Adsorption of Indigo Carmine Dye onto Porous Adsorbent Derived from Banaba Peels Waste
DOI:
https://doi.org/10.48048/tis.2021.9Keywords:
Adsorbent, Adsorption isotherm, Adsorption kinetics, Banana peels, Indigo carmineAbstract
This study investigated the preparation method and adsorption efficiency of porous adsorbents prepared from banana peels (BP) waste. The BP waste was acquired from banana fried chip factories in Sukhothai province. Chemical activation by sulfuric acid was used to activate the raw material, followed by carbonization at 500 °C. The activated BP adsorbent has been characterized by energy dispersive X-ray spectrometer (EDXRF), N2 adsorption-desorption isotherms (BET), scanning electron microscopy (SEM), and zeta potential measurements. The analysis of chemical properties showed that it mainly contained 70.39 % K matter. The specific surface area of the BP sample reached 215.05 m2/g under acid activation and thermal treatment. The SEM images showed the morphologies of the BP adsorbent before and after activation. There was a significant change in the morphology and in the dried BP and activated BP. The pHPZC value of the activated BP obtained under optimal conditions was 4.13. Adsorption of indigo carmine (IC) onto porous activated BP at different initial IC concentrations (10 - 100 mg/L) and contact times (30 - 120 min) was investigated using batch experiments (0.1 g/50 mL). At an optimum contact time of 60 min, maximum adsorption capacity was determined at 56.18 mg/g. The Freundlich isotherm model with a high correlation coefficient was used to describe multilayer adsorption. The kinetic study fit well with a pseudo second-order kinetic model that represented chemisorption. The negative deltaG° value of –14.64 kJ/mol indicated that adsorption of IC onto the BP adsorbent was spontaneous in nature at 305.15 K.
HIGHLIGHTS
- Increase the value of the biomass resources was produced form banana peels as high surface area adsorbent
- The qmaxfor indigo carmine (IC) adsorption was 56.18 mg/g under optimized conditions
- The adsorption of the IC follows the Freundlich model and pseudo-second order kinetic model
- Negative deltaG° value indicated the possibility of IC adsorption process
GRAPHICAL ABSTRACT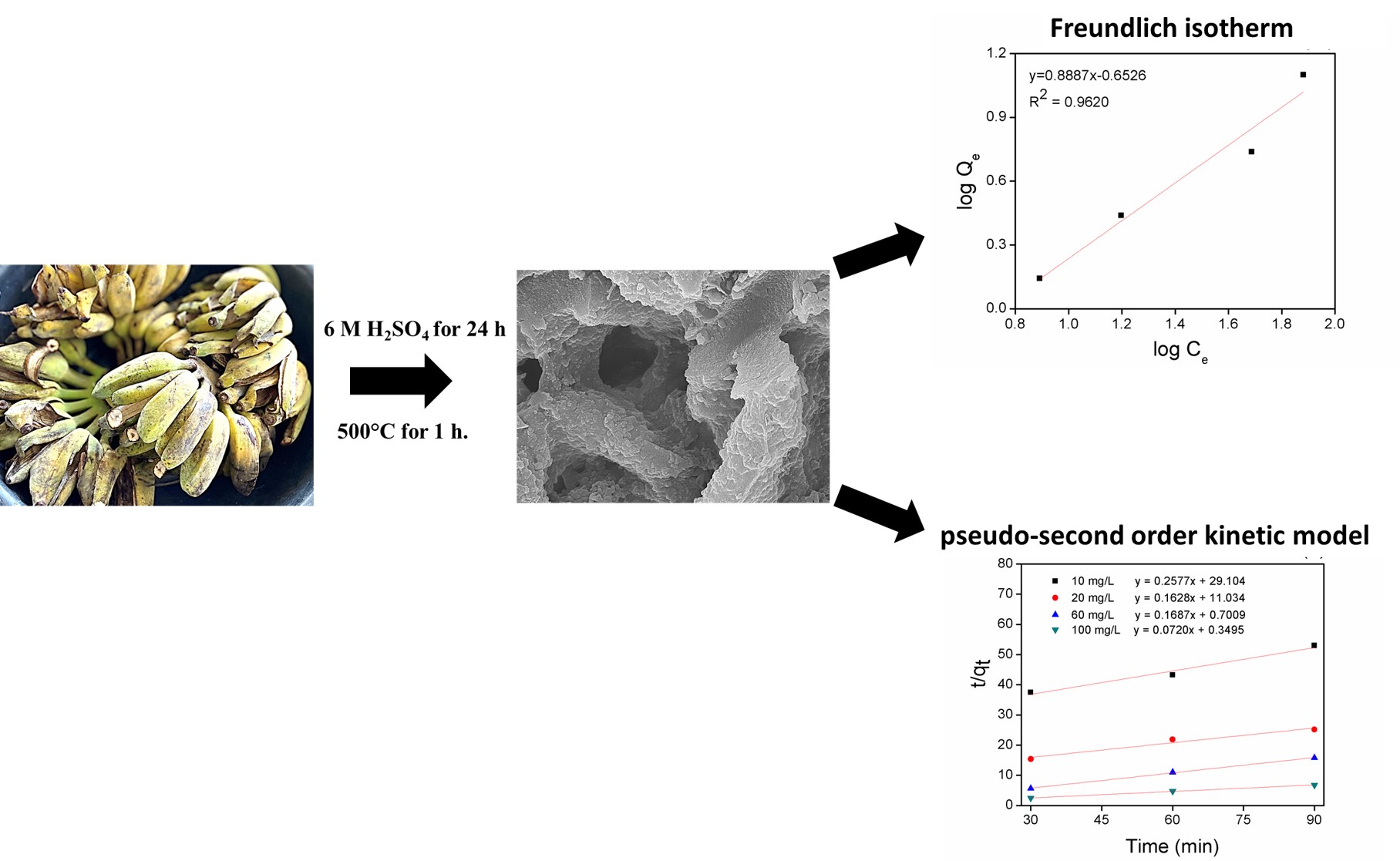
Downloads
References
Z Harrache, M Abbas, T Aksil and M Trari. Thermodynamic and kinetics studies on adsorption of Indigo Carmine from aqueous solution by activated carbon. Microchem J. 2019; 144, 180-9.
HR Rashidi, NM Sulaiman and NA Hashim. Batik industry synthetic waste treatment using nanofiltration membrane. Proc. Eng. 2012; 44, 2010-2.
A Kume. Importance of the green color, absorption gradient, and spectral absorption of chloroplasts for the radiative energy balance of leaves. J. Plant Res. 2017; 130, 501-14.
MI Abdullah, MRSA Janjua, A Mahmood, S Ali and M Ali. Quantum chemical designing of efficient sensitizers for dye sensitized solar cells. Bull. Kor. Chem. Soc. 2013; 34, 2093-8.
RE Palma-Goyes, J Silva-Agredo, I González and RA Torres-Palma. Comparative degradation of indigo carmine by electrochemical oxidation and advanced oxidation processes. Electrochim. Acta 2014; 140, 427-33.
AY Zahrim and N Hilal. Treatment of highly concentrated dye solution by coagulation/flocculation-sand filtration and nanofiltration. Water Resour. Ind. 2013; 3, 23-34.
GZ Kyzas, J Fu and KA Matis. The change from past to future for adsorbent materials in treatment of dyeing wastewaters. Materials 2013; 6, 5131-58.
M Vithanage, SS Mayakaduwa, I Herath, YS Ok and D Mohan. Kinetics, thermodynamics and mechanistic studies of carbofuran removal using biochars from tea waste and rice husks. Chemosphere 2016; 150, 781-9.
GO El-Sayed, MM Yehia and AA Asaad. Assessment of activated carbon prepared from corncob by chemical activation with phosphoric acid. Water Resour. Ind. 2014; 7-8, 66-75.
MC Hoyos-Sánchez, AC Córdoba-Pacheco, LF Rodríguez-Herrera and R Uribe-Kaffur . Removal of Cd (II) from aqueous media by adsorption onto chemically and thermally treated rice husk. J. Chem. 2017; 2017, 5763832.
AB Albadarin, MN Collins, M Naushad, S Shirazian, G Walker and C Mangwandi. Activated lignin-chitosan extruded blends for efficient adsorption of methylene blue. Chem. Eng. J. 2017; 307, 264-72.
HC Tao, HR Zhang, JR Li and WY Ding. Biomass based activated carbon obtained from sludge and sugarcane bagasse for removing lead ion from wastewater. Bioresour. Tech. 2015; 192, 611-7.
ZM Magriotis, PVB Lea, PFD Sales, RM Papini, PRM Vian and PA Arroyo. A comparative study for the removal of mining wastewater by kaolinite, activated carbon and beta zeolite. Appl. Clay Sci. 2014; 91-92, 55-62.
A Omri, A Wali and M Benzina. Adsorption of bentazon on activated carbon prepared from Lawsonia inermis wood: Equilibrium, kinetic and thermodynamic studies. Arab. J. Chem. 2016; 9, S1729-S1739.
GM Couto, AL Dessimoni, ML Bianchi, DM Perígolo and PF Trugilho. Use of sawdust Eucalyptus sp. in the preparation of activated carbons. Ciência e Agrotecnologia 2012; 36, 69-77.
Z Heidarinejad, MH Dehghani, M Heidari, G Javedan, I Ali and M Sillanpää. Methods for preparation and activation of activated carbon: A review. Environ. Chem. Lett. 2020; 18, 393-415.
L Qi, X Tang, Z Wang, and X Peng. Pore characterization of different types of coal from coal and gas outburst disaster sites using low temperature nitrogen adsorption approach. Int. J. Min. Sci. Tech. 2017; 27, 371-7.
P Iacomi and P Llewellyn. pyGAPS: A python-based framework for adsorption isotherm processing and material characterisation. Adsorption 2019; 25, 1533-49.
D Eisenberg, P Prinsen, NJ Geels, W Stroek, N Yana, B Hua JL Luo and G Rothenberg. The evolution of hierarchical porosity in self-templated nitrogen-doped carbons and its effect on oxygen reduction electrocatalysis. RSC Adv. 2016; 6, 80398-407.
D Channei, A Nakaruk, W Khanitchaidecha, P Jannoey and S Phanichphant. Equilibrium, kinetics, and thermodynamic studies concerning the removal of 2-chlorophenol using chemically carbonized rice husk waste. Naresuan Univ. Sci. Tech. 2020; 28, 94-104.
S Phanichphant, A Nakaruk and D Channei. Photocatalytic activity of the binary composite CeO2/SiO2 for degradation of dye. Appl. Surf. Sci. 2016; 387, 214-20.
AM Aljeboree, AN Alshirifi and AF Alkaim. Kinetics and equilibrium study for the adsorption of textile dyes on coconut shell activated carbon. Arab. J. Chem. 2017; 10, S3381-S3393.
DAH Hanaor, M Ghadiri, W Chrzanowski and Y Gan. Scalable surface area characterization by electrokinetic analysis of complex anion Aadsorption. Langmuir 2014; 30, 15143-52.
HD Utomo, XC Ong, SMS Lim, GCB Ong and P Li. Thermally processed sewage sludge for methylene blue uptake. Int. Biodeterior. Biodegrad. 2013; 85, 460-5.
TN Ramesh, DV Kirana, A Ashwini and TR Manasa. Calcium hydroxide as low cost adsorbent for the effective removal of indigo carmine dye in water. J. Saudi Chem. Soc. 2017; 21, 165-71.
A Machrouhi, H Alilou, M Farnane, SE Hamidi, M Sadiq, M Abdennouri, H Tounsadi and N Barka. Statistical optimization of activated carbon from Thapsia transtagana stems and dyes removal efficiency using central composite design. J. Sci. Adv. Mater. Dev. 2019; 4, 544-53.
AN Babu, DS Reddy, P Sharma, GS Kumar, K Ravindhranath and GVK Mohan. Removal of hazardous indigo carmine dye from waste water using treated red mud. Mater. Today. Proc. 2019; 17, 198-208.
Z Harrache, M Abbas, T Aksil and M Trari. Thermodynamic and kinetics studies on adsorption of Indigo Carmine from aqueous solution by activated carbon. Microchem. J. 2019; 144, 180-9.
PS Kumar, S Ramalingam, SD Kirupha, A Murugesan, T Vidhyadevi and S Sivanesan. Adsorption behavior of nickel (II) onto cashew nut shell: Equilibrium, thermodynamics, kinetics, mechanism and process design. Chem. Eng. J. 2011; 167, 122-31.
Z Sun, Z Wu and D Liu. Microwave-assisted modification of activated carbon with cationic surfactants for enhancement of naphthalene adsorption. Kor. J. Chem. Eng. 2018; 35, 557-66.
Downloads
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.







