords:
Influenza, Live influenza vaccine, THP-1, Early cytokines, Type I interferons, Early protection, PandemicAbstract
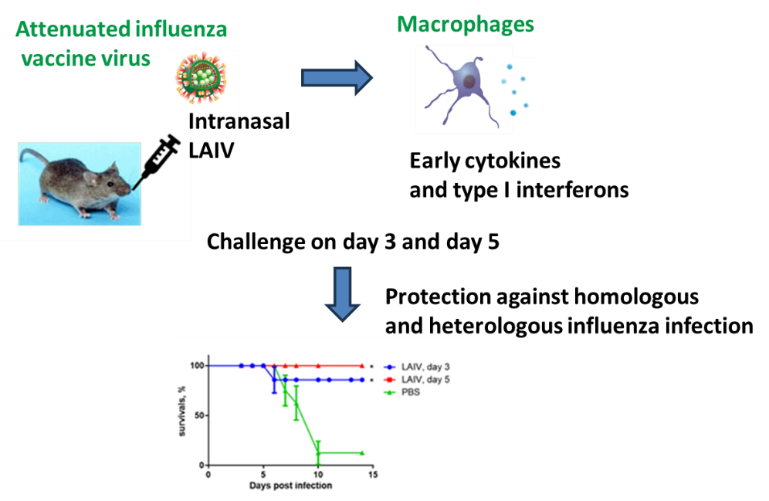
Live influenza vaccine (LAIV) is an effective tool in the fight against influenza infection. LAIV is easy to administer, economical and fast to produce, and stimulates a systemic and local immune response. The aim of the study was to study the early protective effect of LAIV in mice against homologous and heterologous influenza infection within one week of LAIV immunization and possible pathways of immune system activation. We studied expression of early cytokines and type I interferons in response to LAIV introduction in THP-1 cell line. Mice were immunized intranasally with the vaccine virus A/17/South Africa/2013/01(H1N1)pdm09 at a dose of 6 lg EID50. The production of Interleukin-6 (IL-6), Tumor necrosis factor-α (TNF-α) and type I interferons in the respiratory tract of mice was determined by ELISA. Infection was carried out on the 3rd or 5th day after immunization with the influenza viruses A/South Africa/3626/2013(H1N1)pdm09 or A/Indonesia/5/2005(H5N1) IDCDC-RG2. When both the vaccine virus and the parent virus A/South Africa/3626/2013(H1N1)pdm09 were introduced into THP-1 cell culture, an increase in the expression of type I interferon was observed. Immunization with LAIV lead to increase in the production of early cytokines and type I interferons in the respiratory tract of mice. The mice were completely protected on 5 after immunization against lethal homologous and heterologous influenza infection and partially protected on day 3. The data obtained may indicate the benefit of using LAIV during the seasonal increase in acute respiratory viral infections due to stimulation of innate immune factors.
HIGHLIGHTS
- Live influenza vaccine (LAIV) has long been successfully used for the prevention of seasonal influenza.
- The LAIV provides early protection against homologous and heterologous influenza infection in the 1st week after immunization before the formation of an adaptive immune response.
- Introduction of LAIV into a cell culture of macrophage origin and into the nasal passages of mice causes an increase in the expression and production of early cytokines and type I interferon.
- The use of LAIV is useful during the period of circulation of not only influenza viruses, but also other pathogens of respiratory infections
GRAPHICAL ABSTRACT
Downloads
References
M Brisse, SM Vrba, N Kirk, Y Liang and H Ly. Emerging concepts and technologies in vaccine development. Frontiers in Immunology 2020; 11, 583077.
GBD 2019 Mental Disorders Collaborators. Global, regional, and national burden of 12 mental disorders in 204 countries and territories, 1990 - 2019: A systematic analysis for the global burden of disease study 2019. Lancet Psychiatry 2019; 9(2), 137-150.
Q Li, X Guan, P Wu, X Wang, L Zhou, Y Tong, R Ren, KSM Leung, EHY Lau, JY Wong, X Xing, N Xiang, Y Wu, C Li, Q Chen, D Li, T Liu, J Zhao, M Liu, W Tu, …, Z Feng. Early transmission dynamics in Wuhan, China, of novel coronavirus - infected pneumonia. The New England Journal of Medicine 2020; 382(13), 1199-1207.
SG Viveiros-Rosa, CD Mendes, GG Farfán-Cano and M El-Shazly. The race for clinical trials on Omicron-based COVID-19 vaccine candidates: Updates from global databases. Narra J 2022; 2(3), e88.
R Wang, M Liu and J Liu. The association between influenza vaccination and COVID-19 and its outcomes: A systematic review and meta-analysis of observational studies. Vaccines 2021; 9(5), 529.
M Liu, LZX Huang, AA Smits, C Büll, Y Narimatsu, FJMV Kuppeveld, H Clausen, CAMD Haan and ED Vries. Human-type sialic acid receptors contribute to avian influenza A virus binding and entry by hetero-multivalent interactions. Nature Communications volume 2022; 13(1), 4054.
SS Wong and K Yuen. Avian influenza virus infections in humans. Chest 2006; 129(1), 156-168.
Y Desheva. Preparing live influenza vaccines against potential pandemic influenza using nonpathogenic avian influenza viruses and cold-adapted master donor strain. In: SK Saxena (Ed.). Influenza - therapeutics and challenges. IntechOpen, London, 2018, p. 160.
DF Hoft, KR Lottenbach, A Blazevic, A Turan, TP Blevins, TP Pacatte, Y Yu, MC Mitchell, SG Hoft and RB Belshe. Comparisons of the humoral and cellular immune responses induced by live attenuated influenza vaccine and inactivated influenza vaccine in adults. Clinical and Vaccine Immunology 2017; 24(1), e00414-16.
LA Grohskopf, JM Ferdinands, LH Blanton, KR Broder and J Loehr. Prevention and control of seasonal influenza with vaccines: Recommendations of the advisory committee on immunization practices - United States, 2024-25 influenza season. Morbidity and Mortality Weekly Report 2024; 73(5), 1-25.
LG Rudenko, NH Arden, E Grigorieva, A Naychin, A Rekstin, AI Klimov, S Donina, J Desheva, RC Holman, A DeGuzman, NJ Cox and JM Katz. Immunogenicity and efficacy of Russian live attenuated and US inactivated influenza vaccines used alone and in combination in nursing home residents. Vaccine 2000; 19(2-3), 308-318.
EP Grigor’eva, IA Desheva, SA Donina, AN Naĭkhin, AR Rekstin, IB Barantseva, EA Zavitkova, TM Moskvicheva, VG Zhavoronkov and LG Rudenko. The comparative characteristics of the safety, immunogenic activity and prophylactic potency of the adult and children types of live influenza vaccine in schoolchildren aged 7 - 14 years. Voprosy Virusologii 2002; 47(4), 24.
L Rudenko1, AN Slepushkin, AS Monto, AP Kendal, E Grigorieva, E Burtseva, A Rekstin, AL Beljaev, VE Bragina, NJ Cox, YZ Ghendon and GI Alexandrova. Efficacy of live attenuated and inactivated influenza vaccines in schoolchildren and their unvaccinated contacts in Novgorod, Russia. The Journal of Infectious Diseases 1993; 168(4), 881-887.
L Bai, Y Zhao, J Dong, S Liang, M Guo, X Liu, X Wang, Z Huang, X Sun, Z Zhang, L Dong, Q Liu, Y Zheng, D Niu, M Xiang, K Song, J Ye, W Zheng, Z Tang, M Tang, …, K Xu. Coinfection with influenza A virus enhances SARS-Cov-2 infectivity. Cell Research 2021; 31(4), 395-403.
R Rathnasinghe, M Salvatore, H Zheng, S Jangra, T Kehrer, I Mena, M Schotsaert, T Muster, P Palese and A García-Sastre. Interferon mediated prophylactic protection against respiratory viruses conferred by a prototype live attenuated influenza virus vaccine lacking non-structural protein 1. Scientific Reports 2021; 11, 22164.
YJ Lee, JY Lee, YH Jang, S Seo, J Chang and BL Seong. Non-specific effect of vaccines: Immediate protection against respiratory syncytial virus infection by a live attenuated influenza vaccine. Frontiers in Microbiology 2018; 9, 83.
PA Lanthier, GE Huston, A Moquin, SM Eaton, FM Szaba, LW Kummer, MP Tighe, JE Kohlmeier, PJ Blair, M Broderick, ST Smiley and L Haynes. Live attenuated influenza vaccine (LAIV) impacts innate and adaptive immune responses. Vaccine 2011; 29(44), 7849-7856.
AR Rekstin, JA Desheva, IV Kiseleva and IN Isakova-Sivak. Early protection against influenza by pandemic live attenuated influenza vaccines. Medical Academic Journal 2019; 19(3), 37-46.
YA Desheva, GF Leontieva, TA Kramskaya, GO Landgraf, IA Sychev, AR Rekstin and AN Suvorov. Factors of early protective action of live influenza vaccine combined with recombinant bacterial polypeptides against homologous and heterologous influenza infection. Heliyon 2019; 5(2), e01154.
A García-Sastre. Induction and evasion of type I interferon responses by influenza viruses. Virus Research 2011; 162(1-2), 12-18.
P Österlund, V Veckman, J Sirén, KM Klucher, J Hiscott, S Matikainen and I Julkunen. Gene expression and antiviral activity of Alpha/Beta interferons and interleukin-29 in virus-infected human myeloid dendritic cells. Journal of Virology 2005; 79(15), 9608-9617.
GA Kolumam, S Thomas, LJ Thompson, J Sprent and K Murali-Krishna. Type I interferons act directly on CD8 T cells to allow clonal expansion and memory formation in response to viral infection. Journal of Experimental Medicine 2005; 202(5), 637-650.
G Rubino, M Bulati, A Aiello, S Aprile, CM Gambino, F Gervasi, C Caruso and G Accardi. Sicilian centenarian offspring are more resistant to immune ageing. Aging Clinical and Experimental Research 2018; 31(1), 125-133.
X Lu, LE Edwards, JA Desheva, DC Nguyen, A Rekstin, I Stephenson, K Szretter, NJ Cox, LG Rudenko, A Klimov, and JM Katz. Cross-protective immunity in mice induced by live-attenuated or inactivated vaccines against highly pathogenic influenza A (H5N1) viruses. Vaccine 2006; 24(44-46), 6588-6593.
MR Khaitov, V Laza-Stanca, MR Edwards, RP Walton, G Rohde, M Contoli, A Papi, LA Stanciu, SV Kotenko and SL Johnston. Respiratory virus induction of alpha‐, beta‐ and lambda‐interferons in bronchial epithelial cells and peripheral blood mononuclear cells. Allergy 2009; 64(3), 375-386.
I Shannon, CL White and JL Nayak. Understanding immunity in children vaccinated with live attenuated influenza vaccine. J Journal of the Pediatric Infectious Diseases Society 2019; 9(S1), S10-S14.
EY Boravleva, AV Lunitsin, AP Kaplun, NV Bykova, IV Krasilnikov and AS Gambaryan. Immune response and protective efficacy of inactivated and live influenza vaccines against homologous and Heterosubtypic challenge. Biochemistry (Moscow) 2020; 85(5), 553-566.
J Xia, Y Kuang, J Liang, M Jones and SL Swain. Influenza vaccine–induced CD4 effectors require antigen recognition at an effector checkpoint to generate CD4 lung memory and antibody production. The Journal of Immunology 2020; 205(8), 2077-2090.
PJ Turner, AF Abdulla, ME Cole, RR Javan, V Gould, ME O’Driscoll, J Southern, M Zambon, E Miller, NJ Andrews, K Höschler and JS Tregoning. Differences in nasal immunoglobulin a responses to influenza vaccine strains after live attenuated influenza vaccine (LAIV) immunization in children. Clinical and Experimental Immunology 2019; 199(2), 109-118.
T Kishimoto, S Akira, M Narazaki and T Taga. Interleukin-6 family of cytokines and gp130. Blood 1995; 86(4), 1243-1254.
G Herbein and WA O’brien. Tumor necrosis factor (tnf)-α and TNF receptors in Viral Pathogenesis. Proceedings of the Society for Experimental Biology and Medicine 2000; 223(3), 241-257.
SJ Hopkins. The pathophysiological role of cytokines. Legal Medicine 2003; 5(S), S45-S57.
RS Thwaites, ASS Uruchurtu, VA Negri, ME Cole, N Singh, N Poshai, D Jackson, K Hoschler, T Baker, IC Scott, XR Ros, ES Cohen, M Zambon, KM Pollock, TT Hansel and PJM Openshaw. Early mucosal events promote distinct mucosal and systemic antibody responses to live attenuated influenza vaccine. Nature Communications 2023; 14(1), 8053.
T Betakova, A Kostrabova, V Lachova and L Turianova. Cytokines induced during influenza virus infection. Current Pharmaceutical Design 2017; 23(18), 2616-2622.
X Chen, S Liu, MU Goraya, M Maarouf, S Huang and J Chen. Host immune response to influenza A virus infection. Frontiers in Immunology 2018; 9, 320.
K Honda, A Takaoka and T Taniguchi. Type I Interferon gene induction by the interferon regulatory factor family of transcription factors. Immunity 2006; 25(3), 349-360.
MG Netea. Training innate immunity: The changing concept of immunological memory in innate host defence. European Journal of Clinical Investigation 2013; 43(8), 881-884.
C Li, T Wang, Y Zhang and F Wei. Evasion mechanisms of the type I interferons responses by influenza A virus. Critical Reviews in Microbiology 2020; 4(9), 1438-1476.
B Mishra and LB Ivashkiv. Interferons and epigenetic mechanisms in training, priming and tolerance of monocytes and hematopoietic progenitors. Immunological Reviews 2024; 323(1), 257-275.
YH Jang and BL Seong. Immune responses elicited by live attenuated influenza vaccines as correlates of universal protection against influenza viruses. Vaccines 2021; 9(4), 353.
S Sridhar, S Begom, K Hoschler, A Bermingham, W Adamson, W Carman, S Riley and A Lalvani. Longevity and determinants of protective humoral immunity after pandemic influenza infection. American Journal of Respiratory and Critical Care Medicine 2015; 191(3), 325-332.
T Sladkova and F Kostolansky. The role of cytokines in the immune response to influenza A virus infection. Acta Virologica 2006; 50(3), 151-162.
JR Teijaro. Type I interferons in viral control and immune regulation. Current Opinion in Virology 2016; 16, 31-40.
MKJ Kusala, ANM Ansori, RV Nidom, S Indrasari, AF Astutik, I Normalina, MY Alamudi, S Rukmana, K Rachmawati, KP Santoso and CA Nidom. Primary cell culture of zebrafish (Danio rerio) as a material for developing H5N1 avian influenza vaccines. Research Journal of Pharmacy and Technology 2020; 13(12), 6140-6146.
TW Clark, JS Tregoning, H Lister, T Poletti, E Amin and JS Nguyen-Van-Tam. Recent advances in the influenza virus vaccine landscape: A comprehensive overview of technologies and trials. Clinical Microbiology Reviews 2024; 37(4), e00025.
F Krammer. The human antibody response to influenza A virus infection and vaccination. Nature Reviews Immunology 2019; 19, 383-397.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






