Optimization of Acetylcholinesterase and Sucrose Concentration using Response Surface Methodology (RSM) Approach in the Development of Paper-Based Biosensor for Pesticides Detection
DOI:
https://doi.org/10.48048/tis.2025.8849Keywords:
Biosensor, Paper-based, Pesticide, Optimization, RSM, Organophosphate, ColorimetryAbstract
Excessive use of pesticides, particularly organophosphate pesticides (OPs), to increase agricultural productivity may give a risk to consumers if consumed as these pesticides are neurotoxic substances. To increase agricultural productivity, fruits and vegetables are frequently treated with excessive pesticides, particularly organophosphate pesticides (OPs). Several methods have been studied and developed to identify pesticide residue in crops, however the standard laboratory procedures for OPs analysis involves the use of HPLC or GC-MS. These methods are complex in sample preparation and require expert labor to operate. Paper-based biosensors are low-cost platforms for fast detection in the field, offering an easy and reusable option. In this study, a colorimetric paper-based biosensor based on immobilizing acetylcholinesterase (AChE) using sol-gel silica matrices and indoxyl acetate is developed. The Response Surface Methodological (RSM) approach was used to optimize AChE concentration and sucrose as a stabilizer, allowing simultaneous optimization of multiple variables using smaller-size datasets. This study used the central composite design (CCD) fractional factorial design using Design Expert 7.0 software. A quadratic model was selected to represent both immobilization yield and relative AChE activity responses. The model was evaluated using ANOVA values and Lack of Fit test which confirmed that the quadratic models for both responses are suitable for experimental data. Validation of the model demonstrates that the predictive model accurately represents of the validation research. The Limit of Detection (LoD) of the biosensor for pure profenofos pesticide after a 20-minute incubation time is 1 ppm, as indicated by a 32.55 % decrease in color intensity. After a week of storage at 4 °C, the biosensor showed a loss of 5.92 % on immobilization yield and 2.55 % on relative activity indicating a good storage stability. The mean value of lettuce samples was 200.60 ± 1.2 a.u, with pesticide concentration of 2.57 ± 1.2 ppm (n=3).
HIGHLIGHTS
- Incorporating AChE into the sol gel matrices is a great way to enhance the stability of the enzymatic colorimetric pesticide detection.
- Response Surface Methodology (RSM) is considered as useful for optimizing response level of AChE and sucrose.
- Combination of optimum AChE and sucrose concentration show beneficial effect on the stability of the biosensor.
GRAPHICAL ABSTRACT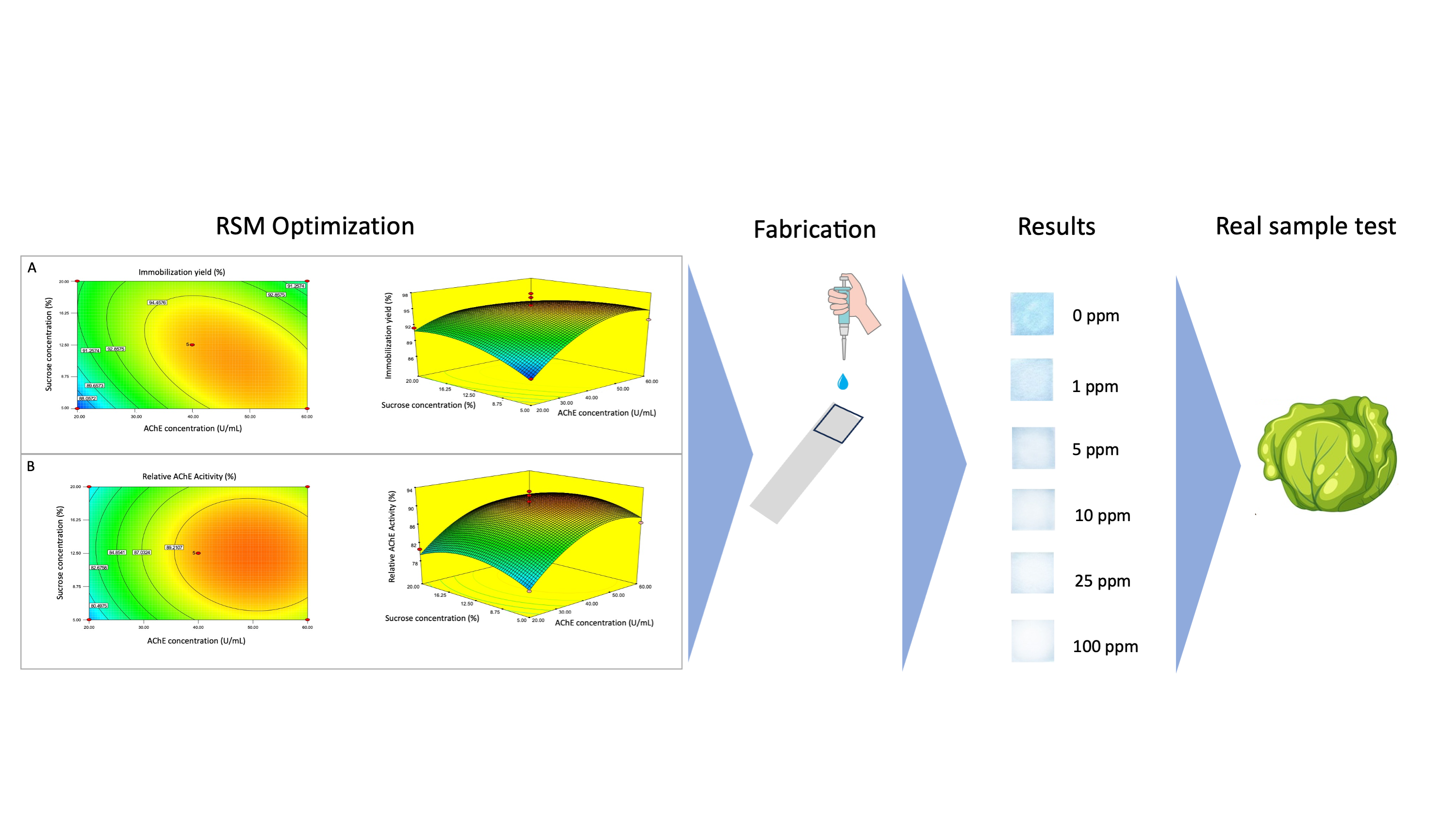
Downloads
References
MG Lionetto, R Caricato, A Calisi, ME Giordano and T Schettino. Acetylcholinesterase as a biomarker in environmental and occupational medicine: New insights and future perspectives. BioMed Research International 2013; 2013, 321213.
D Harshit, K Charmy and P Nrupesh. Organophosphorus pesticides determination by novel HPLC and spectrophotometric method. Food Chemistry 2017; 230, 448-453.
SD Wijayanti, L Tsvik and D Haltrich. Recent advances in electrochemical enzyme-based biosensors for food and beverage analysis. Foods 2023; 12(18), 3355.
B Jiang, M Lu and M Xu. Amperometric sensing of organophosphorus pesticides based on covalently attached multilayer assemblies of diazo-resin, prussian blue single-walled carbon nanotubes, and acetylcholinesterase. Revue Roumaine de Chimie 2019; 64(9), 763-774.
HF Cui, TT Zhang, QY Lv, X Song, XJ Zhai and GG Wang. An acetylcholinesterase biosensor based on doping Au nanorod@SiO2 nanoparticles into TiO2-chitosan hydrogel for detection of organophosphate pesticides. Biosensors and Bioelectronics 2019; 141, 111452.
H Hu, B Wang, Y Li, P Wang and L Yang. Acetylcholinesterase sensor with patterned structure for detecting organophosphorus pesticides based on titanium dioxide sol‐gel carrier. Electroanalysis 2020; 32(8), 1834-1842.
Z Sun, L Tian, M Guo, X Xu, Q Li and H Weng. A double-film screening card for rapid detection of organophosphate and carbamate pesticide residues by one step in vegetables and fruits. Food Control 2017; 81, 23-29.
NA Mohidem, M Mohamad, MU Rashid, MN Norizan, F Hamzah and HB Mat. Recent advances in enzyme immobilisation strategies: An overview of techniques and composite carriers. Journal of Composites Science 2023; 7(12), 488.
DL Jürgen-Lohmann and RL Legge. Immobilization of bovine catalase in sol-gels. Enzyme and Microbial Technology 2006; 39(4), 626-633.
GJ Owens, RK Singh, F Foroutan, M Alqaysi, H Cheol-Mim, Chinmaya Mahapatra, K Hae-Won and JC Knowles. Sol-gel based materials for biomedical applications. Progress in Materials Science 2016; 77, 1-79.
MC Gonçalves. Sol-gel silica nanoparticles in medicine: A natural choice. Design, synthesis and products. Molecules 2018; 23(8), 2021.
R Todorova and K Nedev. Effect of high concentrations of sucrose on the enzymatic activity of a‐chymotrypsin. IUBMB Life 2000; 49(6), 491-496.
P Srirangsan, K Kawai, N Hamada-Sato, M Watanabe and T Suzuki. Stabilizing effects of sucrose-polymer formulations on the activities of freeze-dried enzyme mixtures of alkaline phosphatase, nucleoside phosphorylase and xanthine oxidase. Food Chemistry 2011; 49(1), 271-280.
RA Sheldon and SV Pelt. Enzyme immobilisation in biocatalysis: Why, what and how. Chemical Society Reviews 2013; 42(15), 6223-6235.
M Pohanka, M Hrabinova, K Kuca and J P Simonato. Assessment of acetylcholinesterase activity using indoxylacetate and comparison with the standard Ellman’s method. International Journal of Molecular Sciences 2011; 12(4), 2631-2640.
M Kumari and SK Gupta. Response surface methodological (RSM) approach for optimizing the removal of trihalomethanes (THMs) and its precursor’s by surfactant modified magnetic nanoadsorbents (sMNP) - An endeavor to diminish probable cancer risk. Scientific Reports 2019; 9, 18339.
SK Behera, H Meena, S Chakraborty and BC Meikap. Application of response surface methodology (RSM) for optimization of leaching parameters for ash reduction from low-grade coal. International Journal of Mining Science and Technology 2018; 28(4), 621-629.
K Mahalik, JN Sahu, AV Patwardhan and BC Meikap. Statistical modelling and optimization of hydrolysis of urea to generate ammonia for flue gas conditioning. Journal of Hazardous Materials 2010; 182(1-3), 603-610.
TJ Bergman and JC Beehner. A simple method for measuring colour in wild animals: Validation and use on chest patch colour in geladas (Theropithecus gelada). Biological Journal of the Linnean Society 2008; 94(2), 231-240.
ML Gulrajani. Colour measurement principles, advances and industrial applications. Woodhead Publishing, Cambridgeshire, 2010.
S Arana-Peña, NS Rios, D Carballares, C Mendez-Sanchez, Y Lokha, LRB Gonçalves and R Fernandez-Lafuente. Effects of enzyme loading and immobilization conditions on the catalytic features of lipase from Pseudomonas fluorescens immobilized on octyl-agarose beads. Frontiers in Bioengineering and Biotechnology 2020; 8, 36.
LJ Li, WJ Xia, GP Ma, YL Chen and YY Ma. A study on the enzymatic properties and reuse of cellulase immobilized with carbon nanotubes and sodium alginate. AMB Express 2019; 9, 112.
L Momeni, S Mahmodian, B Shareghi, AA Saboury and S Farhadian. The functional and structural stabilization of trypsin by sucrose. International Journal of Biological Macromolecules 2017; 99, 343-349.
V Minovska, E Winkelhausen and S Kuzmanova. Lipase immobilized by different techniques on various support materials applied in oil hydrolysis. Journal of the Serbian Chemical Society 2005; 70(4), 609-624.
I Alemzadeh, SSS Kavardi and A Kazemi. Optimization of lipase immobilization. International Journal of Engineering 2012; 25(1), 1-10.
SN Timasheff. Control of protein stability and reactions by weakly interacting cosolvents: The simplicity of the complicated. Advances in Protein Chemistry 1998; 51, 355-432.
G Derringer and R Suich. Simultaneous optimization of several response variables. Journal of Quality Technology 1980; 12(4), 214-219.
J Li, C Ma, Y Ma, Y Li, W Zhou and P Xu. Medium optimization by combination of response surface methodology and desirability function: An application in glutamine production. Applied Microbiology and Biotechnology 2007; 74, 563-571.
IJ Jeong and KJ Kim. An interactive desirability function method to multiresponse optimization. European Journal of Operational Research 2009; 195(2), 412-426.
M Kavruk, VC Özalp and HA Öktem. Portable bioactive paper-based sensor for quantification of pesticides. Journal of Analytical Methods in Chemistry 2013; 2013, 932946.
X Guo, X Zhang, Q Cai, T Shen and S Zhu. Developing a novel sensitive visual screening card for rapid detection of pesticide residues in food. Food Control 2013; 30(1), 15-23.

Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






