Understanding the Complex Interplay of Epigenetic Factors in Atherosclerosis: A Review
DOI:
https://doi.org/10.48048/tis.2024.8576Keywords:
Atherosclerosis, Dyslipoproteinaemia, Hyperhomocysteinaemia, Dyslipidemia, Hypermethylation, EpigenticsAbstract
This review focuses on the epigenetic factors that play a vital role in the formation of plaques and lead to atherosclerosis and related diseases in humans during current times. Apart from the epigenetics there are plenty more factors that influence the atherosclerotic risk in the older population of the current era. Lack of physical activities has become one of the most commonly found traits that causes obesity in people especially the middles aged adults. People working 9 - 5 in an office, or an IT firm tend spend idle time with their computer for 4 - 5 consecutive hours with barely changing their position. Lack of exercise tends to affect not only the physical health but also stress a person mentally. Chronic inflammatory diseases like atherosclerosis have a major role in the morbidity and mortality from cardiovascular disease worldwide. Atherosclerotic plaques are created by the pathophysiological process of lipids, inflammatory cells and fibrous components gradually accumulating within artery walls. Endothelial dysfunction signals the start of atherosclerosis and allows low-density lipoproteins (LDL) to enter the subendothelial region. Oxidative stress-induced changes in retained low-density lipoprotein (LDL) set off an inflammatory cascade that draws T cells and monocytes to the site of the lesion. The interaction of these immune cells with the local vascular cells encourages the production of foam cells, which fills the plaque’s centre with lipids. Smooth muscle cells multiply and help produce fibrous caps as the lesion ages, which affects the stability of the plaque. Studies shows how the brain responds to physical labour and how differently the effectively the enzymes and hormones are secreted, and their actions are vastly guided based on the physical stress an individual undergoes. Among the most prominent causes of atherosclerosis epigenetics has been focussed majorly in this review, where we focus on the underlying genetic factors that play a vital role in atherosclerosis and related cardiovascular diseases and also the genetic approach to a treatment is also elaborated. A number of miRNAs are involved in the cause/cure for the atherosclerosis, the following tables give an outlook regarding the same. This review aims to focus on the causes, risk factors, pathophysiology and the treatment and therapies that are currently available all the way concentrating on the genetic approach in the above-mentioned perspective.
HIGHLIGHTS
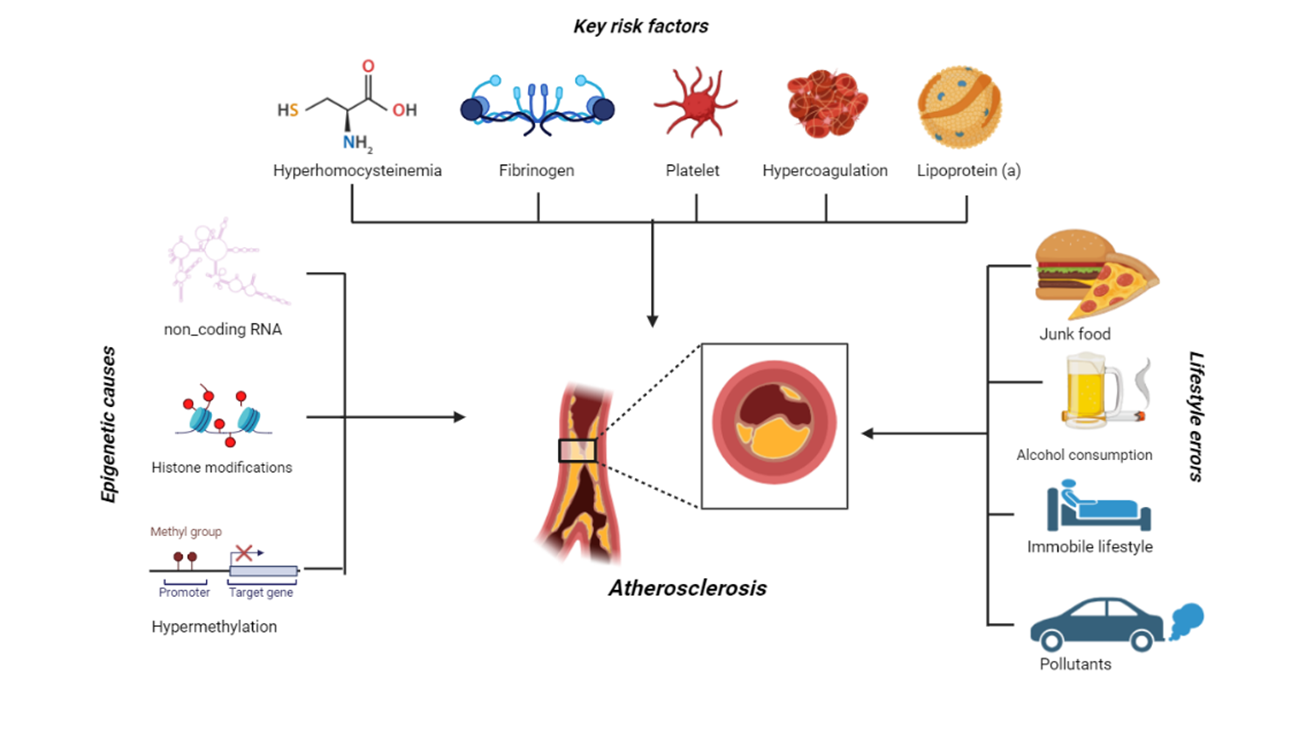
- The review revolves around the modifiable and non-modifiable risk factors leading to atherosclerosis.
- Key risk factors and epigenetics causes of atherosclerosis and atherosclerosis related Cardio vascular Diseases (CVD).
- The article explains in detail regarding the pathophysiology of atherosclerosis.
- The progression and stages of atherosclerosis.
GRAPHICAL ABSTRACT
Downloads
References
S Jebari-Benslaiman, U Galicia-Garcia, A Larrea-Sebal, JR Olaetxea, I Alloza, K Vandenbroeck, A Benito-Vicente and C Martín. Pathophysiology of atherosclerosis. Int. J. Mol. Sci. 2022; 23, 3346.
HT Lee, S Oh, DH Yoo and YW Kwon. The key role of DNA methylation and histone acetylation in epigenetics of atherosclerosis. J. Lipid Atheroscler. 2020; 9, 419.
C Weber and H Noels. Atherosclerosis: Current pathogenesis and therapeutic options. Nat. Med. 2011; 17, 1410-22.
JB Rubin and WB Borden. Coronary heart disease in young adults. Curr. Atheroscler. Rep. 2012; 14, 140-9.
S Kovacic and M Bakran. Genetic susceptibility to atherosclerosis. Stroke Res. Treat. 2012; 26, 362941.
D Skuratovskaia, N Vulf, A Komar, E Kirienkova and L Litvinova. Promising directions in atherosclerosis treatment based on epigenetic regulation using microRNAs and long noncoding RNAs. Biomolecules 2019; 9, 226.
AO Jackson, MA Regine, C Subrata and S Long. Molecular mechanisms and genetic regulation in atherosclerosis. IJC Heart Vasculature 2018; 21, 36-44.
A Fernández-Sanlés, S Sayols-Baixeras, I Subirana, IR Degano and R Elosua. Association between DNA methylation and coronary heart disease or other atherosclerotic events: A systematic review. Atherosclerosis 2017; 263, 325-33.
Y Shen, C Peng, Q Bai, Y Ding, X Yi, H Du, L He, D Zhou and X Chen. Epigenome-wide association study indicates hypomethylation of MTRNR2L8 in large-artery atherosclerosis stroke. Stroke 2019; 50, 1330-8.
DV Fyodorov, BR Zhou, AI Skoultchi and Y Bai. Emerging roles of linker histones in regulating chromatin structure and function. Nature Rev. Mol. Cell Biol. 2018; 19, 192-206.
P Sharma, J Kumar, G Garg, A Kumar, A Patowary, G Karthikeyan, L Ramakrishnan, V Brahmachari and S Sengupta. Detection of altered global DNA methylation in coronary artery disease patients. DNA Cell Biol. 2008; 27, 357-65.
SC Bergheanu, MC Bodde and JW Jukema. Pathophysiology and treatment of atherosclerosis: Current view and future perspective on lipoprotein modification treatment. Neth. Heart J. 2017; 25, 231-42.
S Xu, J Pelisek and ZG Jin. Atherosclerosis is an epigenetic disease. Trends Endocrinol. Metab. 2018; 29, 739-42.
MW Feinberg and KJ Moore. MicroRNA regulation of atherosclerosis. Circ. Res. 2016; 118, 703-20.
B Aryal and Y Suárez. Non-coding RNA regulation of endothelial and macrophage functions during atherosclerosis. Vasc. Pharmacol. 2019; 114, 64-75.
X He, Y Yang, Q Wang, J Wang, S Li, C Li, T Zong, X Li, Y Zhang, Y Zou and T Yu. Expression profiles and potential roles of transfer RNA‐derived small RNAs in atherosclerosis. J. Cell. Mol. Med. 2021; 25, 7052-65.
JB Pierce and MW Feinberg. Long noncoding RNAs in atherosclerosis and vascular injury: Pathobiology, biomarkers, and targets for therapy. Arterioscler. Thromb. Vasc. Biol. 2020; 40, 2002-17.
R Griñán, M Canyelles, D Santos, J Martínez, JC Souto, A Martínez-Perez, JM Soria, J Julve, C Rodríguez, F Blanco-Vaca and N Rotllan. Identification of a potential microRNA as an important regulator in HHCY-related atherosclerosis. Atherosclerosis 2023; 379, S11.
P Skroblin and M Mayr. “Going long”: Long non-coding RNAs as biomarkers. Circ. Res. 2014; 115, 607-9.
J Chi, J Li, J Jia, T Zhang, X Liu and L Yi. Long non-coding RNA ANRIL in gene regulation and its duality in atherosclerosis. Curr. Med. Sci. 2017; 37, 816-22.
B Xu, Z Xu, Y Chen, N Lu, Z Shu and X Tan. Genetic and epigenetic associations of ANRIL with coronary artery disease and risk factors. BMC Med. Genomics 2021; 14, 240.
I Razeghian-Jahromi, A Karimi Akhormeh and MJ Zibaeenezhad. The role of ANRIL in atherosclerosis. Dis. Markers 2022; 2022, 8859677.
F Carbone, F Montecucco, S Xu, M Banach, T Jamialahmadi and A Sahebkar. Epigenetics in atherosclerosis: Key features and therapeutic implications. Expert Opin. Ther. Targets 2020; 24, 719-21.
YC Chen, AL Huang, TS Kyaw, A Bobik and K Peter. Atherosclerotic plaque rupture: Identifying the straw that breaks the camel’s back. Arterioscler. Thromb. Vasc. Biol. 2016; 36, e63-e72.
MP Turunen, E Aavik and S Ylä-Herttuala. Epigenetics and atherosclerosis. Biochimica et Biophysica Acta 2009; 1790, 886-91.
DA Chistiakov, AN Orekhov and YV Bobryshev. Treatment of cardiovascular pathology with epigenetically active agents: Focus on natural and synthetic inhibitors of DNA methylation and histone deacetylation. Int. J. Cardiol. 2017; 227, 66-82.
Y Dai, D Chen and T Xu. DNA methylation aberrant in atherosclerosis. Front. Pharmacol. 2022; 13, 815977.
S Tabaei and SS Tabaee. DNA methylation abnormalities in atherosclerosis. Artif. Cells Nanomed. Biotechnol. 2019; 47, 2031-41.
D Weiss, JJ Kools and WR Taylor. Angiotensin II-induced hypertension accelerates the development of atherosclerosis in apoE-deficient mice. Circulation 2001; 103, 448-54.
A Daugherty, MW Manning and LA Cassis. Angiotensin II promotes atherosclerotic lesions and aneurysms in apolipoprotein E-deficient mice. J. Clin. Invest. 2000; 105, 1605-12.
I Nicorescu, GM Dallinga, MPD Winther, ES Stroes and M Bahjat. Potential epigenetic therapeutics for atherosclerosis treatment. Atherosclerosis 2019; 281, 189-97.
Z Ding, S Liu, X Wang, X Deng, Y Fan, C Sun, Y Wang and JL Mehta. Hemodynamic shear stress via ROS modulates PCSK9 expression in human vascular endothelial and smooth muscle cells and along the mouse aorta. Antioxid. Redox Signaling 2015; 22, 760-71.
HS Bassiouny, CK Zarins, MH Kadowaki and S Glagov. Hemodynamic stress and experimental aortoiliac atherosclerosis. J. Vasc. Surg. 1994; 19, 426-34.
W Jiang, DK Agrawal and CS Boosani. Cell specific histone modifications in atherosclerosis. Mol. Med. Rep. 2018; 18, 1215-24.
AW Khan, F Paneni and KA Jandeleit-Dahm. Cell-specific epigenetic changes in atherosclerosis. Clin. Sci. 2021; 135, 1165-87.
A Greißel, M Culmes, R Burgkart, A Zimmermann, HH Eckstein, A Zernecke and J Pelisek. Histone acetylation and methylation significantly change with severity of atherosclerosis in human carotid plaques. Cardiovasc. Pathol. 2016; 25, 79-86.
BK Zhang, X Lai and S Jia. Epigenetics in atherosclerosis: A clinical perspective. Discovery Med. 2015; 19, 73-80.
P Bompada, I Goncalves, C Wu, R Gao, J Sun, BA Mir, C Luan, E Renström, L Groop, J Weng and O Hansson. Epigenome-wide histone acetylation changes in peripheral blood mononuclear cells in patients with type 2 diabetes and atherosclerotic disease. Biomedicines 2021; 9, 1908.
DV Mohankrishna. Histone acetylation and gene expression in neural cells-probed by small molecule modulators. Ph. D. Jawaharlal Nehru Centre for Advanced Scientific Research.
S Ye, CR Gale and CN Martyn. Variation in the matrix metalloproteinase-1 gene and risk of coronary heart disease. Eur. Heart J. 2003; 24, 1668-71.
RJ Wierda, IM Rietveld, MCV Eggermond, JA Belien, EWV Zwet, JH Lindeman and PJVD Elsen. Global histone H3 lysine 27 triple methylation levels are reduced in vessels with advanced atherosclerotic plaques. Life Sci. 2015; 129, 3-9.
A Greißel, M Culmes, R Napieralski, E Wagner, H Gebhard, M Schmitt, A Zimmermann, HH Eckstein, A Zernecke and J Pelisek. Alternation of histone and DNA methylation in human atherosclerotic carotid plaques. Thromb. Haemostasis 2015; 114, 390-402.
S Xu, D Kamato, PJ Little, S Nakagawa, J Pelisek and ZG Jin Targeting epigenetics and non-coding RNAs in atherosclerosis: From mechanisms to therapeutics. Pharmacol. Ther. 2019; 196, 15-43.
IS Shin, JM Kim, KL Kim, SY Jang, ES Jeon, SH Choi, DK Kim, W Suh and YW Kim. Early growth response factor-1 is associated with intraluminal thrombus formation in human abdominal aortic aneurysm. J. Am. Coll. Cardiol. 2009; 53, 792-9.
NA Abdel-Malak, M Mofarrahi, D Mayaki, LM Khachigian and SNA Hussain. Early growth response-1 regulates angiopoietin-1-induced endothelial cell proliferation, migration, and differentiation. Arterioscler. Thromb. Vasc. Biol. 2009; 29, 209-16.
S Legartová, L Stixova, H Strnad, S Kozubek, N Martinet, FJ Dekker, M Franek and E Bartova. Basic nuclear processes affected by histone acetyltransferases and histone deacetylase inhibitors. Epigenomics 2013; 5, 379-96.
JC Rice and CD Allis. Histone methylation versus histone acetylation: New insights into epigenetic regulation. Curr. Opin. Cell Biol. 2001; 13, 263-73.
HJ Kim and SC Bae. Histone deacetylase inhibitors: Molecular mechanisms of action and clinical trials as anti-cancer drugs. Am. J. Transl. Res. 2011; 3, 166.
JH Choi, KH Nam, J Kim, MW Baek, JE Park, HY Park, HJ Kwon, OS Kwon, DY Kim and GT Oh. Trichostatin a exacerbates atherosclerosis in low density lipoprotein receptor - deficient mice. Arterioscler. Thromb. Vasc. Biol. 2005; 25, 2404-9.
AJ Bowes, MI Khan, Y Shi, L Robertson and GH Werstuck. Valproate attenuates accelerated atherosclerosis in hyperglycemic apoE-deficient mice: Evidence in support of a role for endoplasmic reticulum stress and glycogen synthase kinase-3 in lesion development and hepatic steatosis. Am. J. Pathol. 2009; 174, 330-42.
KM VanderMolen, W McCulloch, CJ Pearce and NH Oberlies. Romidepsin (Istodax, NSC 630176, FR901228, FK228, depsipeptide): A natural product recently approved for cutaneous T-cell lymphoma. J. Antibiot. 2011; 64, 525-31.
M Salmon, D Gomez, E Greene, L Shankman and GK Owens. Cooperative binding of KLF4, pELK-1, and HDAC2 to a G/C repressor element in the SM22α promoter mediates transcriptional silencing during SMC phenotypic switching in vivo. Circ. Res. 2012; 111, 685-96.
X Dong and Z Weng. The correlation between histone modifications and gene expression. Epigenomics 2013; 5, 113-6.
EL Greer and Y Shi. Histone methylation: A dynamic mark in health, disease and inheritance. Nat. Rev. Genet. 2012; 13, 343-57.
Y Xiaoling, Z Li, L ShuQiang, M Shengchao, Y Anning, D Ning, L Nan, J Yuexia, Y Xiaoming, L Guizhong and J Yideng. Hyperhomocysteinemia in ApoE-/-mice leads to overexpression of enhancer of zeste homolog 2 via miR-92a regulation. PLoS One 2016; 11, e0167744.
H Hou and H Zhao. Epigenetic factors in atherosclerosis: DNA methylation, folic acid metabolism, and intestinal microbiota. Clin. Chim. Acta 2020; 512, 7-11.
JE Fish, CC Matouk, A Rachlis, S Lin, SC Tai, C D’Abreo and PA Marsden. The expression of endothelial nitric-oxide synthase is controlled by a cell-specific histone code. J. Biol. Chem. 2005; 280, 24824-38.
Y Chan, JE Fish, C D’Abreo, S Lin, GB Robb, AM Teichert, F Karantzoulis-Fegaras, A Keightley, BM Steer and PA Marsden. The cell-specific expression of endothelial nitric-oxide synthase: A role for DNA methylation. J. Biol. Chem. 2004; 279, 35087-100.
GC Chan, JE Fish, IA Mawji, DD Leung, AC Rachlis and PA Marsden. Epigenetic basis for the transcriptional hyporesponsiveness of the human inducible nitric oxide synthase gene in vascular endothelial cells. J. Immunol. 2005; 175, 3846-61.
AM Devlin, R Singh, RE Wade, SM Innis, T Bottiglieri and SR Lentz. Hypermethylation of Fads2 and altered hepatic fatty acid and phospholipid metabolism in mice with hyperhomocysteinemia. J. Biol. Chem. 2007; 282, 37082-90.
NE Hastings, MB Simmers, OG McDonald, BR Wamhoff and BR Blackman. Atherosclerosis-prone hemodynamics differentially regulates endothelial and smooth muscle cell phenotypes and promotes pro-inflammatory priming. Am. J. Physiol. Cell Physiol. 2007; 293, C1824-C1833.
WS Post, PJ Goldschmidt-Clermont, CC Wilhide, AW Heldman, MS Sussman, P Ouyang, EE Milliken and JP Issa. Methylation of the estrogen receptor gene is associated with aging and atherosclerosis in the cardiovascular system. Cardiovasc. Res. 1999; 43, 985-91.
J Kim, JY Kim, KS Song, YH Lee, JS Seo, J Jelinek, PJ Goldschmidt-Clermont and JPJ Issa. Epigenetic changes in estrogen receptor β gene in atherosclerotic cardiovascular tissues and in-vitro vascular senescence. Biochim. Biophys. Acta Mol. Basis Dis. 2007; 1772, 72-80.
C Liu, D Xu, J Sjöberg, P Forsell, M Björkholm and HE Claesson. Transcriptional regulation of 15-lipoxygenase expression by promoter methylation. Exp. Cell Res. 2004; 297, 61-7.
MO Laukkanen, S Mannermaa, MO Hiltunen, S Aittomäki, K Airenne, J Jänne and S Ylä-Herttuala. Local hypomethylation in atherosclerosis found in rabbit ec-sod gene. Arterioscler. Thromb. Vasc. Biol. 1999; 19, 2171-8.
AM Devlin, T Bottiglieri, FE Domann and SR Lentz. Tissue-specific changes in H19 methylation and expression inmice with hyperhomocysteinemia. J. Biol. Chem. 2005; 280, 25506-11.
KC Chen, YS Wang, CY Hu, WC Chang, YC Liao, CY Dai and SHH Juo. OxLDL up‐regulates microRNA‐29b, leading to epigenetic modifications of MMP‐2/MMP‐9 genes: A novel mechanism for cardiovascular diseases. FASEB J. 2011; 25, 1718-28.
A Wild, A Ramaswamy, P Langer, I Celik, V Fendrich, B Chaloupka, B Simon and DK Bartsch. Frequent methylation-associated silencing of the tissue inhibitor of metalloproteinase-3 gene in pancreatic endocrine tumors. J. Clin. Endocrinol. Metab. 2003; 88, 1367-73.
GP White, PM Watt, BJ Holt and PG Holt. Differential patterns of methylation of the IFN-γ promoter at CpG and non-CpG sites underlie differences in IFN-γ gene expression between human neonatal and adult CD45RO - T cells. J. Immunol. 2002; 168, 2820-7.
XH Lin, C Guo, LJ Gu and TF Deuel. Site-specific methylation inhibits transcriptional activity of platelet-derived growth factor A-chain promoter. J. Biol. Chem. 1993; 268, 17334-40.
Y Tanaka, K Fukudome, M Hayashi, S Takagi and O Yoshie. Induction of ICAM‐1 and LFA‐3 by Tax1 of human T‐cell leukemia virus type 1 and mechanism of down‐regulation of ICAM‐1 or LFA‐1 in adult‐T‐cell‐leukemia cell lines. Int. J. Cancer 1995; 60, 554-61.
M Schroeder and MJ Mass. CpG methylation inactivates the transcriptional activity of the promoter of the humanp53tumor suppressor gene. Biochem. Biophys. Res. Commun. 1997; 235, 403-6.
DA Pan and DG Hardie. A homologue of AMP-activated protein kinase in Drosophila melanogaster is sensitive to AMP and is activated by ATP depletion. Biochem. J. 2002; 367, 179-86.
IJ Kullo, GT Gau and AJ Tajik. Novel risk factors for atherosclerosis. Mayo Clin. Proc. 2000; 75, 369-80.
SG Chrysant and GS Chrysant. The current status of homocysteine as a risk factor for cardiovascular disease: A mini review. Expert Rev. Cardiovasc. Ther. 2018; 16, 559-65.
R Guieu, J Ruf and G Mottola. Hyperhomocysteinemia and cardiovascular diseases. Ann. Biol. Clin. 2022; 80, 7-14.
D Yuan, J Chu, H Lin, G Zhu, J Qian, Y Yu, T Yao, F Ping, F Chen and X Liu. Mechanism of homocysteine-mediated endothelial injury and its consequences for atherosclerosis. Front. Cardiovasc. Med. 2023; 9, 1109445.
KS McCully. Homocysteine and the pathogenesis of atherosclerosis. Expert Rev. Clin. Pharmacol. 2015; 8, 211-9.
JC Fruchart, MC Nierman, ESG Stroes, JJP Kastelein and P Duriez. New risk factors for atherosclerosis and patient risk assessment. Circulation 2004; 109, III-15-III-19.
E Ernst and KL Resch. Fibrinogen as a cardiovascular risk factor: A meta-analysis and review of the literature. Ann. Intern. Med. 1993; 118, 956-63.
E Ernst. The role of fibrinogen as a cardiovascular risk factor. Atherosclerosis 1993; 100, 1-12.
R Vilar, RJ Fish, A Casini and M Neerman-Arbez. Fibrin (ogen) in human disease: Both friend and foe. Haematologica 2020; 105, 284.
G Murdaca, F Spanò, P Cagnati and F Puppo. Free radicals and endothelial dysfunction: Potential positive effects of TNF-α inhibitors. Redox Rep. 2013; 18, 95-9.
MD Trip, VM Cats, FJLV Capelle and J Vreeken. Platelet hyperreactivity and prognosis in survivors of myocardial infarction. N. Engl. J. Med. 1990; 322, 1549-54.
E Thaulow, J Erikssen, L Sandvik, H Stormorken and PF Cohn. Blood platelet count and function are related to total and cardiovascular death in apparently healthy men. Circulation 1991; 84, 613-7.
J Kutti. Smoking, platelet reactivity and fibrinogen. Adv. Exp. Med. Biol. 1990; 273, 129-34.
DP Thomas and HR Roberts. Hypercoagulability in venous and arterial thrombosis. Ann. Intern. Med. 1997; 126, 638-44.
M Margaglione, V Brancaccio, N Giuliani, G D’nous thrombosis in carriers of the prothrombin G→ A20210 gene variant. Ann. Intern. Med. 1998; 129, 89-93.
RA Asherson, MA Khamashta, JO Ordi-Ros, RH Derksen, SJ Machin, JO Barquinero, HH Outt, EN Harris, MI Vilardell-Torres and GR Hughes. The “primary” antiphospholipid syndrome: Major clinical and serological features. Medicine 1989; 68, 366-74.
AM Scanu. Lipoprotein (a): A genetic risk factor for premature coronary heart disease. JAMA 1992; 267, 3326-9.
CG Fabricant, J Fabricant, MM Litrenta and CR Minick. Virus-induced atherosclerosis. J. Exp. Med. 1978; 148, 335-40.
J Danesh, R Collins and R Peto. Chronic infections and coronary heart disease: Is there a link? Lancet 1997; 350, 430-6.
JB Muhlestein. Antibiotic therapy for treatment of Chlamydia to prevent coronary heart disease events. Curr. Atheroscler. Rep. 2000; 2, 336-41.
K Sakakura, M Nakano, F Otsuka, E Ladich, FD Kolodgie and R Virmani. Pathophysiology of atherosclerosis plaque progression. Heart Lung Circ. 2013; 22, 399-411.
AL Catapano, I Graham, G De Backer, O Wiklund, M Chapman, H Drexel, AW Hoes, CS Jennings, U Landmesser, TR Pedersen and Z Reiner. Wytyczne ESC/EAS dotyczace leczenia zaburzeń lipidowych w 2016 roku (in Polish). Kardiol. Pol. 2016; 74, 1234-318.
V Grimaldi, MT Vietri, C Schiano, A Picascia, MR de Pascale, C Fiorito, A Casamassimi and C Napoli. Epigenetic reprogramming in atherosclerosis. Curr. Atheroscler. Rep. 2015; 17, 1-2.
F Kronenberg and G Utermann. Lipoprotein (a): Resurrected by genetics. J. Intern. Med. 2013; 273, 6-30.
A Rognoni, C Cavallino, A Veia, S Bacchini, R Rosso, M Facchini, GG Secco, A Lupi, F Nardi, F Rametta and AS Bongo. Pathophysiology of atherosclerotic plaque development. Cardiovasc. Hematol. Agents Med. Chem. 2015; 13, 10-3.
Y Zhang, J Mei, J Li, Y Zhang, Q Zhou and F Xu. DNA methylation in atherosclerosis: A new perspective. Evidence Based Complementary Altern. Med. 2021; 2021, 6623657.
PM Ridker. Anticytokine agents: Targeting interleukin signaling pathways for the treatment of atherothrombosis. Circ. Res. 2019; 124, 437-50.
PM Ridker, M Devalaraja, FMM Baeres, MDM Engelmann, GK Hovingh, M Ivkovic, L Lo, D Kling, P Pergola, D Raj and P Libby. IL-6 inhibition with ziltivekimab in patients at high atherosclerotic risk (rescue): A double-blind, randomised, placebo-controlled, phase 2 trial. Lancet 2021; 397, 2060-9.
RF DeBusk, NH Miller, HR Superko, CA Dennis, RJ Thomas, HT Lew, WE Berger, RS Heller, J Rompf, D Gee and HC Kraemer. A case-management system for coronary risk factor modification after acute myocardial infarction. Ann. Intern. Med. 1994; 120, 721-9.
GC Fonarow, A Gawlinski, S Moughrabi and JH Tillisch. Improved treatment of coronary heart disease by implementation of a Cardiac Hospitalization Atherosclerosis Management Program (CHAMP). Am. J. Cardiol. 2001; 87, 819-22.
DE Harris, NB Record, GW Gipson and TA Pearson. Lipid lowering in a multidisciplinary clinic compared with primary physician management. Am. J. Cardiol. 1998; 81, 929-33.
DI Swerdlow, DA Rider, A Yavari, MW Lindholm, GV Campion and SE Nissen. Treatment and prevention of lipoprotein (a)-mediated cardiovascular disease: The emerging potential of RNA interference therapeutics. Cardiovasc. Res. 2022; 118, 1218-31.
KM Ali, A Wonnerth, K Huber and J Wojta. Cardiovascular disease risk reduction by raising HDL cholesterol - current therapies and future opportunities. Br. J. Pharmacol. 2012; 167, 1177-94.
AL Catapano, I Graham, GD Backer, O Wiklund, MJ Chapman, H Drexel, AW Hoes, CS Jennings, U Landmesser, TR Pedersen and Ž Reiner. 2016 ESC/EAS guidelines for the management of dyslipidaemias. Polish Heart J. 2016; 74, 1234-318.
MH Davidson, MA Dillon, B Gordon, P Jones, J Samuels, S Weiss, J Isaacsohn, P Toth and SK Burke. Colesevelam hydrochloride (cholestagel): A new, potent bile acid sequestrant associated with a low incidence of gastrointestinal side effects. Arch. Intern. Med. 1999; 159, 1893-900.
T Umemoto, S Subramanian, Y Ding, L Goodspeed, S Wang, CY Han, AS Teresa, J Kim, KD O’Brien and A Chait. Inhibition of intestinal cholesterol absorption decreases atherosclerosis but not adipose tissue inflammation. J. Lipid Res. 2012; 53, 2380-9.
J Yoon, S Subramanian, Y Ding, S Wang, L Goodspeed, B Sullivan, J Kim, KD O’Brien and A Chait. Chronic insulin therapy reduces adipose tissue macrophage content in LDL-receptor-deficient mice. Diabetologia 2011; 54, 1252-60.
R Tummala, M Gupta, AR Devanabanda, D Bandyopadhyay, WS Aronow, KK Ray, M Mamas and RK Ghosh. Bempedoic acid and its role in contemporary management of hyperlipidemia in atherosclerosis. Ann. Med. 2022; 54, 1287-96.
M Florentin, EN Liberopoulos, DP Mikhailidis and MS Elisaf. Colesevelam hydrochloride in clinical practice: A new approach in the treatment of hypercholesterolaemia. Curr. Med. Res. Opin. 2008; 24, 995-1009.
AFG Cicero, F Fogacci and I Cincione. Evaluating pharma-cokinetics of bempedoic acid in the treatment of hypercholesterolemia. Expert Opin. Drug Metab. Toxicol. 2021; 17, 1031-8.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






