Microanatomy of the Testes and Male Genital Ducts of Banded Krait Bungarus fasciatus (Schneider, 1801)
DOI:
https://doi.org/10.48048/tis.2024.8332Keywords:
Banded krait, Histochemistry, Histology, Male genital duct, Reproductive system, TestisAbstract
The Banded krait is a species of venomous snakes that ranks among the top 5 in Thailand, with a significance role as a predator for controlling other animals. Despite numerous studies on the ecology, the microanatomy of body systems has still not reported clearly. The present study was to investigate the microanatomy of the testes and male genital ducts of Banded krait that collected from Phatthalung Province, Thailand. The snakes were collected after anesthetized, and their testes and genital ducts were fixed in Bouin’s fixative. The reproductive organs were collected during July to October 2022, and were sectioned of 5-µm-thick pieces for staining with Harris’s hematoxylin and eosin (H&E), periodic acid Schiff’s (PAS), alcian blue (AB) pH 2.5 and pH 1.0, bromophenol blue (BB) and Masson’s trichrome (MT). The testes contained the seminiferous tubules with 7 stages of germ cells: (I) type A spermatogonium, (II) type B spermatogonium, (III) primary spermatocyte, (IV) secondary spermatocyte, (V) round spermatid, (VI) elongated spermatid and (VII) spermatozoa. Besides, Sertoli and Leydig’s cells were appeared in the seminiferous tubules and the interstitial area, respectively. During the sampling period, the spermatozoa were observed in the seminiferous tubules and genital ducts. The extra-testicular rete testis, ductuli efferentes and ductus epididymis were embedded in the epididymal sheath. The epithelial cells of rete testis, ductus epididymis, as well as ductus deferens possessed microvilli, whereas those of the ductuli efferentes composed of cilia. The epithelial cells of all portions showed the positive staining with PAS and BB due to the presence of neutral glycoproteins and proteins, respectively, and the negative staining for AB pH 2.5 and pH 1.0 because of the absence of acidic glycoproteins.
HIGHLIGHTS
- During the sampling period from July to October, the Banded Krait exhibited active spermatogenesis
- The rete testis could be divided into 2 portions: intra-rete testis and extra-rete testis
- The ductus epididymis was divided into 3 portions: proximal, middle and distal ductus epididymis
- All portions of the male genital ducts, the epithelium showed the secretion of neutral glycoproteins and proteins without sulfated and carboxylated glycoproteins
GRAPHICAL ABSTRACT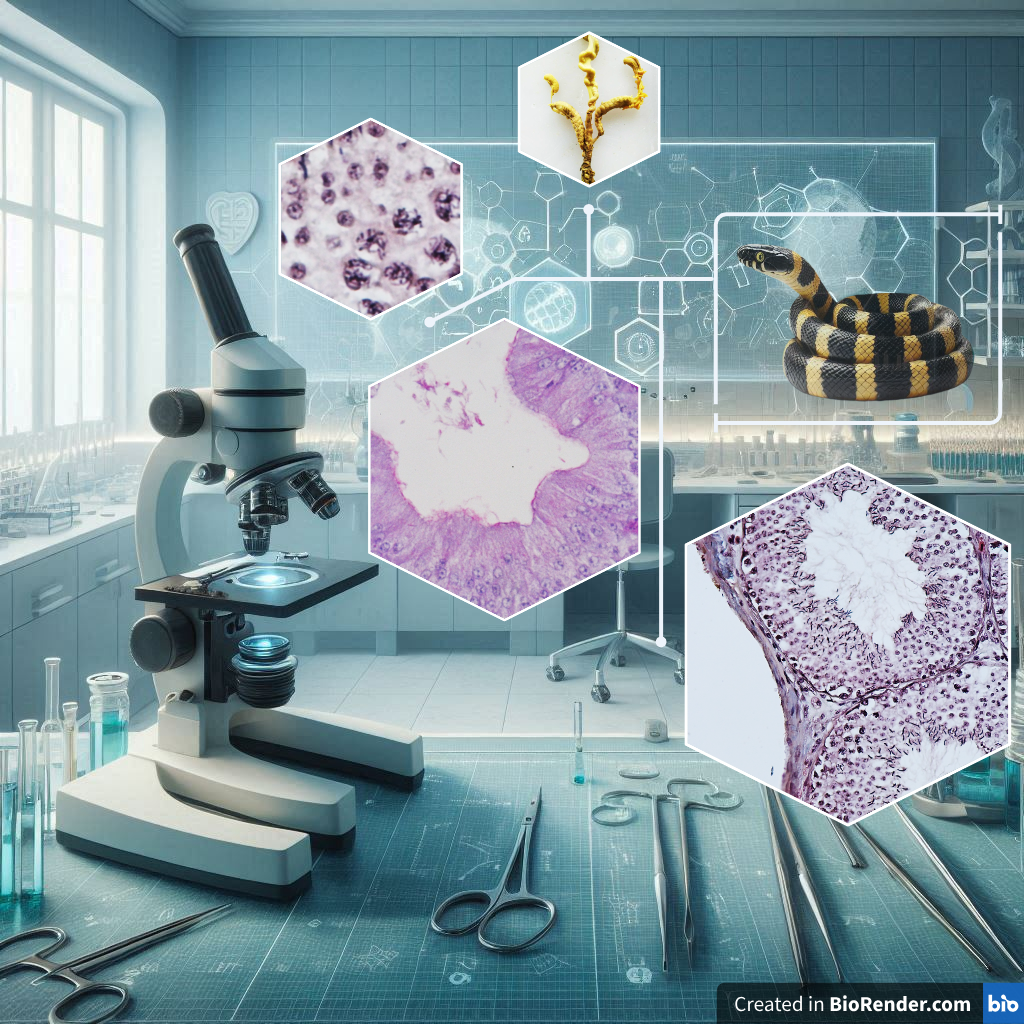
Downloads
References
LJ Vitt and JP Caldwell. Reproduction and life histories. In: LJ Vitt and JP Caldwell (Eds.). Herpetology: An introductory biology of amphibians and reptiles. 4th ed. Academic Press, San Diego, 2014, p. 117-55.
L Loebens, SZ Cechin, TF Theis, LB Moura and SM Almeida-Santos. Reproductive biology of Philodryas patagoniensis (Snakes: Dipsadidae) in South Brazil: Male reproductive cycle. Acta Zool. 2016; 98, 329-39.
T Vasaruchapong. Snake reproductive system. Thai Vet. Med. 2014; 41, 89-91.
P Tumkiratiwong, W Meesuk, L Chanhome and A Aowphol. Reproductive pattern of captive male and female monocled cobra, Naja kaouthia (Lesson, 1831). Zool. Stud. 2012; 51, 692-700.
SE Trauth and DM Sever. Male urogenital ducts and cloacal anatomy. In: RD Aldridge and DM Sever (Eds.). Reproductive biology and phylogeny of snakes. Science Publishers, New Hampshire, 2011, p. 411-75.
E Hondo, M Kurohmaru, M Toriba and Y Hayashi. Seasonal changes in spermatogenesis and ultrastructure of developing spermatids in the Japanese rat snake, Elaphe climacophora. J. Vet. Med. Sci. 1994; 56, 835-40.
KM Gribbins, JL Rheubert, MH Collier, DS Siegel and DM Sever. Histological analysis of spermatogenesis and the germ cell development strategy within the testis of the male western Cottonmouth Snake, Agkistrodon piscivorus. Ann. Anat. 2008; 190, 461-76.
E Hondo, N Kitamura, M Toriba, M Kurohmaru, Y Hayashi and J Yamada. Histological study of the seminiferous epithelium in the Japanese rat snake, Elaphe climacophora: Identification of spermatogonium. J. Vet. Med. Sci. 1997; 59, 23-9.
OA Al-Dokhi, YZ Al-Onazee and M Mubarak. Light and electron microscopy of the testicular tissue of the snake Eryx jayakari (Squamata, Reptilia) with a reference to the dividing germ cells. J. Biol. Sci. 2004; 4, 345-51.
DM Sever. Ultrastructure of the reproductive system of the black swamp snake (Seminatrix pygaea). VI. Anterior testicular ducts and their nomenclature. J. Morphol. 2010; 271, 104-15.
T Mathies, JA Cruz, VA Lance and JA Savidge. Reproductive biology of male brown tree snakes (Boiga irregularis) on Guam. J. Herpetol. 2010; 44, 209-21.
L Gang, L Qiao-Qiao, Y Hu-Hu and W Qiong-Xia. Histological and immunocytochemical study of deferens ducts in the Chinese rat snake (Zaocys dhumnades). Zool. Res. 2011; 32, 66-9.
DM Sever and LR Freeborn. Observations on the anterior testicular ducts in snakes with emphasis on sea snakes and ultrastructure in the Yellow-Bellied Sea Snake, Pelamis platurus. J. Morphol. 2012; 273, 324-36.
T Likhityungyuen, R Pimsorn, T Prompa, M Pongsuchart and P tumkiratiwong. Reproductive morphology of the golden tree snake Chrysopelea ornata (Serpentes: Colubridae) from Ao Nang, Krabi Province, Thailand. Sains Malays. 2022; 51, 1293-303.
HJ Bento, A Ferreira, GA Iglesias, FF Curcio, HAS Lima, TG Araújo, AH Kuczmarski and RCR Paz. Testicle histology of the Epicrates cenchria: A morphological and reproductive biology analysis. Arq. Bras. Med. Vet. Zootec. 2022; 74, 853-61.
L Chanhome, MJ Cox, T Vasaruchapong, N Chaiyabutr and V Sitprija. Characterization of venomous snakes of Thailand. Asian Biomed. 2011; 5, 311-28.
S Dutta, HP Jana, S Saha and SK Mukhopadhyay. The cause and consequences of road mortality of herpetofauna in Durgapur, West Bengal, India. Russ. J. Ecol. 2016; 47, 88-95.
TK Knierim. 2018, The spatial ecology, habitat use and activity of Banded Kraits (Bungarus fasciatus) in the Sakaerat biosphere reserve. Ph. D. Dissertation. Suranaree University of Technology, Nakhonratchasima, Thailand.
JD Bancroft and M Gamble. Theory and practice of histologial techniques. 6th ed. Churchill Livingstone, Edinburgh, Scotland, 2008.
SG Olukole, MO Oyeyemi and BO Oke. Biometrical and histometrical observations on the testis and epididymis of the African sideneck turtle (Pelusios castaneus). Eur. J. Anat. 2014; 18, 102-8.
KM Gribbins. Reptilian spermatogenesis: A histological and ultrastructural perspective. Spermatogenesis 2011; 1, 250-69.
M Mckinley, V O’Loughlin and T Bidle. Anatomy & physiology. McGraw-Hill, New York, 2013.
KM Gribbins and DH Gist. The cytological evaluation of the germinal epithelium and the germ cell cycle in an introduced population of European Wall Lizard, Podarics muralis. J. Morphol. 2003; 256, 296-306.
KM Gribbins and JL Rheubert. The ophidian testis, spermatogenesis and mature spermatozoa. In: RD Aldridge and DM Sever (Eds.). Reproductive biology and phylogeny of snakes. Science Publishers, New Hampshire, United States, 2011, p. 183-264.
A Pewhom, T Supapakorn and N Srakaew. Male reproductive cycle in a population of the endermic butterfly lizard, Leiolepis ocellata Peters, 1971 (Squamata: Agamidae) from northern Thailand. BMC Zool. 2022; 7, 45.
KM Gribbins, CS Happ and DM Sever. Ultrastructure of the reproductive system of the Black Swamp Snake (Seminatrix pygaea) V. The temporal germ cell development strategy of the testis. Acta Zool. 2005; 86, 223-30.
TA Aire. Anatomy of the testis and male reproductive tract. In: BBG Jamieson (Ed.). Reproductive biology and phylogeny of birds. Science Publishers, New Hampshire, United States, 2007, p. 37-113.
DS Siegel, DM Sever, JL Rheubert and KM Gribbins. Reproductive biology of Agkistrodon piscivorus Lace´pe`de (Squamata, Serpentes, Viperidae, Crotalinae). Herpetol. Monogr. 2009; 23, 74-107.
KY Ilio and RA Hess. Structure and function of the ductuli efferentes: A review. Microsc. Res. Tech. 1994; 29, 432-67.
L Hermo and B Robaire. Epididymal cell types and their functions. In: B Robaire and BT Hinton (Eds.). The epididymis: From molecules to clinical practice. Plenum Publishers, New York, 2002, p. 81-102.
S Desantis, M Labate, GM Labate and F Cirillo. Evidence of regional differences in the lectin histochemistry along the ductus epididymis of the lizard, Podarcis sicula Raf. Histochem. J. 2002; 34, 123-30.
MA Akbarsha, B Kadalmani and V Tamilarasan. Histological variation along and ultrastructural organization of the epithelium of the ductus epididymides of the fan-throated lizard Sitana ponticeriana Cuvier. Acta Zool. 2006; 87, 181-96.
DM Sever. Ultrastructure of the reproductive system of the black swamp snake (Seminatrix pygaea). IV. Occurrence of an ampulla ducuts deferentis. J. Morphol. 2004; 262, 714-20.
S Haider and U Rai. Epididymis of the Indian wall lizard (Hemidactylus flaviviridis) during the sexual cycle and in response to mammalian pituitary gonadotropins and testosterone. J. Morphol. 1987; 191, 151-60.
M Manimekalai and MA Akbarsha. Secretion of glycoprotein granules in the epididymis of the agamid lizard Calotes versicolor (Daudin) is region-specific. Biol. Struct. Morphog. 1992; 4, 96-101.
G Liang, Q Liu, H Yu and Q Wang. Histological and immunocytochemical study of deferens ducts in the Chinese rat snake (Zaocys dhumnades). Zool. Res. 2011; 32, 663-9.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






