Different Doxorubicin Sensitivity Across Various Human Cancer Cell Lines
DOI:
https://doi.org/10.48048/tis.2024.8566Keywords:
Doxorubicin, Cancer cell lines, Drug sensitivity, Anticancer drug resistance, ChemotherapyAbstract
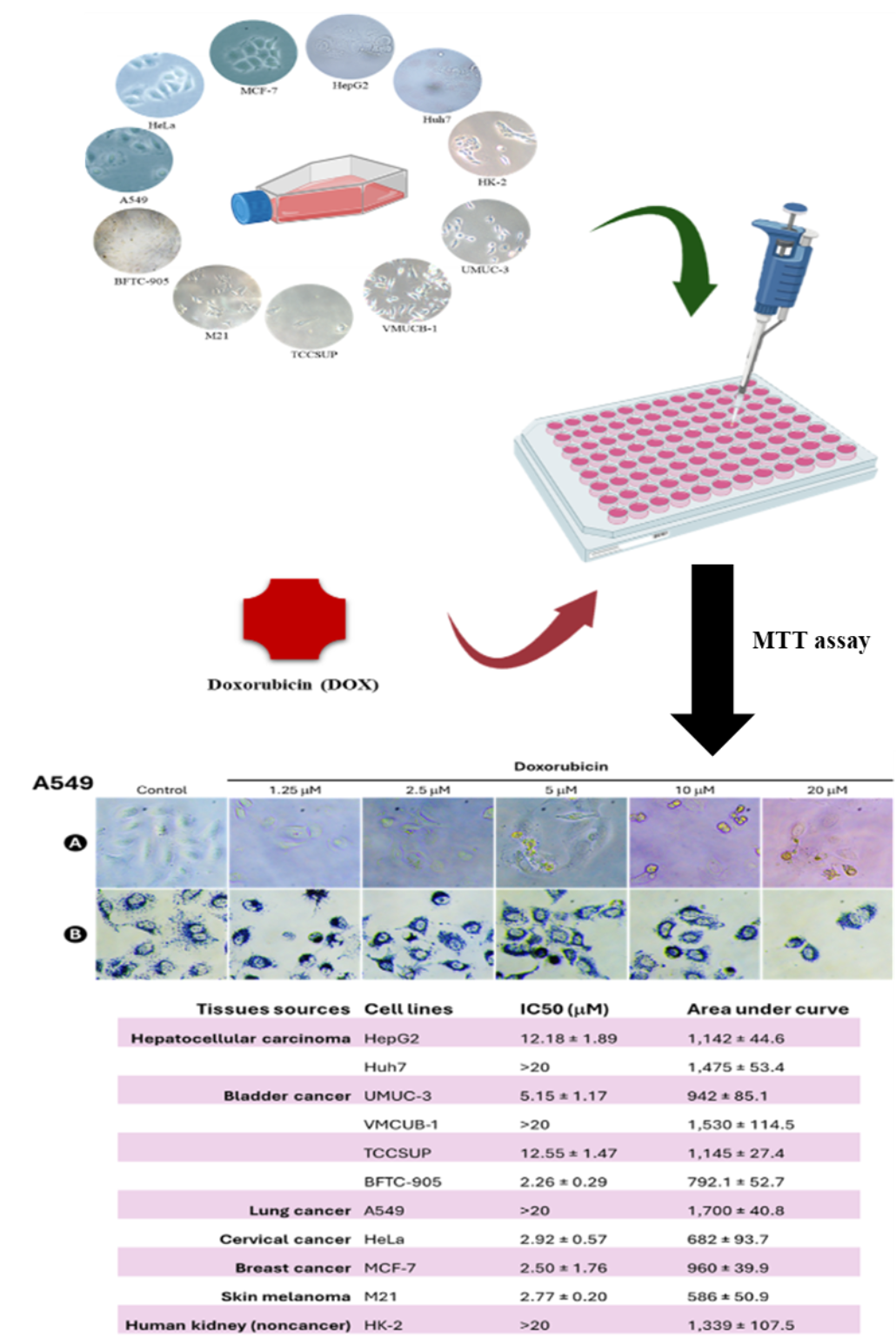
Doxorubicin (Dox) is a highly potent chemotherapeutic agent, approved by FDA since 1974, for treating a broad spectrum of cancers. However, development of severe side effects and drug resistance are main issues that limit the clinical use of Dox. Herein, we aimed to investigate the sensitivity of Dox in a variety of human cancer cell lines. Eleven cell lines were tested in this study including 2 hepatocellular carcinoma (HCC) cell lines (HepG2 and Huh7), 4 bladder cancer (BlCa) cell lines (UMUC-3, VMCUB-1, TCCSUP and BFTC-905), a lung cancer cell line (A549), a cervical carcinoma cell line (HeLa), a breast cancer cell line (MCF-7), a skin melanoma cell line (M21) and a noncancer human kidney cell line (HK-2). The MTT assay was employed for the determination of cytotoxicity. Cells were treated with various concentrations of Dox for 24 h. The half-maximal inhibitory concentration (IC50) of Dox was calculated to compare the Dox sensitivity among tested cell lines. The result showed that IC50 of Dox in HepG2, Huh7, UMUC-3, VMCUB-1, TCCSUP, BFTC-905, A549, HeLa, MCF-7, M21 and HK-2 cells were 12.2, > 20, 5.1, > 20, 12.6, 2.3, > 20, 2.9, 2.5, 2.8 and > 20 mM, respectively. BFTC-905 had the lowest IC50 value, therefore, it was the most sensitive to Dox. In contrast, Huh7, VMCUB-1 and A549 cells were resistant to Dox with IC50 values > 20 mM. Conclusions, we demonstrated that different human malignant cell lines had different sensitivity to Dox. BFTC-905 BlCa cell line had the highest sensitivity to Dox. Huh7, VMCUB-1 and A549 cell lines were resistant to Dox. Perhaps, the different Dox sensitivity in different cell lines was due to the different acquisition of Dox resistance mechanisms. Huh7, VMCUB-1 and A549 cell lines could be suitable cell models to investigate the molecular mechanism of Dox resistance in different cancers.
HIGHLIGHTS
- Different types of cancer cell lines exhibited different sensitivity to doxorubicin.
- BFTC-905, MCF-7 and M21 cell lines were sensitive to doxorubicin.
- HepG2, UMUC-3, TCCSUP and HeLa cells were moderately sensitive to doxorubicin.
- Huh7, VMCUB-1, A549 and HK-2 cells were resistant to doxorubicin.
- Huh7, VMCUB-1 and A549 cell lines could be suitable cell models for investigating the molecular pathways of Dox resistance in different types of cancers.
GRAPHICAL ABSTRACT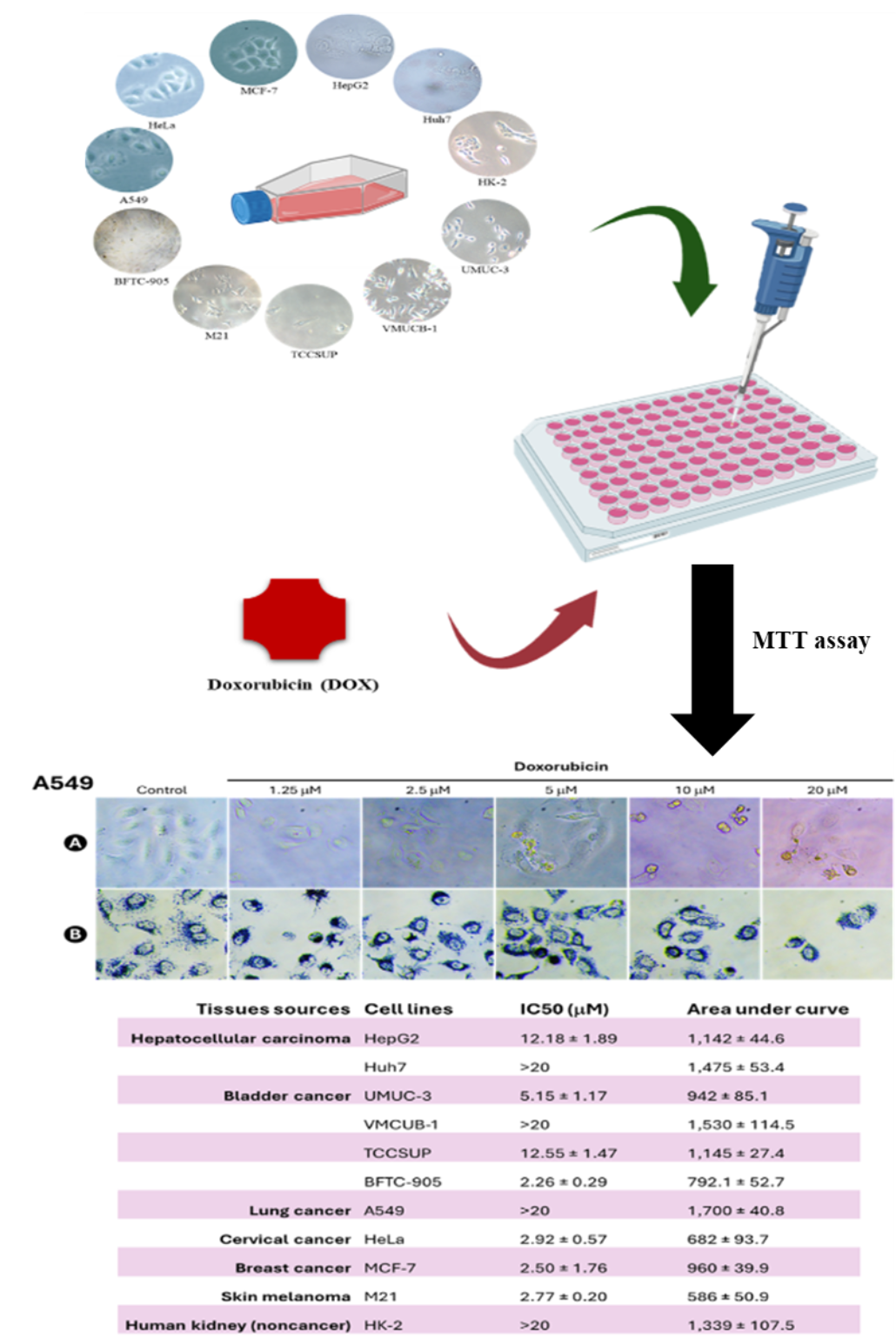
Downloads
References
G Cassinelli. The roots of modern oncology: From discovery of new antitumor anthracyclines to their clinical use. Tumori 2016; 2016, 226-35.
M Cagel, E Grotz, E Bernabeu, MA Moretton and DA Chiappetta. Doxorubicin: Nanotechnological overviews from bench to bedside. Drug Discovery Today 2017; 22, 270-81.
AN Linders, IB Dias, TL Fernandez, CG Tocchetti, N Bomer and PVD Meer. A review of the pathophysiological mechanisms of doxorubicin-induced cardiotoxicity and aging. NPJ Aging 2024; 10, 9.
K Chatterjee, J Zhang, N Honbo and JS Karliner. Doxorubicin cardiomyopathy. Cardiology 2010; 115, 155-62.
M Kciuk, A Gielecinska, S Mujwar, D Kolat, Z Kaluzinska-Kolat, I Celik and R Kontek. Doxorubicin-an agent with multiple mechanisms of anticancer activity. Cells 2023; 12, 659.
R Mattioli, A Ilari, B Colotti, L Mosca, F Fazi and G Colotti. Doxorubicin and other anthracyclines in cancers: Activity, chemoresistance and its overcoming. Mol. Aspects Med. 2023; 93, 101205.
O Tacar, P Sriamornsak and CR Dass. Doxorubicin: An update on anticancer molecular action, toxicity and novel drug delivery systems. J. Pharm. Pharmacol. 2013; 65, 157-70.
CF Thorn, C Oshiro, S Marsh, T Hernandez-Boussard, H McLeod, TE Klein and RB Altman. Doxorubicin pathways: Pharmacodynamics and adverse effects. Pharmacogenet. Genomics 2011; 21, 440-6.
MC Asensio-Lopez, F Soler, D Pascual-Figal, F Fernandez-Belda and A Lax. Doxorubicin-induced oxidative stress: The protective effect of nicorandil on HL-1 cardiomyocytes. PLoS One 2017; 12, e0172803.
CY Kong, Z Guo, P Song, X Zhang, YP Yuan, T Teng, L Yan and QZ Tang. Underlying the mechanisms of doxorubicin-induced acute cardiotoxicity: Oxidative stress and cell death. Int. J. Biol. Sci. 2022; 18, 760-70.
B Mansoori, A Mohammadi, S Davudian, S Shirjang and B Baradaran. The different mechanisms of cancer drug resistance: A brief review. Adv. Pharm. Bull. 2017; 7, 339-48.
C Christowitz, T Davis, A Isaacs, G van Niekerk, S Hattingh and AM Engelbrecht. Mechanisms of doxorubicin-induced drug resistance and drug resistant tumour growth in a murine breast tumour model. BMC Cancer 2019; 19, 757.
J Cox and S Weinman. Mechanisms of doxorubicin resistance in hepatocellular carcinoma. Hepatic Oncol. 2016; 3, 57-9.
G Kibria, H Hatakeyama, K Akiyama, K Hida and H Harashima. Comparative study of the sensitivities of cancer cells to doxorubicin, and relationships between the effect of the drug-efflux pump P-gp. Biol. Pharm. Bull. 2014; 37, 1926-35.
JL Sebaugh. Guidelines for accurate EC50/IC50 estimation. Pharm. Stat. 2011; 10, 128-34.
S Aykul and E Martinez-Hackert. Determination of half-maximal inhibitory concentration using biosensor-based protein interaction analysis. Anal. Biochem. 2016; 508, 97-103.
Y He, Q Zhu, M Chen, Q Huang, W Wang, Q Li, Y Huang and W Di. The changing 50 % inhibitory concentration (IC50) of cisplatin: A pilot study on the artifacts of the MTT assay and the precise measurement of density-dependent chemoresistance in ovarian cancer. Oncotarget 2016; 7, 70803-21.
MJ Garnett, EJ Edelman, SJ Heidorn, CD Greenman, A Dastur, KW Lau, P Greninger, IR Thompson, X Luo, J Soares, Q Liu, F Iorio, D Surdez, L Chen, RJ Milano, GR Bignell, AT Tam, H Davies, JA Stevenson, …, CH Benes. Systematic identification of genomic markers of drug sensitivity in cancer cells. Nature 2012; 483, 570-5.
Sanger Institute. Genomics of drug sensitivity in cancer, Available at: https://www.cancerrxgene.org/ compound/Doxorubicin/133/overview/ic50?tissue=PANCANCER&screening_set=GDSC1, accessed October 2023.
T Kalliokoski, C Kramer, A Vulpetti and P Gedeck. Comparability of mixed IC(5)(0) data - a statistical analysis. PLoS One 2013; 8, e61007.
IR Dubbelboer, N Pavlovic, F Heindryckx, E Sjogren and H Lennernas. Liver cancer cell lines treated with doxorubicin under normoxia and hypoxia: Cell viability and oncologic protein profile. Cancers 2019; 11, 1024.
M Louisa, FD Suyatna, SI Wanandi, PB Asih and D Syafruddin. Differential expression of several drug transporter genes in HepG2 and Huh-7 cell lines. Adv. Biomed. Res. 2016; 5, 104.
S Phoyen, A Sanpavat, C Ma-On, U Stein, N Hirankarn, P Tangkijvanich, D Jindatip, P Whongsiri and C Boonla. H4K20me3 upregulated by reactive oxygen species is associated with tumor progression and poor prognosis in patients with hepatocellular carcinoma. Heliyon 2023; 9, e22589.
P Whongsiri, C Pimratana, U Wijitsettakul, D Jindatip, A Sanpavat, WA Schulz, MJ Hoffmann, W Goering and C Boonla. LINE-1 ORF1 protein is up-regulated by reactive oxygen species and associated with bladder urothelial carcinoma progression. Cancer Genomics Proteomics 2018; 15, 143-51.
P Whongsiri, C Pimratana, U Wijitsettakul, A Sanpavat, D Jindatip, MJ Hoffmann, W Goering, WA Schulz and C Boonla. Oxidative stress and LINE-1 reactivation in bladder cancer are epigenetically linked through active chromatin formation. Free Radical Biol. Med. 2019; 134, 419-28.
S Homma, Y Ishii, Y Morishima, T Yamadori, Y Matsuno, N Haraguchi, N Kikuchi, H Satoh, T Sakamoto, N Hizawa, K Itoh and M Yamamoto. Nrf2 enhances cell proliferation and resistance to anticancer drugs in human lung cancer. Clin. Cancer Res. 2009; 15, 3423-32.
V Ngo and ML Duennwald. Nrf2 and oxidative stress: A general overview of mechanisms and implications in human disease. Antioxidants 2022; 11, 2345.
B Shrestha, AR Pokhrel, S Darsandhari, P Parajuli, JK Sohng and RP Pandey. Engineering Streptomyces peucetius for doxorubicin and daunorubicin biosynthesis. In: D Arora, C Sharma, S Jaglan and E Lichtfouse (Eds.). Pharmaceuticals from microbes. Springer, Cham, Switzerland, 2019, p. 191-209.
P Paramasivan, JD Kumar, R Baskaran, CF Weng and VV Padma. Reversal of doxorubicin resistance in lung cancer cells by neferine is explained by nuclear factor erythroid-derived 2-like 2 mediated lung resistance protein down regulation. Cancer Drug Resist. 2020; 3, 647-65.
H Sadeghi-Aliabadi, M Minaiyan and A Dabestan. Cytotoxic evaluation of doxorubicin in combination with simvastatin against human cancer cells. Res. Pharm. Sci. 2010; 5, 127-33.
SH Tsou, TM Chen, HT Hsiao and YH Chen. A critical dose of doxorubicin is required to alter the gene expression profiles in MCF-7 cells acquiring multidrug resistance. PLoS One 2015; 10, e0116747.
XJ Fang, H Jiang, YQ Zhu, LY Zhang, QH Fan and Y Tian. Doxorubicin induces drug resistance and expression of the novel CD44st via NF-kappaB in human breast cancer MCF-7 cells. Oncol. Rep. 2014; 31, 2735-42.
L Panasci, BJ Jean-Claude, D Vosilescu, A Mustafa, S Damian, Z Damian, E Georges, Z Liu, G Batist and B Leyland-Jones. Sensitization to doxorubicin resistance in breast cancer cell lines by tamoxifen and megestrol acetate. Biochem. Pharmacol. 1996; 52, 1097-102.
Y Guo, Y Tang, G Lu and J Gu. p53 at the crossroads between doxorubicin-induced cardiotoxicity and resistance: A nutritional balancing act. Nutrients 2023; 15, 2259.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






