The Effect of Mixed Lipid Concentrations and Sucrose on the Size of the Lipid Nanoparticles Containing mRNAs
DOI:
https://doi.org/10.48048/tis.2025.8985Keywords:
Lipid nanoparticles (LNPs), Microfluidic, Sucrose, Mixed lipid molar concentration, Size stabilityAbstract
The development of messenger RNA (mRNA) vaccines has been transformed using lipid nanoparticles (LNPs) as delivery systems. This study evaluates the impact of lipid mixture molar concentration and the addition of sucrose as a cryoprotectant on the size and stability of LNPs encapsulating mRNA. Using a microfluidic mixing technique, we formulated LNPs with varying lipid concentrations and analyzed their size, size distribution, and encapsulation efficiency through dynamic light scattering and a RiboGreen assay. Results indicated that higher lipid concentrations not only improved encapsulation efficiency but also maintained LNPs within the ideal size range of 80-120 nm, crucial for optimal biodistribution and immune cell uptake. Furthermore, the addition of sucrose significantly stabilized the LNPs against size changes during freeze-thaw cycles, particularly at lower temperatures. This study underscores the importance of optimizing lipid and sucrose concentrations to enhance the stability and functionality of LNPs, providing insights that could enhance the efficacy and storage stability of mRNA vaccines.
HIGHLIGHTS
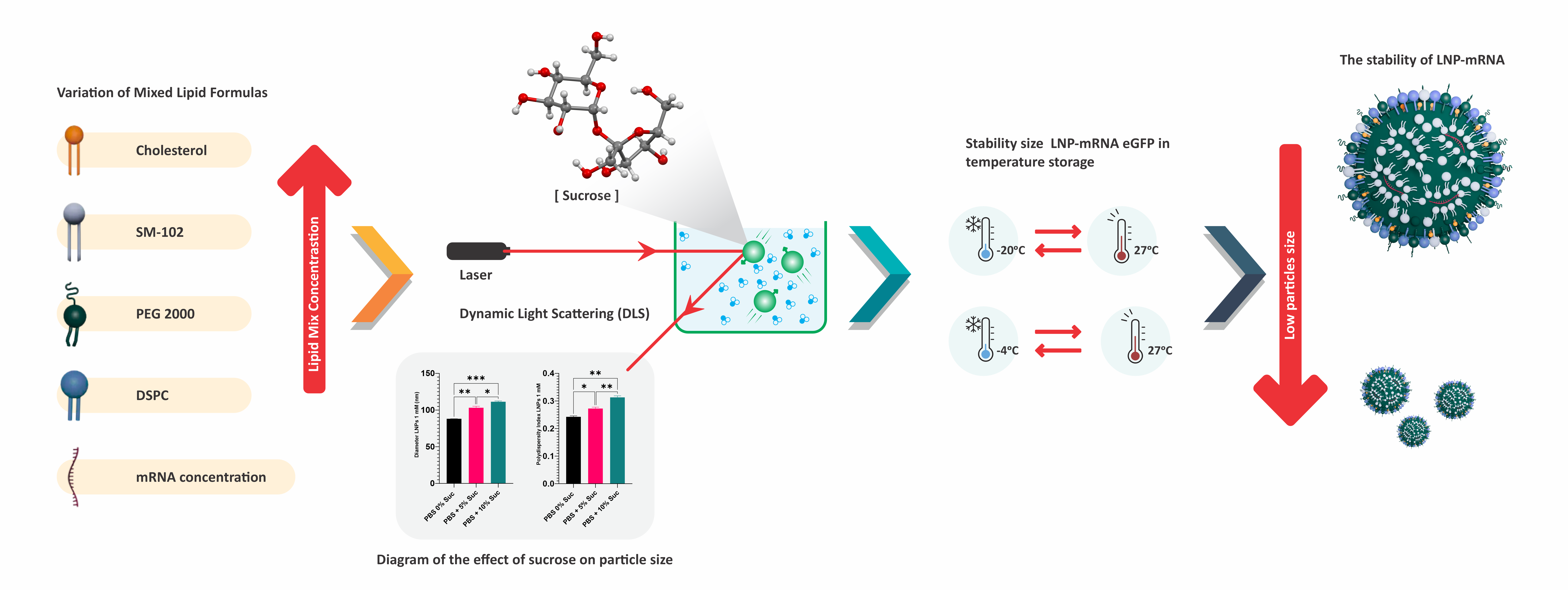
- The variation in molar concentrations of mixed lipids affects the particle size of LNP-mRNA.
- Buffer exchange using a membrane filter with centrifugation technique can reduce the size and uniformity of LNP particle distribution.
- The addition of sucrose affects the particle size of LNP-mRNA.
- The addition of 10 % sucrose as a cryoprotectant can maintain the particle size of LNP-mRNA at 4 and –20 °C.
GRAPHICAL ABSTRACT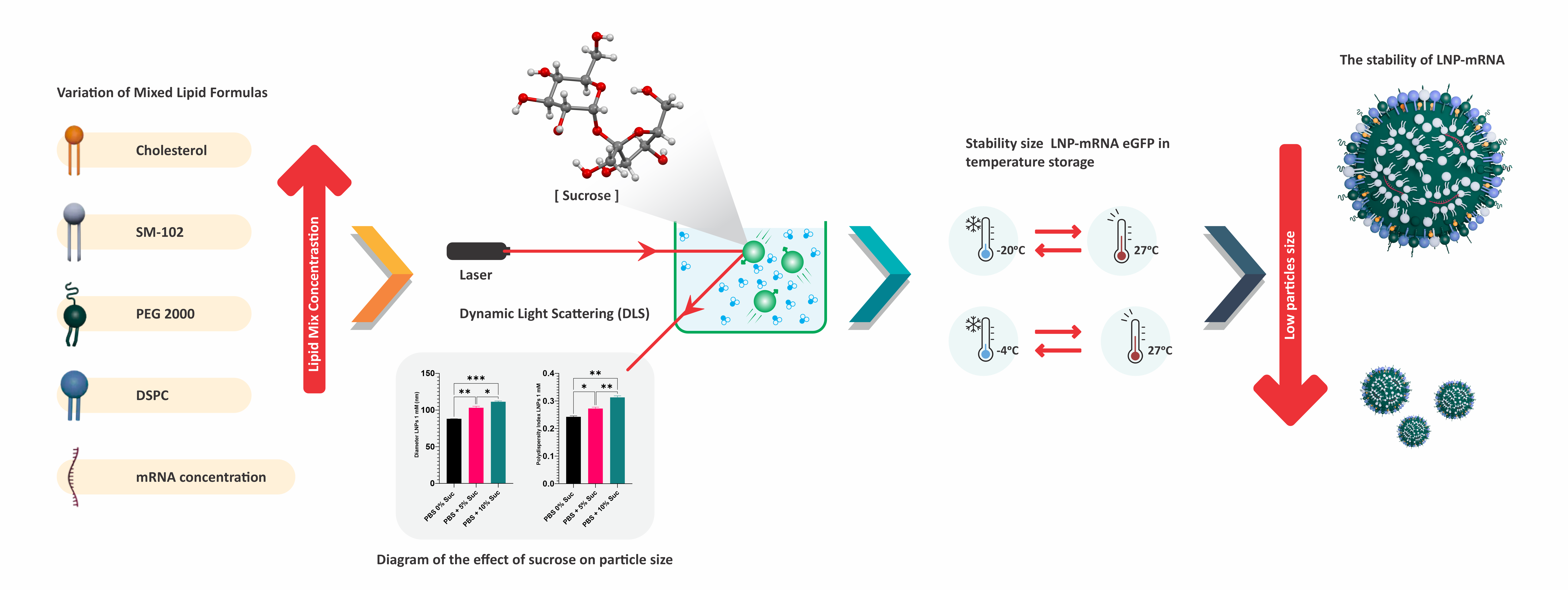
Downloads
References
A Saleh, S Qamar, A Tekin, R Singh and R Kashyap. Vaccine development throughout history. Cureus 2021; 13(7), e16635.
A Fortner and D Schumacher. First COVID-19 vaccines receiving the US FDA and EMA emergency use authorization. Discoveries 2021; 9(1), e122.
GS Laurini, N Montanaro, M Broccoli, G Bonaldo and D Motola. Real-life safety profile of mRNA vaccines for COVID-19: An analysis of VAERS database. Vaccine 2023; 41(18), 2879-2886.
M Schlich, R Palomba, G Costabile, S Mizrahy, M Pannuzzo, D Peer and P Decuzzi. Cytosolic delivery of nucleic acids: The case of ionizable lipid nanoparticles. Bioengineering & Translational Medicine 2021; 6(2), e10213.
MCP Mendonça, A Kont, PS Kowalski and CM O’Driscoll. Design of lipid-based nanoparticles for delivery of therapeutic nucleic acids. Drug Discovery Today 2023; 28(3), 103505.
BZ Igyártó and Z Qin. The mRNA-LNP vaccines - the good, the bad and the ugly? Frontiers in Immunology 2024; 15, 1336906.
M Sedic, JJ Senn, A Lynn, M Laska, M Smith, SJ Platz, J Bolen, S Hoge, A Bulychev, E Jacquinet, V Bartlett and PF Smith. Safety evaluation of lipid nanoparticle-formulated modified mRNA in the Sprague-Dawley rat and cynomolgus monkey. Veterinary Pathology 2018; 55(2), 341-354.
M Yang, Z Zhang, P Jin, K Jiang, Y Xu, F Pan, K Tian, Z Yuan, XE Liu, J Fu, B Wang, H Yan, C Zhan and Z Zhang. Effects of PEG antibodies on in vivo performance of LNP-mRNA vaccines. International Journal of Pharmaceutics 2024; 650, 123695.
R Tenchov, R Bird, AE Curtze and Q Zhou. Lipid nanoparticles - From liposomes to mRNA vaccine delivery, a landscape of research diversity and advancement. ACS Nano 2021; 15(11), 16982-17015.
E Álvarez-Benedicto, L Farbiak, MM Ramírez, X Wang, LT Johnson, O Mian, ED Guerrero and DJ Siegwart. Optimization of phospholipid chemistry for improved lipid nanoparticle (LNP) delivery of messenger RNA (mRNA). Biomaterials Science 2022; 10(2), 549-559.
CH Albertsen, JA Kulkarni, D Witzigmann, M Lind, K Petersson and JB Simonsen. The role of lipid components in lipid nanoparticles for vaccines and gene therapy. Advanced Drug Delivery Reviews 2022; 188, 114416.
HN Jung, SY Lee, S Lee, H Youn and HJ Im. Lipid nanoparticles for delivery of RNA therapeutics: Current status and the role of in vivo imaging. Theranostics 2022; 12(17), 7509-7531.
M Tang, A Sagawa, N Inoue, S Torii, K Tomita and Y Hattori. Efficient mRNA delivery with mRNA lipoplexes prepared using a modified ethanol injection method. Pharmaceutics 2023; 15(4), 1141.
C Zhang, Y Ma, J Zhang, JCT Kuo, Z Zhang, H Xie, J Zhu and T Liu. Modification of lipid-based nanoparticles: An efficient delivery system for nucleic acid-based immunotherapy. Molecules 2022; 27, 1943.
AD Bangham, MM Standish and JC Watkins. Diffusion of univalent ions across the lamellae of swollen phospholipids. Journal of Molecular Biology 1965; 13(1), 238-252.
RE McKenzie, JJ Minnell, M Ganley, GF Painter and SL Draper. mRNA synthesis and encapsulation in ionizable lipid nanoparticles. Current Protocols 2023; 3(9), e898.
CJM Rivas, M Tarhini, W Badri, K Miladi, H Greige-Gerges, QA Nazari, SAG Rodríguez, RÁ Román, H Fessi and A Elaissari. Nanoprecipitation process: From encapsulation to drug delivery. International Journal of Pharmaceutics 2017; 532(1), 66-81.
BG Carvalho, BT Ceccato, M Michelon, SW Han and LGDL Torre. Advanced microfluidic technologies for lipid nano-microsystems from synthesis to biological application. Pharmaceutics 2022; 14(1), 141.
S Salatin, J Barar, M Barzegar-Jalali, K Adibkia, F Kiafar and M Jelvehgari. Development of a nanoprecipitation method for the entrapment of a very water soluble drug into Eudragit RL nanoparticles. Research in Pharmaceutical Sciences 2017; 12(1), 1-14.
M Maeki, T Saito, Y Sato, T Yasui, N Kaji, A Ishida, H Tani, Y Baba, H Harashima and M Tokeshi. A strategy for synthesis of lipid nanoparticles using microfluidic devices with a mixer structure. RSC Advances 2015; 5(57), 46181-46185.
J Nguyen, CL Walsh, JPM Motion, EK Perttu and F Szoka. Controlled nucleation of lipid nanoparticles. Pharmaceutical Research 2012; 29(8), 2236-2248.
KJ Wu, ECM Tse, C Shang and Z Guo. Nucleation and growth in solution synthesis of nanostructures - From fundamentals to advanced applications. Progress in Materials Science 2022; 123, 100821.
M Maeki, S Uno, A Niwa, Y Okada and M Tokeshi. Microfluidic technologies and devices for lipid nanoparticle-based RNA delivery. Journal of Controlled Release 2022; 344, 80-96.
AD Stroock, SKW Dertinger, A Ajdari, I Mezić, HA Stone and GM Whitesides. Chaotic mixer for microchannels. Science 2002; 295(5555), 647-651.
KJ Hassett, J Higgins, A Woods, B Levy, Y Xia, CJ Hsiao, E Acosta, Ö Almarsson, MJ Moore and LA Brito. Impact of lipid nanoparticle size on mRNA vaccine immunogenicity. Journal of Controlled Release 2021; 335, 237-246.
T Nakamura, M Kawai, Y Sato, M Maeki, M Tokeshi and H Harashima. The effect of size and charge of lipid nanoparticles prepared by microfluidic mixing on their lymph node transitivity and distribution. Molecular Pharmaceutics 2020; 17(3), 944-953.
M Dhayalan, W Wang, SUM Riyaz, RA Dinesh, J Shanmugam, SS Irudayaraj, A Stalin, J Giri, S Mallik and R Hu. Advances in functional lipid nanoparticles: From drug delivery platforms to clinical applications. 3 Biotech 2024; 14(2), 57.
MF Bachmann and GT Jennings. Vaccine delivery: A matter of size, geometry, kinetics and molecular patterns. Nature Reviews Immunology 2010; 10, 787-796.
A Ji, M Xu, Y Pan, L Diao, L Ma, L Qian, J Cheng and M Liu. Lipid microparticles show similar efficacy with lipid nanoparticles in delivering mRNA and preventing cancer. Pharmaceutical Research 2023; 40(1), 265-279.
M Kovacsovics-Bankowski, K Clark, B Benacerraf and KL Rock. Efficient major histocompatibility complex class I presentation of exogenous antigen upon phagocytosis by macrophages. Proceedings of the National Academy Sciences of the United States of America 1993; 90(11), 4942-4946.
H Pflicke and M Sixt. Preformed portals facilitate dendritic cell entry into afferent lymphatic vessels. The Journal of Experimental Medicine 2009; 206(13), 2925-2935.
GB Schober, S Story and DP Arya. A careful look at lipid nanoparticle characterization: Analysis of benchmark formulations for encapsulation of RNA cargo size gradient. Scientific Reports 2024; 14(1), 2403.
A Lamoot, J Lammens, ED Lombaerde, Z Zhong, M Gontsarik, Y Chen, TRMD Beer and BGD Geest. Successful batch and continuous lyophilization of mRNA LNP formulations depend on cryoprotectants and ionizable lipids. Biomaterials Science 2023; 11(12), 4327-4334.
P Zhao, X Hou, J Yan, S Du, Y Xue, W Li, G Xiang and Y Dong. Long-term storage of lipid-like nanoparticles for mRNA delivery. Bioactive Materials 2020; 5(2), 358-363.
A Akinc, MA Maier, M Manoharan, K Fitzgerald, M Jayaraman, S Barros, S Ansell, X Du, MJ Hope, TD Madden, BL Mui, SC Semple, YK Tam, M Ciufolini, D Witzigmann, JA Kulkarni, RVD Meel and PR Cullis. The Onpattro story and the clinical translation of nanomedicines containing nucleic acid-based drugs. Nature Nanotechnology 2019; 14(12), 1084-1087.
C Walsh, K Ou, NM Belliveau, TJ Leaver, AW Wild, J Huft, PJ Lin, S Chen, AK Leung, JB Lee, CL Hansen, RJ Taylor, EC Ramsay and PR Cullis. Microfluidic-based manufacture of siRNA-lipid nanoparticles for therapeutic applications. Methods in Molecular Biology 2014; 1141, 109-120.
NM Belliveau, J Huft, PJC Lin, S Chen, AKK Leung, TJ Leaver, AW Wild, JB Lee, RJ Taylor, YK Tam, CL Hansen and PR Cullis. Microfluidic synthesis of highly potent limit-size lipid nanoparticles for in vivo delivery of siRNA. Molecular Therapy Nucleic Acids 2012; 1, e37.
CM Bailey-Hytholt, P Ghosh, J Dugas, IE Zarraga and A Bandekar. Formulating and characterizing lipid nanoparticles for gene delivery using a microfluidic mixing platform. Journal of Visualized Experiments 2021. https://doi.org/10.3791/62226.
R Shi, X Liu, Y Wang, M Pan, S Wang, L Shi and B Ni. Long-term stability and immunogenicity of lipid nanoparticle COVID-19 mRNA vaccine is affected by particle size. Human Vaccines & Immunotherapeutics 2024; 20, 2342592.
M Mehta, TA Bui, X Yang, Y Aksoy, EM Goldys and W Deng. Lipid-based nanoparticles for drug/gene delivery: An overview of the production techniques and difficulties encountered in their industrial development. ACS Materials Au 2023; 3(6), 600-619.
M Maeki, Y Fujishima, Y Sato, T Yasui, N Kaji, A Ishida, H Tani, Y Baba, H Harashima and M Tokeshi. Understanding the formation mechanism of lipid nanoparticles in microfluidic devices with chaotic micromixers. PLoS One 2017; 12(11), e0187962.
SM D’Addio and RK Prud’Homme. Controlling drug nanoparticle formation by rapid precipitation. Advanced Drug Delivery Reviews 2011; 63(6), 417-426.
JG Hashimoto, AS Beadles-Bohling and KM Wiren. Comparison of RiboGreen® and 18S rRNA quantitation for normalizing real-time RT-PCR expression analysis. Biotechniques 2004; 36(1), 54-60.
LJ Jones, ST Yue, CY Cheung and VL Singer. RNA quantitation by fluorescence-based solution assay: RiboGreen reagent characterization. Analytical Biochemistry 1998; 265(2), 368-374.
CB Roces, G Lou, N Jain, S Abraham, A Thomas, GW Halbert and Y Perrie. Manufacturing considerations for the development of lipid nanoparticles using microfluidics. Pharmaceutics 2020; 12(11), 1095.
EVL Grgacic and DA Anderson. Virus-like particles: Passport to immune recognition. Methods 2006; 40(1), 60-65.
R Mihaila, S Chang, AT Wei, ZY Hu, D Ruhela, TR Shadel, S Duenwald, E Payson, JJ Cunningham, N Kuklin and DJ Mathre. Lipid nanoparticle purification by Spin Centrifugation-Dialysis (SCD): A facile and high-throughput approach for small scale preparation of siRNA-lipid complexes. International Journal of Pharmaceutics 2011; 420(1), 118-121.
R Vargas, M Romero, T Berasategui, DA Narváez-Narváez, P Ramirez, A Nardi-Ricart, E García-Montoya, P Pérez-Lozano, JM Suñe-Negre, C Moreno-Castro, C Hernández-Munain, C Suñe and M Suñe-Pou. Dialysis is a key factor modulating interactions between critical process parameters during the microfluidic preparation of lipid nanoparticles. Colloid and Interface Science Communications 2023; 54, 100709.
L Schoenmaker, D Witzigmann, JA Kulkarni, R Verbeke, G Kersten, W Jiskoot and DJA Crommelin. mRNA-lipid nanoparticle COVID-19 vaccines: Structure and stability. International Journal of Pharmaceutics 2021; 601, 120586.
A Hardianto, ZS Muscifa, W Widayat, M Yusuf and T Subroto. The effect of ethanol on lipid nanoparticle stabilization from a molecular dynamics simulation perspective. Molecules 2023; 28(12), 4836.
GF Boafo, KT Magar, MD Ekpo, W Qian, S Tan and C Chen. The role of cryoprotective agents in liposome stabilization and preservation. International Journal of Molecular Sciences 2022; 23(20), 12487.
C Cacela and DK Hincha. Low amounts of sucrose are sufficient to depress the phase transition temperature of dry phosphatidylcholine, but not for lyoprotection of liposomes. Biophysical Journal 2006; 90(8), 2831.
D Guimarães, J Noro, C Silva, A Cavaco-Paulo and E Nogueira. Protective effect of saccharides on freeze-dried liposomes encapsulating drugs. Frontiers in Bioengineering and Biotechnology 2019; 7, 424.
HD Andersen, C Wang, L Arleth, GH Peters and P Westh. Reconciliation of opposing views on membrane-sugar interactions. Proceedings of the National Academy of Sciences of the United States of America 2011; 108(5), 1874-1878.
JH Crowe, LM Crowe, JF Carpenter, AS Rudolph, CA Wistrom, BJ Spargo and TJ Anchordoguy. Interactions of sugars with membranes. Biochimica et Biophysica Acta 1988; 947(2), 367-384.
JH Crowe, FA Hoekstra, KHN Nguyen and LM Crowe. Is vitrification involved in depression of the phase transition temperature in dry phospholipids? Biochimica et Biophysica Acta 1996; 1280(2), 187-196.
IJ Vereyken, V Chupin, RA Demel, SC Smeekens and BD Kruijff. Fructans insert between the headgroups of phospholipids. Biochimica et Biophysica Acta 2001; 1510(1-2), 307-320.
A Dhaliwal, A Khondker, R Alsop and MC Rheinstädter. Glucose can protect membranes against dehydration damage by inducing a glassy membrane state at low hydrations. Membranes 2019; 9(1), 15.
A Roy, R Dutta, N Kundu, D Banik and N Sarkar. A comparative study of the influence of sugars sucrose, trehalose, and maltose on the hydration and diffusion of DMPC lipid bilayer at complete hydration: Investigation of structural and spectroscopic aspect of lipid-sugar interaction. Langmuir 2016; 32(20), 5124-5134.
GVD Bogaart, N Hermans, V Krasnikov, AHD Vries and B Poolman. On the decrease in lateral mobility of phospholipids by sugars. Biophysical Journal 2007; 92(5), 1598-1605.
Ü Kartoglu, NK Özgüler, LJ Wolfson and W Kurzatkowski. Validation of the shake test for detecting freeze damage to adsorbed vaccines. Bulletin of the World Health Organization 2010; 88(8), 624-631.
B Kim, RR Hosn, T Remba, D Yun, N Li, W Abraham, MB Melo, M Cortes, B Li, Y Zhang, Y Dong and DJ Irvine. Optimization of storage conditions for lipid nanoparticle-formulated self-replicating RNA vaccines. Journal of Controlled Release 2023; 353, 241-253.
SC Rumsey, NF Galeano, Y Arad and RJ Deckelbaum. Cryopreservation with sucrose maintains normal physical and biological properties of human plasma low density lipoproteins. Journal of Lipid Research 1992; 33(10), 1551-1561.
G Yu, R Li and A Hubel. Interfacial interactions of sucrose during cryopreservation detected by Raman spectroscopy. Langmuir 2019; 35(23), 7388-7395.
MJGW Roozen and MA Hemminga. Molecular motion in sucrose-water mixtures in the liquid and glassy state as studied by spin probe ESR. Journal of Physical Chemistry 1990; 94(18), 7326-7329.
WQ Sun, AC Leopold, LM Crowe and JH Crowe. Stability of dry liposomes in sugar glasses. Biophysical Journal 1996; 70(4), 1769-1776.
G Anderluzzi, G Lou, S Woods, ST Schmidt, S Gallorini, M Brazzoli, R Johnson, CW Roberts, DT O’Hagan, BC Baudner and Y Perrie. The role of nanoparticle format and route of administration on self-amplifying mRNA vaccine potency. Journal of Controlled Release 2022; 342, 388-399.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






