Molecular Identification and Antifungal Activity of Candida chrysomelidarum and Rhodotorula toruloides Isolated from Indonesian Indigenous Fruit Flacourtia inermis Roxb. (Lobi-lobi) against Pathogenic Mold Aspergillus flavus
DOI:
https://doi.org/10.48048/tis.2024.8321Keywords:
Molecular identification, Antifungal activity, Candida chrysomelidarum, Rhodotorula toruloides, Flacourtia inermis fruit, Aspergillus flavusAbstract
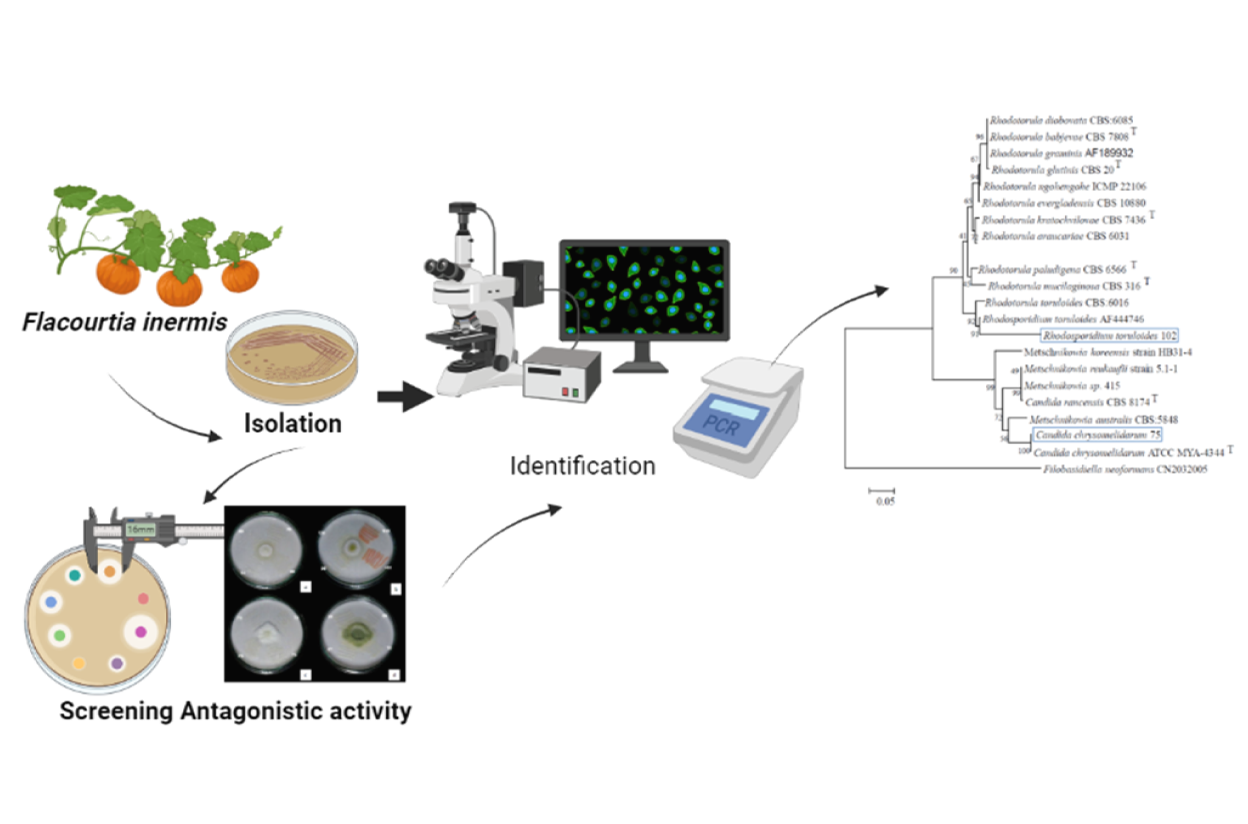
Aspergillus flavus is one of the main contaminating mold species in most feed products, significantly impacting human and animal health. This study aims to isolate the epiphytic yeasts from the fruit Flacourtia inermis Roxb and test their potential ability to act as biocontrol agents for A. flavus. The antagonistic test was performed using a dual culture method, and the potential yeast isolates were then identified using a combination of morphology characterization and molecular sequence analysis. From 42 yeast isolated from F. inermis fruit, 2 yeast isolates (LCA 75 and LCA 102) demonstrated the highest inhibition growth against A. flavus (32.32 and 32.30 %, respectively). The isolates LCA 75 and LCA 102 were identified as Candida chrysomelidarum and Rhodotorula toruloides, respectively. Rhodotorula toruloides, identified as one of the yeast isolates (LCA 102), is an unconventional red yeast demonstrating notable antagonistic effects against A. flavus, inhibiting its growth by 32.30 %. This yeast, also known as Rhodosporidium toruloides, R. rubescens, R. glutinis, or R. gracilis, belongs to the subphylum Pucciniomycotina within the division Basidiomycota. The significant inhibition of mycelium growth and sporulation observed in our study highlights the potential of R. toruloides, along with C. chrysomelidarum (LCA 75), as effective biocontrol agents to reduce mycotoxin contamination in chicken feed by A. flavus. This study represents a significant advancement in the management of A. flavus contamination in animal feed. The findings not only underscore the viability of yeast-based biocontrol strategies but also contribute to a safer, more sustainable approach to preventing mycotoxin proliferation in feedstocks.
HIGHLIGHTS
- Founding Out of 42 yeast isolates, two (LCA 75 and LCA 102) demonstrated the highest inhibition growth against A. flavus, with inhibition rates of 32.32 and 32.30 %, respectively.
- Identified Yeasts: LCA 75 as Candida chrysomelidarum LCA 102 as Rhodotorula toruloides (alternatively referred to as Rhodosporidium toruloides, R. rubescens, R. glutinis, or R. gracilis).
- Characteristics of Rhodotorula toruloides Unconventional red yeast belonging to the subphylum Pucciniomycotina within Basidiomycota. Commonly associated with various plant species and found on plant surfaces, including leaves, stems, and fruits.
- Identified yeast isolates exhibited inhibition activities leading to reduced mycelium growth and sporulation of A. flavus.
- These yeast isolates have potential as biocontrol agents to prevent mycotoxin contamination in chicken feed caused by A. flavus.
GRAPHICAL ABSTRACT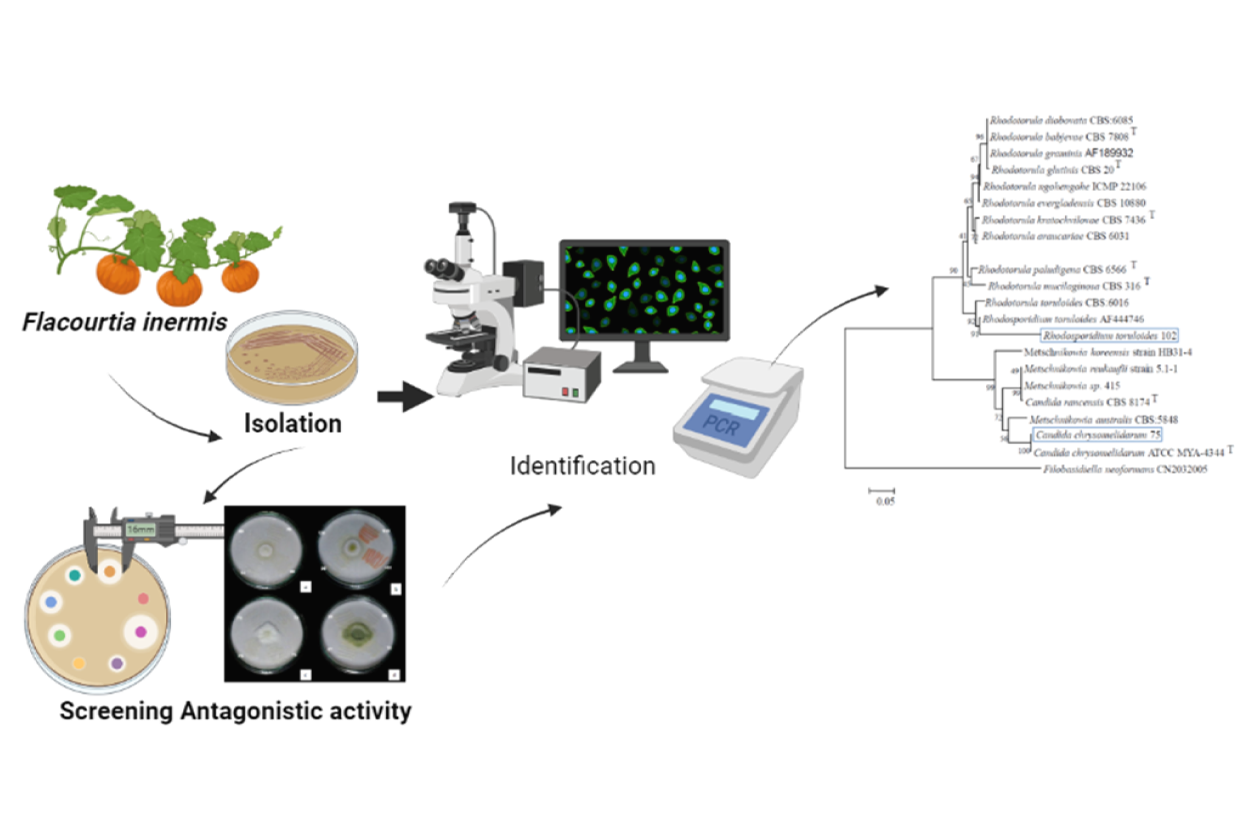
Downloads
References
IH Attitalla, LKT Al-Ani, MA Nasib, IAA Balal, M Zakaria, SSM El-Maraghy and SMR Karim. Screening of fungi associated with commercial grains and animal feeds in Al-Bayda governorate, Libya. World Appl. Sci. J. 2010; 9, 746-56.
B Tai, J Chang, Y Liu and F Xing. Recent progress of the effect of environmental factors on Aspergillus flavus growth and aflatoxins production on foods. Food Qual. Saf. 2020; 4, 21-8.
L Afsah-Hejri. Saprophytic yeasts: Effective biocontrol agents against Aspergillus flavus. Int. F. Res. J. 2013; 20, 3403-9.
Z Siddiqui, D Hagare, V Jayasena, R Swick, MM Rahman, N Boyle and M Ghodrat. Recycling of food waste to produce chicken feed and liquid fertiliser. Waste Manage. 2021; 131, 386-93.
I Podgórska-Kryszczuk. Biological control of Aspergillus flavus by the yeast Aureobasidium pullulans in vitro and on tomato fruit. Plants 2023; 12, 236.
K Yang, Z Zhang, F Zhang, F Song, H Zhong, F Ran, S Yu, G Xu, F Lan and S Wang. Inhibition of aflatoxin metabolism and growth of Aspergillus flavus in liquid culture by a DNA methylation inhibitor. Food Addit. Contam. 2014; 32, 554-63.
P Khlangwiset, GS Shephard and F Wu. Aflatoxins and growth impairment: A review. Crit. Rev. Toxicol. 2011; 41, 740-55.
P Bishnu, P De, A Hayat, D Roy, S Das and MK Mahato. Modern approaches in aflatoxin free groundnut production. In: RK Naresh (Ed.). Advances in agriculture sciences. AkiNik Publications, New Delhi, India, 2022.
X Lai, H Zhang, R Liu and C Liu. Potential for aflatoxin B1 and B2 production by Aspergillus flavus strains isolated from rice samples. Saudi J. Biol. Sci. 2015; 22, 176-80.
M Norlia, S Jinap, MAR Nor-Khaizura, S Radu, NIP Samsudin and FA Azri. Aspergillus section Flavi and aflatoxins: Occurrence, detection, and identification in raw peanuts and peanut-based products along the supply chain. Front. Microbiol. 2019; 10, 2602.
A Kumar, H Pathak, S Bhadauria and J Sudan. Aflatoxin contamination in food crops: Causes, detection, and management: A review. Food Prod. Process. Nutr. 2021; 3, 17.
MAH Rahman, J Selamat, NIP Samsudin, K Shaari, N Mahror and JM John. Antagonism of nonaflatoxigenic Aspergillus flavus isolated from peanuts against aflatoxigenic A. flavus growth and aflatoxin B1 production in vitro. Food Sci. Nutr. 2022; 10, 3993-4002.
C Payne and A Bruce. The yeast Debaryomyces hansenii as a short-term biological control agent against fungal spoilage of sawn Pinus sylvestris timber. Biol. Control 2001; 22, 22-8.
N Fardaila, D Sukmawati and TH Kurniyati. Potensi Khamir Asal Buah Lobi-Lobi (Flacourtia Inermis Roxb.) Dalam Menghambat Pertumbuhan Kapang Aspergillus Flavus. Skripsi, 2018.
D Sukmawati, A Setyaningsih, KT Handayani, S Rahayu, Y Rustam, M Moersilah and SNA Husna. Isolation and characterization of aflatoxigenic Aspergillus spp. from maize of livestock feed from Bogor. In: Proceedings of the 3rd Annual Applied Science and Engineering Conference, Bandung, Indonesia. 2018, p. 12105.
D Sukmawati, Z Arman, R Hasanah, M Balqis, A Setiawan, F Tafrijiyyah, R Sari, A Supiyani, NB Prihantini, SNA Husna, HAE Enshasy and DJ Dailin. Application of yeasts isolated from fermented cocoa beans for biocontrol of pathogenic mold in chocolate fruit. In: Proceedings of the 2nd Annual Conference of Science and Technology, Malang, Indonesia. 2020, p. 12042.
D Sukmawati, MH Andrianto, Z Arman, NI Ratnaningtyas, SNA Husna, HA El-Enshasy, D Dailin and AA Kenawy. Antagonistic activity of phylloplane yeasts from Moringa oleifera Lam. leaves against Aspergillus flavus UNJCC F-30 from chicken feed. Indian Phytopathol. 2020; 73, 79-88.
ML Penna, A Nesci and M Etcheverry. In vitro studies on the potential for biological control on Aspergillus section Flavi by Kluyveromyces spp. Lett. Appl. Microbiol. 2004; 38, 257-64.
D Sukmawati, Z Arman, GA Sondana, NN Fikriyah, R Hasanah, ZN Afifah, M Balqis, HE Enshasy, SNA Husna, S Rahayu, TH Kurniati and R Puspitaningrum. Potential amylase-producing yeast isolated from indigenous fermented beverages originating from Bali, Indonesia. J. Phys. Conf. Ser. 2019; 1402, 55021.
V Sharma, R Salwan and LKT Al-Ani. Molecular aspects of plant beneficial microbes in agriculture. Academic press, London, 2020.
L Aguilar-Marcelino, LKT Al-Ani, GS Castañeda-Ramirez, V Garcia-Rubio and JJ Ojeda-Carrasco. Microbial technologies to enhance crop production for future needs. In: AA Rastegari, AN Yadav and N Yadav (Eds.). New and future developments in microbial biotechnology and bioengineering. Elsevier, Amsterdam, The Netherlands, 2020, p. 29-47.
D Dellanerra, A Risandi, A Sunari, D Sukmawati, SNA Husna and HA El-Enshasy. Screening and characterization of amylolitic mold originated from ghost crab (Ocypode sp.) in Cidaon, Ujung Kulon National Park, Indonesia. AIP Conf. Pro. 2019; 2120, 070008.
L Ling, K Jiang, W Cheng, Y Wang, M Pang, H Luo, L Lu, K Gao and Y Tu. Biocontrol of volatile organic compounds obtained from Bacillus subtilis CL2 against Aspergillus flavus in peanuts during storage. Biol. Control 2022; 176, 105094.
AM Mohammed, LKT AL-Ani, L Bekbayeva and B Salleh. Biological control of Fusarium oxysporum f. sp. cubense by Pseudomonas fluorescens and BABA in vitro. World Appl. Sci. J. 2011; 15, 189-91.
J Kowalska, J Krzymińska and J Tyburski. Yeasts as a potential biological agent in plant disease protection and yield improvement - A short review. Agriculture 2022; 12, 1404.
CAT Gava, CA Pereira, PFDS Tavares and CDD Paz. Applying antagonist yeast strains to control mango decay caused by Lasiodiplodia theobromae and Neofusicoccum parvum. Biol. Control 2022; 170, 104912.
S Jaibangyang, R Nasanit and S Limtong. Biological control of aflatoxin-producing Aspergillus flavus by volatile organic compound-producing antagonistic yeasts. BioControl 2020; 65, 377-86.
MC Moure, RP Torrado, G Garmendia, S Vero, A Querol, T Alconada and ÁL Peláez. Characterization of kefir yeasts with antifungal capacity against Aspergillus species. Int. Microbiol. 2023; 26, 361-70.
TK Lim. Edible medicinal and non-medicinal plants: Volume 10, modified stems, roots, bulbs. Springer Dordrecht, Dordrecht, The Netherlands, 2016.
L Jayasinghe, M Lakdusinghe, N Hara and Y Fujimoto. Phenolic constituents from the fruit juice of Flacourtia inermis. Nat. Prod. Res. 2012; 26, 278-81.
AG Alakolanga, NS Kumar, L Jayasinghe and Y Fujimoto. Antioxidant property and α-glucosidase, α-amylase and lipase inhibiting activities of Flacourtia inermis fruits: Characterization of malic acid as an inhibitor of the enzymes. J. Food. Sci. Tech. 2015; 52, 8383-8.
PJ Benny, G Shibumon, K Sunny and G Cincy. 2, 3-dihydroxybenzoic acid: An effective antifungal agent isolated from Flacourtia inermis fruit. Int. J. Pharm. Clin. Res. 2010; 2, 101-5.
S George, PJ Benny, S Kuriakose and C George. Antibiotic activity of 2, 3-dihydroxybenzoic acid isolated from Flacourtia inermis fruit against multidrug resistant bacteria. Asian J. Pharm. Clin. Res. 2011; 4, 126-30.
D Sukmawati, A Oetari, D Hendrayanti, M Atria and W Sjamsuridzal. Identification of phylloplane yeasts from paper mulberry (Broussonetia papyrifera (L.) L’Hér. ex Vent.) in Java, Indonesia. Malays. J. Microbiol. 2015; 11, 324-40.
A Chanchaichaovivat, P Ruenwongsa and B Panijpan. Screening and identification of yeast strains from fruits and vegetables: Potential for biological control of postharvest chilli anthracnose (Colletotrichum capsici). Biol. Control 2007; 42, 326-35.
JG Cappuccino and CT Welsh. Microbiology: A laboratory manual. Pearson, Essex, 2018.
EJ Rangel-Muñoz, AG Valdivia-Flores, S Hernández-Delgado, C Cruz-Vázquez, MC De-Luna-López, T Quezada-Tristán, R Ortiz-Martínez and N Mayek-Pérez. Assessment of the potential of a native non-aflatoxigenic Aspergillus flavus isolate to reduce aflatoxin contamination in dairy feed. Toxins 2022; 14, 437.
MH El-Katatny, MS El-Katatny, EM Fadl-Allah and AS Emam. Antagonistic effect of two isolates of Trichoderma harzianum against postharvest pathogens of tomato (Lycopersicon esculentum). Arch. Phytopathol. Plant Prot. 2011; 44, 637-54.
CP Kurtzman, JW Fell and T Boekhout. The yeasts: A taxonomic study. Elsevier, Amsterdam, The Netherlands, 2011.
I Maulana. Identifikasi isolat-isolat khamir dari saluran pencernaan apis cerana (Fabricius, 1793) di apiari berdasarkan data sequence daerah ITS rDNA (in Indonesian). Universitas Indonesia, Depok, Indonesia, 2011.
K Tamura, G Stecher and S Kumar. MEGA11: Molecular evolutionary genetics analysis version 11. Mol. Biol. Evol. 2021; 38, 3022-7.
LC Mata-Gómez, JC Montañez, A Méndez-Zavala and CN Aguilar. Biotechnological production of carotenoids by yeasts: An overview. Microb. Cell Fact. 2014; 13, 12.
MG Kreusch and RTD Duarte. Photoprotective compounds and radioresistance in pigmented and non-pigmented yeasts. Appl. Microbiol. Biotechnol. 2021; 105, 3521-32.
ABM Vaz, LH Rosa, MLA Vieira, VD Garcia, LR Brandão, LCR Teixeira, M Moliné, D Libkind, MV Broock and CA Rosa. The diversity, extracellular enzymatic activities and photoprotective compounds of yeasts isolated in Antarctica. Braz. J. Microbiol. 2011; 42, 937-47.
DJ Dailin, EA Elsayed, RA Malek, SZ Hanapi, S Selvamani, S Ramli, D Sukmawati, RZ Sayyed and HAE Enshasy. Efficient kefiran production by Lactobacillus kefiranofaciens ATCC 43761 in submerged cultivation: Influence of osmotic stress and nonionic surfactants, and potential bioactivities. Arabian J. Chem. 2020; 13, 8513-23.
LK Nyanga, MJR Nout, TH Gadaga, B Theelen, T Boekhout and MH Zwietering. Yeasts and lactic acid bacteria microbiota from masau (Ziziphus mauritiana) fruits and their fermented fruit pulp in Zimbabwe. Int. J. Food Microbiol. 2007; 120, 159-66.
M Lentz, T Putzke, R Hessler and E Luman. Genetic and physiological characterization of yeast isolated from ripe fruit and analysis of fermentation and brewing potential. J. Inst. Brew. 2014; 120, 559-64.
L Dufossé. Current and potential natural pigments from microorganisms (bacteria, yeasts, fungi, and microalgae). In: Handbook on natural pigments in food and beverages. Woodhead Publishing, Cambridge, 2016, p. 337-54.
LADC Cardoso, KYF Kanno and SG Karp. Microbial production of carotenoids: A review. Afr. J. Biotechnol. 2017; 16, 139-46.
M Moliné, MR Flores, D Libkind, MDC Diéguez, ME Farías and MV Broock. Photoprotection by carotenoid pigments in the yeast Rhodotorula mucilaginosa: The role of torularhodin. Photochem. Photobiol. Sci. 2010; 9, 1145-51.
MHS Rani, T Ramesh, J Subramanian and M Kalaiselvam. Production and characterization of melanin pigment from halophilic black yeast Hortaea werneckii. Int. J. Pharma Res. Rev. 2013; 2, 9-17.
R Vadkertiová, J Molnárová, D Vránová and E Sláviková. Yeasts and yeast-like organisms associated with fruits and blossoms of different fruit trees. Can. J. Microbiol. 2012; 58, 1344-52.
SB Swami, NJ Thakor, PM Haldankar and SB Kalse. Jackfruit and its many functional components as related to human health: A review. Compr. Rev. Food Sci. Food Saf. 2012; 11, 565-76.
V Ambikapathy, S Gomathi and A Panneerselvam. Effect of antifungal activity of some medicinal plants against Pythium debaryanum (Hesse). Asian J. Plant Sci. Res. 2011; 1, 131-4.
M Bar-Shimon, H Yehuda, L Cohen, B Weiss, A Kobeshnikov, A Daus, M Goldway, M Wisniewski and S Droby. Characterization of extracellular lytic enzymes produced by the yeast biocontrol agent Candida oleophila. Curr. Genet. 2004; 45, 140-8.
MW Haggag and HALA Mohamed. Biotechnological aspects of microorganisms used in plant biological control. Eurasian J. Sustainable Agr. 2007; 1, 7-12.
LKT Al-Ani, Surono, L Aguilar-Marcelino, VE Salazar-Vidal, AG Becerra and W Raza. Role of useful fungi in agriculture sustainability. In: AN Yadav (Ed.). Recent trends in mycological research: Volume 1: Agricultural and medical perspective. Springer, Cham, Switzerland, 2021, p. 1-44.
L Aguilar-Marcelino, P Mendoza-De-Gives, LKT Al-Ani, ME López-Arellano, O Gómez-Rodríguez, E Villar-Luna and DE Reyes-Guerrero. Using molecular techniques applied to beneficial microorganisms as biotechnological tools for controlling agricultural plant pathogens and pest. In: V Sharma, R Salwan and LKT Al-Ani (Eds.). Molecular aspects of plant beneficial microbes in agriculture. Academic Press, London, 2020, p. 333-49.
EI Masih and B Paul. Secretion of β-1,3-glucanases by the yeast Pichia membranifaciens and its possible role in the biocontrol of Botrytis cinerea causing grey mold disease of the grapevine. Curr. Microbiol. 2002; 44, 391-5.
A Santos and D Marquina. Killer toxin of Pichia membranifaciens and its possible use as a biocontrol agent against grey mould disease of grapevine. Microbiology 2004; 150, 2527-34.
PJ Boynton. The ecology of killer yeasts: Interference competition in natural habitats. Yeast 2019; 36, 473-85.
HP Dhiani. Kemampuan antagonisme khamir filum Ascomycota dari tanaman saeh (Broussonetia papyrifera Vent.) asal Trowulan terhadap Aspergilus spp. UICC (in Indonesian). Universitas Indonesia, Depok, Indonesia, 2012.
F Bordet, A Joran, G Klein, C Roullier-Gall and H Alexandre. Yeast-yeast interactions: Mechanisms, methodologies and impact on composition. Microorganisms 2020; 8, 600.
M Cortés-Higareda, MDL Ramos-García, ZN Correa-Pacheco, JCD Río-García and S Bautista-Baños. Nanostructured chitosan/propolis formulations: Characterization and effect on the growth of Aspergillus flavus and production of aflatoxins. Heliyon 2019; 5, e01776.
B Li, H Peng and S Tian. Attachment capability of antagonistic yeast Rhodotorula glutinis to Botrytis cinerea contributes to biocontrol efficacy. Front. Microbiol. 2016; 7, 601.
SB Mekbib, TJC Regnier and L Korsten. Efficacy and mode of action of yeast antagonists for control of Penicillium digitatum in oranges. 2011; 36, 233-40.
AC Ritter, M Hoeltz and IB Noll. Toxigenic potential of Aspergillus flavus tested in different culture conditions. Food Sci. Tech. 2011; 31, 623-8.
PB Ouattara-Sourabie, PA Nikiema, N Barro, A Savadogo and AS Traore. Aflatoxigenic potential of Aspergillus spp. isolated from groundnut seeds, in Burkina Faso, West Africa. Afr. J. Microbiol. Res. 2012; 6, 2603-9.
M DebMandal and S Mandal. Coconut (Cocos nucifera L.: Arecaceae): In health promotion and disease prevention. Asian Pac. J. Trop. Med. 2011; 4, 241-7.
J Xu. Fungal species concepts in the genomics era. Genome 2020; 63, 459-68.
AR Hafsari, A Oetari, A Salamah and W Sjamsuridzal. Pengujian kemampuan antagonistik khamir Rhodotorula spp. Asal kebun raya cibodas terhadap kapang dari tanaman tomat terinfeksi dengan co culture (in Indonesian). Jurnal Istek 2011; 5, 149-60.
K Hayer, M Stratford and DB Archer. Structural features of sugars that trigger or support conidial germination in the filamentous fungus Aspergillus niger. Appl. Environ. Microbiol. 2013; 79, 6924-31.
N Osherov and G May. Conidial germination in Aspergillus nidulans requires RAS signaling and protein synthesis. Genetics 2000; 155, 647-56.
J Gholamnejad, HR Etebarian and N Sahebani. Biological control of apple blue mold with Candida membranifaciens and Rhodotorula mucilaginosa. Afr. J. Food Sci. 2010; 4, 1-7.
LKT Al-Ani. 2017, Potential of utilizing biological and chemical agents in the control of Fusarium wilt of banana. Ph.D. Dissertation. Universiti Sains Malaysia, Pulau Pinang, Malaysia.
LKT Al-Ani, ES Negim, B Salleh, AM Mohammed and MI Saleh. Utilization of amino azines polymers as antifungal activity for banana. In: Proceedings of the 1st USM-KAZNU International Conference on Challenges of Teaching and Chemistry Research in Institutions of Higher Learning, Pulau Pinang, Malaysia. 2012, p. 29.
GH Jatoi, S Muhammad, WA Metlo, LKT Al-Ani, Haseenullah, MA Abro, MA Gadhi, NW Awan and MA Reki. Efficacy of different essential oils, fungicides and biocontrol agents against Aspergillus niger the causal agent of fruit rot in Pomegranate. Int. J. Biosci. 2020; 16, 51-65.
L Parafati, C Restuccia and G Cirvilleri. Efficacy and mechanism of action of food isolated yeasts in the control of Aspergillus flavus growth on pistachio nuts. Food Microbiol. 2022; 108, 104100.
SA James and M Stratford. Spoilage yeasts with special emphasis on the genus Zygosaccharomyces. In: T Boekhout and V Robert (Eds.). Yeasts in food. Woodhead Publishing, Cambridge, 2003, p. 171-91.
Ł Paśko, PGP Ericson and A Elzanowski. Phylogenetic utility and evolution of indels: A study in neognathous birds. Mol. Phylogenet. Evol. 2011; 61, 760-71.
NN Wijayawardene, KD Hyde, LKT Al-Ani, L Tedersoo, D Haelewaters, KC Rajeshkumar, RL Zhao, A Aptroot, D Leontyev, RK Saxena, YS Tokarev, DQ Dai, PM Letcher, SL Stephenson, D Ertz, HT Lumbsch, M Kukwa, I Issi, H Madrid and M Thines. Outline of fungi and fungus-like taxa. Mycosphere 2020; 11, 1060-456.
M Raza, P Razaghi, LKT Al-Ani and L Aguilar-Marcelino. Underpinning the phylogeny and taxonomy of phytofungi through computational biology. In: Phytomycology and molecular biology of plant pathogen interactions. CRC Press, Florida, 2022, p. 149-69.
F Ekawasti, U Cahyaningsih, NLPI Dharmayanti, S Sa’diah, DT Subekti, Z Azmi and MI Desem. Restriction fragment length polymorphism analysis of genes of virulent strain isolate of Toxoplasma gondii using enzyme DdeI. Int. J. One Health 2021; 7, 196-203.
MG Simpson. Plant systematics. Academic Press, Massachusetts, 2019.
SK Biswas, K Yokoyama, K Nishimura and M Miyaji. Molecular phylogenetics of the genus Rhodotorula and related basidiomycetous yeasts inferred from the mitochondrial cytochrome b gene. Int. J. System. Evol. Microbiol. 2001; 51, 1191-9.
JL Staton. Understanding phylogenies: Constructing and interpreting phylogenetic trees. J. S. Carol. Acad. Sci. 2015; 13, 6.
NH Nguyen, SO Suh, CK Erbil and M Blackwell. Metschnikowia noctiluminum sp. nov., Metschnikowia corniflorae sp. nov., and Candida chrysomelidarum sp. nov., isolated from green lacewings and beetles. Mycol. Res. 2006; 110, 346-56.
MK Dhami, T Hartwig, AD Letten, M Banf and T Fukami. Genomic diversity of a nectar yeast clusters into metabolically, but not geographically, distinct lineages. Mol. Ecol. 2018; 27, 2067-76.
JA Barnett, RW Payne, D Yarrow and L Barnett. Yeasts: Characteristics and identification. 3rd eds. Cambridge University Press, Cambridge, 2000.
M Garcia‐Gonzalez, FJ Plou, FV Cervantes, M Remacha, A Poveda, J Jiménez‐Barbero and M Fernandez‐Lobato. Efficient production of isomelezitose by a glucosyltransferase activity in Metschnikowia reukaufii cell extracts. Microb. Biotechnol. 2019; 12, 1274-85.
CC Rering, JJ Beck, GW Hall, MM McCartney and RL Vannette. Nectar‐inhabiting microorganisms influence nectar volatile composition and attractiveness to a generalist pollinator. New Phytol. 2018; 220, 750-9.
AB Wróbel, P Farkaš, P Chraniuk, D Popielarz, A Synowiec, K Pobiega and M Janowicz. Antimicrobial and prebiotic activity of mannoproteins isolated from conventional and nonconventional yeast species - The study on selected microorganisms. World J. Microbiol. Biotechnol. 2022; 38, 256.
QM Wang, AM Yurkov, M Göker, HT Lumbsch, SD Leavitt, M Groenewald, B Theelen, XZ Liu, T Boekhout and FY Bai. Phylogenetic classification of yeasts and related taxa within Pucciniomycotina. Stud. Mycol. 2015; 81, 149-89.
J Goordial, A Davila, D Lacelle, W Pollard, MM Marinova, CW Greer, J CiRuggiero, CP McKay and LG Whyte. Nearing the cold-arid limits of microbial life in permafrost of an upper dry valley, Antarctica. ISME J. 2016; 10, 1613-24.
S Fenina. Kemampuan antagonisme khamir filum Basidiomycota dari tanaman saeh (Broussonetia papyfera Vent.) asal Trowulan terhadap Aspergilus spp. UICC (in Indonesian). Universitas Indonesia, Depok, Indonesia, 2012.
Z Wen, S Zhang, CK Odoh, M Jin and ZK Zhao. Rhodosporidium toruloides - A potential red yeast chassis for lipids and beyond. FEMS Yeast Res. 2020; 20, foaa038.
CC Wu, K Honda and F Kazuhito. Current advances in alteration of fatty acid profile in Rhodotorula toruloides: A mini-review. World J. Microbiol. Biotechnol. 2023; 39, 234.
Y Zhao, B Song, J Li and J Zhang. Rhodotorula toruloides: An ideal microbial cell factory to produce oleochemicals, carotenoids, and other products. World J. Microbiol. Biotechnol. 2022; 38, 13.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






