Molecular Identification of Some Selected Cyanobacteria and Their Antioxidant Activities
DOI:
https://doi.org/10.48048/tis.2025.8950Keywords:
Cyanobacteria, Antioxidant activities, 16S rRNA sequences, Molecular identification, ABTS radical scavenging assay, ORAC-fluorescein assay, Metal chelating assay, Taxonomic relationshipsAbstract
Cyanobacteria, photosynthetic prokaryotes, are promising sources of biologically active compounds with antioxidant properties, attracting interest in their potential applications in the food, pharmaceutical, and cosmetic sectors. This present study aimed to use 16S rRNA sequences for identification of some selected strains of cyanobacteria which were previously morphologically identified. The investigation of their antioxidant activities was also performed by ABTS radical scavenging assay, ORAC-fluorescein assay and metal chelating assay in this study. Molecular identification of 8 isolates revealed 1 Calothrix sp., 2 Nostoc sp., 2 Amazonocrinis sp., 2 Aulosila sp., and 1 from the Hapalosiphonaceae family. Aulosila sp. exhibited the highest ABTS inhibition (IC50: 18.87 µg/µL), followed by a Hapalosiphonaceae cyanobacterium (19.11 µg/µL) and Calothrix sp. (21.74 µg/µL). The ORAC-fluorescein assay showed Nostoc sp. TUBT05 and a Hapalosiphonaceae cyanobacterium had the highest peroxyl radical scavenging with TE values of 0.22 and 0.21 µg/µL, respectively. Nostoc sp. TUBT01 demonstrated the highest metal chelating activity (IC50: 36.48 µg/µL). The combined analysis of selected cyanobacteria revealed insights into the relationship between their taxonomy and antioxidant profiles, highlighting their potential as natural antioxidant sources.
HIGHLIGHTS
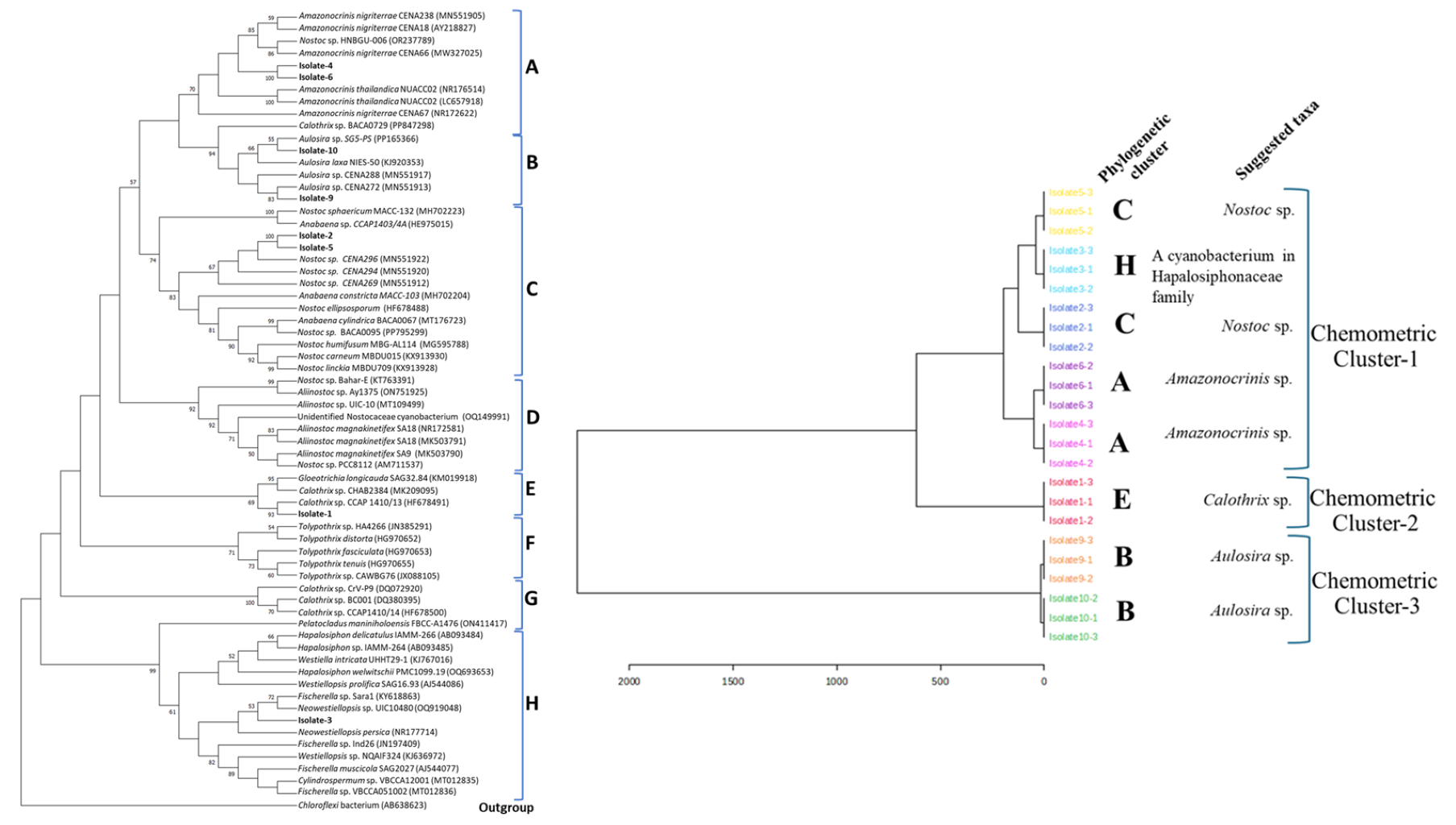
- 16S rRNA sequencing, BLAST, and phylogenetic analysis successfully reclassified previously morphologically identified cyanobacterial isolates.
- Eight cyanobacterial strains were found to have antioxidant activity, indicating their potential as a source of antioxidants.
- Antioxidant profiling of the selected cyanobacteria classified them into 3 major groups, revealing interesting correlations between taxonomic classification and antioxidant potential.
GRAPHICAL ABSTRACT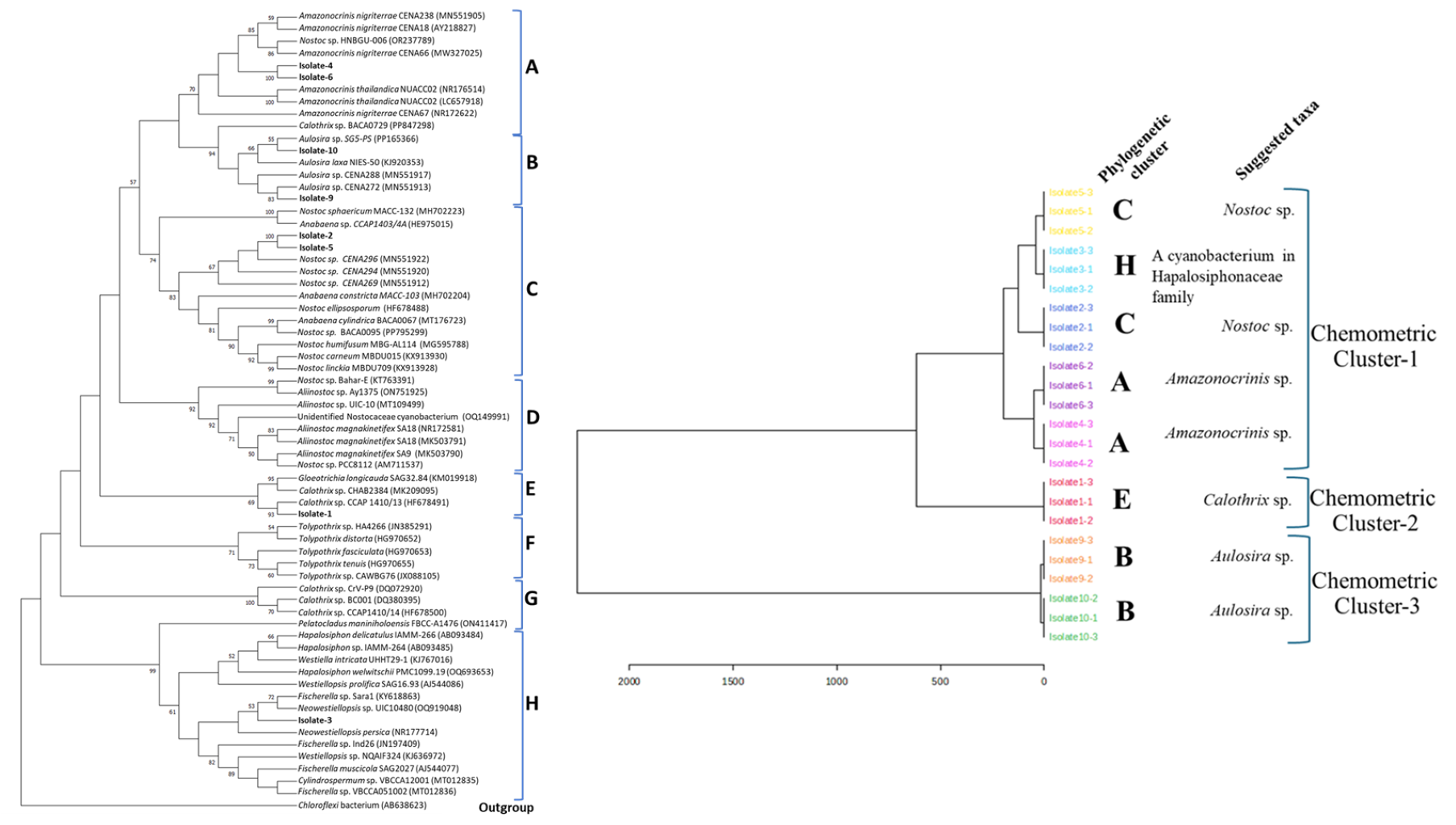
Downloads
References
A Guerreiro, MA Andrade, C Menezes, F Vilarinho and E Dias. Antioxidant and cytoprotective properties of cyanobacteria: Potential for biotechnological applications. Toxins 2020; 12(9), 548.
DP Singh, R Prabha, S Verma, KK Meena and M Yandigeri. Antioxidant properties and polyphenolic content in terrestrial cyanobacteria. 3 Biotech 2017; 7(2), 134.
K Jomova, R Raptova, SY Alomar, SH Alwasel, E Nepovimova, K Kuca and M Valko. Reactive oxygen species, toxicity, oxidative stress, and antioxidants: Chronic diseases and aging. Archives of Toxicology 2023; 97(10), 2499-2574.
Y Kumar, A Tarafdar, D Kumar, C Saravanan, PC Badgujar, A Pharande, S Pareek and OA Fawole. Polyphenols of edible macroalgae: Estimation of in vitro bio-accessibility and cytotoxicity, quantification by LC-MS/MS and potential utilization as an antimicrobial and functional food ingredient. Antioxidants 2022; 11(5), 993.
S Lomakool, K Ruangrit, I Jeerapan, Y Tragoolpua, C Pumas, S Srinuanpan, J Pekkoh and K Duangjan. Biological activities and phytochemicals profiling of different cyanobacterial and microalgal biomass. Biomass Conversion and Biorefinery 2021; 13, 4195-4211.
R Liu and SA Mabury. Synthetic phenolic antioxidants: A review of environmental occurrence, fate, human exposure, and toxicity. Environmental Science & Technology 2020; 54(19), 11706-11719.
HTL Pham, LTT Nguyen, TA Duong, DTT Bui, QT Doan, HTT Nguyen and S Mundt. Diversity and bioactivities of nostocacean cyanobacteria isolated from paddy soil in Vietnam. Systematic and Applied Microbiology 2017; 40(8), 470-481.
C Akoijam, AK Singh and AN Rai. Characterization of free-living cyanobacterial strains and their competence to colonize rice roots. Biology and Fertility of Soils 2012; 48(6), 679-687.
N Wada, T Sakamoto and S Matsugo. Mycosporine-like amino acids and their derivatives as natural antioxidants. Antioxidants 2015; 4(3), 603-646.
IY Massey, MA Osman and F Yang. An overview on cyanobacterial blooms and toxins production: Their occurrence and influencing factors. Toxin Reviews 2022; 41(1), 326-346.
B Ramakrishnan, NR Maddela, K Venkateswarlu and M Megharaj. Potential of microalgae and cyanobacteria to improve soil health and agricultural productivity: A critical view. Environmental Science: Advances 2023; 2(4), 586-611.
D Chittora, M Meena, T Barupal, P Swapnil and K Sharma. Cyanobacteria as a source of biofertilizers for sustainable agriculture. Biochemistry and Biophysics Reports 2020; 22, 100737.
MF Hossain, RR Ratnayake, K Meerajini and KLW Kumara. Antioxidant properties in some selected cyanobacteria isolated from freshwater bodies of Sri Lanka. Food Science & Nutrition 2016; 4(5), 753-758.
U Nübel, F Garcia-Pichel and G Muyzer. PCR primers to amplify 16S rRNA genes from cyanobacteria. Applied and Environmental Microbiology 1997; 63(8), 3327-3332.
JM Janda and SL Abbott. 16S rRNA gene sequencing for bacterial identification in the diagnostic laboratory: Pluses, perils, and pitfalls. Journal of Clinical Microbiology 2007; 45(9), 2761-2764.
S Chittapun and T Charoenrat. Isolation and growth of N2-fixing cyanobacteria from organic agricultural areas in Sanamchaikate, Chachoeng-Sao Province, Thailand. Thammasat International Journal of Science and Technology 2015; 20(2), 27-32.
J Doyle. DNA protocols for plants. In: Molecular techniques in taxonomy. Springer, Germany, 1991, p. 283-293.
K Tamura, G Stecher and S Kumar. MEGA11: Molecular evolutionary genetics analysis version 11. Molecular Biology and Evolution 2021; 38(7), 3022-3027.
R Re, N Pellegrini, A Proteggente, A Pannala, M Yang and C Rice-Evans. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radical Biology and Medicine 1999; 26(9-10), 1231-1237.
A Dávalos, C Gómez-Cordovés and B Bartolomé. Extending applicability of the oxygen radical absorbance capacity (ORAC- fluorescein) assay. Journal of Agricultural and Food Chemistry 2004; 52(1), 48-54.
FC Wong, AL Yong, EP Ting, SC Khoo, HC Ong and TT Chai. Antioxidant, metal chelating, anti-glucosidase activities and phytochemical analysis of selected tropical medicinal plants. Iranian Journal of Pharmaceutical Research 2014; 13(4), 1409-1415.
Z Pang, Y Lu, G Zhou, F Hui, L Xu, C Viau, AF Spigelman, PE MacDonald, DS Wishart, S Li and J Xia. MetaboAnalyst 6.0: Towards a unified platform for metabolomics data processing, analysis and interpretation. Nucleic Acids Research 2024; 52(W1), W398-W406.
J Komárek. A polyphasic approach for the taxonomy of cyanobacteria: Principles and applications. European Journal of Phycology 2016; 51(3), 346-353.
DO Alvarenga, APD Andreote, LHZ Branco, E Delbaje, RB Cruz, ADM Varani and MF Fiore. Amazonocrinis nigriterrae gen. nov., sp. nov., Atlanticothrix silvestris gen. nov., sp. nov. and Dendronalium phyllosphericum gen. nov., sp. nov., nostocacean cyanobacteria from Brazilian environments. International Journal of Systematic and Evolutionary Microbiology 2021; 71(5), 4811.
TT Ngo, BLT Nguyen, TA Duong, THT Nguyen, TL Nguyen, KT Kieu, MHT Do, SV Nguyen, ND Thang and HTL Pham. Polyphasic evaluation and cytotoxic investigation of isolated cyanobacteria with an emphasis on potent activities of a Scytonema strain. Frontiers in Microbiology 2022; 13, 1025755.
BF Tan, SH Te, CY Boo, KYH Gin and JR Thompson. Insights from the draft genome of the subsection V (Stigonematales) cyanobacterium Hapalosiphon sp. Strain MRB220 associated with 2-MIB production. Standards in Genomic Sciences 2016; 11(1), 58.
B Nowruzi, L Hutarova, D Vešelenyiova and JS Metcalf. Characterization of Neowestiellopsis persica A1387 (Hapalosiphonaceae) based on the cpcA, psbA, rpoC1, nifH and nifD gene sequences. BMC Ecology and Evolution 2024; 24(1), 57.
P Rajaniemi, P Hrouzek, K Kaštovska, R Willame, A Rantala, L Hoffmann, J Komárek and K Sivonen. Phylogenetic and morphological evaluation of the genera Anabaena, Aphanizomenon, Trichormus and Nostoc (Nostocales, Cyanobacteria). International Journal of Systematic and Evolutionary Microbiology 2005; 55(Pt 1), 11-26.
W Tawong, P Pongcharoen, P Pongpadung, S Ponza and W Saijuntha. Amazonocrinis thailandica sp. nov. (Nostocales, Cyanobacteria), a novel species of the previously monotypic Amazonocrinis genus from Thailand. Algae 2022; 37(1), 1-14.
F Cai, G Yu, Y Liu, Y Sun and R Li. Description of two new species of Nostoc from China based on the polyphasic approach. Fottea 2021; 21(2), 259-271.
ISS Pinto, IFF Neto and HMVM Soares. Biodegradable chelating agents for industrial, domestic, and agricultural applications—a review. Environmental Science and Pollution Research 2014; 21, 11893-11906.
M AAlikhani, M Khalili and M Jahanshahi. The natural iron chelators’ ferulic acid and caffeic acid rescue mice’s brains from side effects of iron overload. Frontiers in Neurology 2022; 13, 951725.
MMS Ismaiel, YM El-Ayouty and MD Piercey-Normore. Antioxidants characterization in selected cyanobacteria. Annals of Microbiology 2014; 64, 1223-1230.
WSM Aly and SC Andrews. Iron regulation of growth and heterocyst formation in the nitrogen fixing cyanobacterium Nostoc sp. PCC 7120. Journal of Ecology of Health & Environment 2016; 4(3), 103-109.
R Rai, S Singh, KK Rai, A Raj, S Sriwastaw and LC Rai. Regulation of antioxidant defense and glyoxalase systems in cyanobacteria. Plant Physiology and Biochemistry 2021; 168, 353-372.
Y Kumar, A Tarafdar, D Kumar, C Saravanan, PC Badgujar, A Pharande, S Pareek and OA Fawole. Polyphenols of edible macroalgae: Estimation of in vitro bio-accessibility and cytotoxicity, quantification by LC-MS/MS and potential utilization as an antimicrobial and functional food ingredient. Antioxidants 2022; 11(5), 993.
C Kranzler, H Lis, Y Shaked and N Keren. The role of reduction in iron uptake processes in a unicellular, planktonic cyanobacterium. Environmental Microbiology 2011; 13(11), 2990-2999.
A Al-Amin, F Parvin, J Chakraborty and YI Kim. Cyanobacteria mediated heavy metal removal: A review on mechanism, biosynthesis, and removal capability. Environmental Technology Reviews 2021; 10(1), 44-57.
JS Boden, KO Konhauser, LJ Robbins and P Sánchez-Baracaldo. Timing the evolution of antioxidant enzymes in cyanobacteria. Nature Communications 2021; 12, 4742.
M Bernroitner, M Zamocky, PG Furtmüller, GA Peschek and C Obinger. Occurrence, phylogeny, structure, and function of catalases and peroxidases in cyanobacteria. Journal of Experimental Botany 2009; 60(2), 423-440.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






