Current Progress in Exploring Structural Changes in Brown Algae Fucoxanthin and Its Potential Bioactivity for Human Health
DOI:
https://doi.org/10.48048/tis.2024.7984Keywords:
Bioactivity, Brown seaweed, Cis-trans isomers, Fucoxanthin, Stereomutation, Structure changesAbstract
Fucoxanthin is a carotenoid found in brown seaweed. Its potential anti-cancer, anti-obesity, anti-inflammatory and antioxidant properties have recently attracted interest. This interest is driven by the growing awareness of natural compounds promoting well-being, ongoing research into their health impacts and their appeal to the food and supplement industries. Brown seaweed is a major source of fucoxanthin and its global cultivation is on the rise. Fucoxanthin’s structural characteristics contribute to its bioactivity. Isomerization, influenced by factors like light and heat, can alter its biological activity. Recent studies highlight the importance of the cis/trans isomer ratio in determining fucoxanthin’s biological effects. This review uses narrative approaches to explore fucoxanthin’s potential applications in the food and pharmaceutical industries, emphasizing its source from brown seaweed and considerations for maximizing bioavailability and stability. The dynamic landscape of fucoxanthin research underscores its promising role in human health, encapsulating its multifaceted benefits within a concise framework.
HIGHLIGHTS
- Fucoxanthin from brown seaweed has potential anti-cancer, anti-obesity, anti-inflammatory, and antioxidant properties
- Interest in fucoxanthin is rising due to its natural origin and health benefits.
- The global cultivation of brown seaweed is increasing, driven by demand for fucoxanthin
- Fucoxanthin’s bioactivity is influenced by its structure and isomer ratios
- Stability and extraction methods are crucial for optimizing fucoxanthin’s use in food and pharmaceuticals
GRAPHICAL ABSTRACT

Downloads
References
AM Pyszniak and SP Gibbs. Immunocytochemical localization of photosystem I and the fucoxanthin-chlorophyll a/c fight-harvesting complex in the diatom Phaeodactylum tricornutum, 1992; 166, 208-17.
J Peng, JP Yuan, CF Wu and JH Wang. Fucoxanthin, a marine carotenoid present in brown seaweeds and diatoms: Metabolism and bioactivities relevant to human health. Mar. Drugs 2011; 9, 1806-28.
Y Li, Y Liu, Y Wang, Y Yu, Y Zeng, L Li and L Wang. The bioactivity of fucoxanthin from Undaria pinnatifida in vitro. Am. J. Biochem. Biotechnol. 2016; 12, 139-48.
JM Lorenzo, R Agregán, PES Munekata, D Franco, J Carballo, S Şahin, R Lacomba and FJ Barba. Proximate composition and nutritional value of three macroalgae: Ascophyllum nodosum, Fucus vesiculosus, and Bifurcaria bifurcate. Mar. Drugs 2017; 15, 360.
Centre D’etude and De Valorisation Des Algues. Edible seaweed and microalgae Regulatory status in France and Europe, Available at: https://www.ceva-algues.com/wp-content/uploads/2020/03/CEVA-Edible-algae-FR-and-EU-regulatory-update-2019.pdf, accessed December 2023.
M Terasaki, A Hirose, B Narayan, Y Baba, C Kawagoe, H Yasui, N Saga, M Hosokawa and K Miyashita. Evaluation of recoverable functional lipid components of several brown seaweeds (phaeophyta) from Japan with special reference to fucoxanthin and fucosterol contents1. J. Phycol. 2009; 45, 974-80.
E Shannon and N Abu-Ghannam. Optimization of fucoxanthin extraction from Irish seaweeds by response surface methodology. J. Appl. Phycol. 2017; 29, 1027-36.
C Lourenço-Lopes, M Fraga-Corral, P Garcia-Perez, A Carreira-Casais, A Silva, J Simal-Gandara and MA Prieto. An HPLC‐DAD method for identifying and estimating the content of fucoxanthin, β‐carotene, and chlorophyll in brown algal extracts. Food Chem. Adv. 2022; 1, 100095.
T Mise, M Ueda and T Yasumoto. Production of fucoxanthin-rich powder from Cladosiphon okamuranus. Adv. J. Food Sci. Technol. 2011; 3, 73-6.
P Verma, M Kumar, G Mishra and D Sahoo. Multivariate analysis of fatty acid and biochemical constituents of seaweeds to characterize their potential as a bioresource for biofuel and fine chemicals. Bioresour. Technol. 2017; 226, 132-44.
SJ Heo, WJ Yoon, KN Kim, GN Ahn, SM Kang, DH Kang, A affan, C Oh, WK Jung and YJ Jeon. Evaluation of the anti-inflammatory effect of fucoxanthin isolated from brown algae in lipopolysaccharide-stimulated RAW 264.7 macrophages. Food Chem. Toxicol. 2010; 48, 2045-51.
Y Heriyanto, AD Shioi, Juliadiningtyas and L Limantara. Analysis of pigment composition of brown seaweeds collected from Panjang Island, Central Java, Indonesia. Philipp J. Sci. 2017; 146, 323-30.
E Susanto, AS Fahmi, M Abe, M Hosokawa and K Miyashita. Lipids, fatty acids, and fucoxanthin content from temperate and tropical brown seaweeds. Aquat. Procedia 2016; 7, 66-75.
N Heffernan, TJ Smyth, RJ FitzGerald, A Vila-Soler, J Mendiola, E Ibáñez and NP Brunton. Comparison of extraction methods for selected carotenoids from macroalgae and the assessment of their seasonal/spatial variation. Innov. Food Sci. Emerg. Technol. 2016; 37, 221-8.
G Rajauria, B Foley and N Abu-Ghannam. Characterization of dietary fucoxanthin from Himanthalia elongata brown seaweed. Food Res. Int. 2017; 99, 995-1001.
SC Foo, FM Yusoff, M Ismail, M Basri, SK Yau, NMH Khong, KW Chan and M Ebrahimi. Antioxidant capacities of fucoxanthin-producing algae as influenced by their carotenoid and phenolic contents. J. Biotechnol. 2017; 241, 175-83.
K Kanazawa, Y Ozaki, T Hashimoto, SK Das, S Matsushita, M Hirano, T Okada, A Komoto, N Mori and M Nakatsuka. Commercial-scale preparation of biofunctional fucoxanthin from waste parts of brown sea algae Laminalia japonica. Food Sci. Technol. Res. 2008; 14, 573-82.
K Mori, T Ooi, M Hiraoka, N Oka, H Hamada, M Tamura and T Kusumi. Fucoxanthin and its metabolites in edible brown algae cultivated in deep seawater. Mar. Drugs 2004; 2, 63-72.
GR Seely, MJ Duccan and WE Vidiver. Preparative and analytical extraction of pigments from brown algae with dimethyl sulfoxide. Mar. Biol. 1972; 12, 184-8.
V Raguraman, SAL, D MubarakAli, G Narendrakumar, R Thirugnanasambandam, R Kirubagaran and N Thajuddin. Unraveling rapid extraction of fucoxanthin from Padina tetrastromatica: Purification, characterization, and biomedical application. Process Biochem. 2018; 73, 211-9.
S Agatonovic-Kustrin and DW Morton. High-performance thin-layer chromatography HPTLC-direct bioautography as a method of choice for alpha-amylase and antioxidant activity evaluation in marine algae. J. Chromatogr. A 2017; 1530, 197-203.
I Jaswir, D Noviendri, HM Salleh and K Miyashita. Fucoxanthin extractions of brown seaweeds and analysis of their lipid fraction in methanol. Food Sci. Technol. Res. 2012; 18, 251-7.
J Li, Y Liu, Y Liu, Q Wang, X Gao and Q Gong. Effects of temperature and salinity on the growth and biochemical composition of the brown alga Sargassum fusiforme (Fucales, Phaeophyceae). J. Appl. Phycol. 2019; 31, 3061-8.
H Kanda, Y Kamo, S Machmudah, Wahyudiono and M Goto. Extraction of fucoxanthin from raw macroalgae excluding drying and cell wall disruption by liquefied dimethyl ether. Mar. Drugs 2014; 12, 2383-96.
F Sultana, MA Wahab, M Nahiduzzaman, M Mohiuddin, MZ Iqbal, A Shakil, AA Mamun, MSR Khan, LL Wong and M Asaduzzaman. Seaweed farming for food and nutritional security, climate change mitigation and adaptation, and women empowerment: A review. Aquac. Fish. 2023; 8, 463-80.
J Cai, A Lovatelli, GE Gammaro, J Geehan, D Lucente, G Mair, W Miao, M Reantaso, R Roubach, X Yuan, J Aguilar-Manjarrez, L Cornish, L Dabbadie, A Desrochers, S Diffey, M Tauati, A Hurtado, P Potin and C Przybyla. Seaweed and microalgae: an overview for unlocking their potential in global aquaculture development. FAO Fisheries and Aquaculture Circular Rome, Rome, Italy, 2021; 1229, p. 48.
D Desideri, C Cantaluppi, F Ceccotto, MA Meli, C Roselli and L Feduzi. Essential and toxic elements in seaweeds for human consumption. J. Toxicol. Environ. Health A 2016; 79, 112-22.
J Feldmann and EM Krupp. Critical review or scientific opinion paper: Arsenosugars-a class of benign arsenic species or justification for developing partly speciated arsenic fractionation in foodstuffs? Anal. Bioanal. Chem. 2011; 399, 1735-41.
JM Ronan, DB Stengel, A Raab, J Feldmann, L O’Hea, E Bralatei and E McGovern. High proportions of inorganic arsenic in Laminaria digitata but not in Ascophyllum nodosum samples from Ireland. Chemosphere 2017; 186, 17-23.
T Davydiuk, J Tao, X Lu and XC Le. Effects of dietary intake of arsenosugars and other organic arsenic species on studies of arsenic methylation efficiency in humans. Environ. Health 2023; 1, 236-48.
MV Hulle, C Zhang, B Schotte, L Mees, F Vanhaecke, R Vanholder, XR Zhang and R Cornelis. Identification of some arsenic species in human urine and blood after ingestion of Chinese seaweed Laminaria. J. Anal. At. Spectrom. 2004; 19, 58-64.
JTV Elteren, Z Šlejkovec, M Kahn and W Goessler. A systematic study on the extractability of arsenic species from algal certified reference material IAEA-140/TM (Fucus sp., sea plant homogenate) using methanol/water extractant mixtures. Anal. Chim. Acta 2007; 585, 24-31.
A Morales-Rodríguez, M Pérez-López, E Puigpelat, À Sahuquillo, D Barrón and JF López-Sánchez. Arsenosugar extracted from algae: Isolation by anionic exchange solid‐phase extraction. J. Chromatogr. A 2022; 1684, 463549.
K Gokulan, A Mathur, A Kumar, MM Vanlandingham and S Khare. Route of arsenic exposure differentially impacts the expression of genes involved in gut-mucosa-associated immune responses and gastrointestinal permeability. Int. J. Mol. Sci. 2023; 24, 6352.
P Andrewes, DM Demarini, K Funasaka, K Wallace, VWM Lai, H Sun, WR Cullen and KT Kitchin. Do arsenosugars pose a risk to human health? The comparative toxicities of a trivalent and pentavalent arsenosugar. Environ. Sci. Technol. 2004; 38, 4140-8.
VF Taylor and BP Jackson. Concentrations and speciation of arsenic in New England seaweed species harvested for food and agriculture. Chemosphere 2016; 163, 6-13.
Y Zhao, D Shang, J Ning and Y Zhai. Arsenic and cadmium in the marine macroalgae (Porphyra yezoensis and Laminaria japonica)-forms and concentrations. Chem. Speciat. Bioavailab. 2012; 24, 197-203.
The French Food Safety Agency (AFSSA). Opinion1 of the French food safety agency on the recommended maximum inorganic arsenic content of laminaria and consumption of these seaweeds in light of their high iodine content. The French Food Safety Agency (AFSSA), Maisons-Alfort, France, 2009, p. 20.
C Peter, W Jackie, G Abhishek, H Beverley, W Tom, H Hanna, H Tim, L Janet, N Jeane, T Kate and E Christine. Evaluation of food safety risks associated with seaweed and seaweed products. New Zealand Food Safety Science & Research Centre, Palmerston North, New Zealand, 2023, p. 192.
Y Yamashita. Method of removing inorganic arsenic from dried hijiki seaweed products. Nippon Suisan Gakk. 2014; 80, 973-8.
GY Park, DE Kang, M Davaatseren, C Shin, GJ Kang and MS Chung. Reduction of total, organic, and inorganic arsenic content in Hizikia fusiforme (Hijiki). Food Sci. Biotechnol. 2019; 28, 615-22.
L Wang, YR Cui, S Oh, MJ Paik, JG Je, JH Heo, TK Lee, X Fu, J Xu, X Gao and YJ Jeon. Arsenic removal from the popular edible seaweed Sargassum fusiforme by sequential processing involving hot water, citric acid, and fermentation. Chemosphere 2022; 292, 133409.
D Zhao, SM Kim, CH Pan and D Chung. Effects of heating, aerial exposure, and illumination on the stability of fucoxanthin in canola oil. Food Chem. 2014; 145, 505-13.
D Zhao, D Yu, M Kim, MY Gu, SM Kim, CH Pan, GH Kim and D Chung. Effects of temperature, light, and pH on the stability of fucoxanthin in an oil-in-water emulsion. Food Chem. 2019; 291, 87-93.
JA Haugan and S Liaaen-Jensen. Naturally occurring stereoisomers of fucoxanthin. Phytochemistry 1992; 31, 1359-61.
M Honda, K Murakami, S Takasu and M Goto. Extraction of fucoxanthin isomers from the edible brown seaweed Undaria pinnatifida using supercritical CO2: Effects of extraction conditions on isomerization and recovery of fucoxanthin. J. Oleo Sci. 2022; 71, 1097-106.
AA Wibowo, PE Elim, Heriyanto, MNU Prihastyanti, JR Yoewono, Y Shioi, L Limantara and THP Brotosudarmo. Effect of drying on the production of fucoxanthin isomers from brown seaweeds. J. Food Process. Preserv. 2022; 46, 1-8.
AA Wibowo, Heriyanto, Y Shioi, L Limantara and THP Brotosudarmo. Simultaneous purification of fucoxanthin isomers from brown seaweeds by open-column and high-performance liquid chromatography. J. Chrom. B 2022; 1193, 123166.
P Crupi, AT Toci, S Mangini, F Wrubl, L Rodolfi, MR Tredici, A Coletta and D Antonacci. Determination of fucoxanthin isomers in microalgae (Isochrysis sp.) by high-performance liquid chromatography coupled with diode-array detector multistage mass spectrometry coupled with positive electrospray ionization. Rapid Comm. Mass Spectrom. 2013; 27, 1027-35.
Y Nakazawa, T Sashima, M Hosokawa and K Miyashita. Comparative evaluation of growth inhibitory effect of stereoisomers of fucoxanthin in human cancer cell lines. J. Funct. Foods 2009; 1, 88-97.
S Tachihana, N Nagao, T Katayama, M Hirahara, FM Yusoff, S Banerjee, M Shariff, N Kurosawa, T Toda and K Furuya. High productivity of eicosapentaenoic acid and fucoxanthin by a marine diatom Chaetoceros gracilis in a semi-continuous culture. Front. Bioeng. Biotechnol. 2020; 8, 602721.
RGD Oliveira-Júnior, R Grougnet, PE Bodet, A Bonnet, E Nicolau, A Jebali, J Rumin and L Picot. Updated pigment composition of Tisochrysis lutea and purification of fucoxanthin using centrifugal partition chromatography coupled to flash chromatography for the chemosensitization of melanoma cells. Algal Res. 2020; 51, 102035.
V Rajendran, YS Pu and BH Chen. An improved HPLC method for determination of carotenoids in human serum. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2005; 824, 99-106.
LC Sander, KE Sharpless and M Pursch. C30 stationary phases for the analysis of food by liquid chromatography. J. Chromatogr. A 2000; 880, 189-202.
JS Palmer, LA Lawton, R Kindt and C Edwards. Rapid analytical methods for the microalgal and cyanobacterial biorefinery: Application on strains of industrial importance. Microbiologyopen 2021; 10, e1156.
LC Sander, KE Sharpless, NE Craft and SA Wise. Development of engineered stationary phases for the separation of carotenoid isomers. Anal. Chem. 1994; 66, 1667-74.
JA Haugan, G Englert, E Glinz, S Liaaen-Jensen, M Vuoristo, J Sandström and P Krogsgaard-Larsen. Algal carotenoids. 48. Structural assignments of geometrical isomers of fucoxanthin. Acta Chem. Scand. 1992; 46, 389-95.
M Honda, K Murakami, Y Watanabe, T Higashiura, T Fukaya, Wahyudiono, H Kanda and M Goto. The E/Z isomer ratio of lycopene in foods and the effect of heating with edible oils and fats on isomerization of (all-E)-lycopene. Eur. J. Lipid Sci. Technol. 2017; 119, 1600389.
M Honda, T Kodama, H Kageyama, T Hibino, Wahyudiono, H Kanda and M Goto. Enhanced solubility and reduced crystallinity of carotenoids, β-Carotene, and astaxanthin, by Z-isomerization. Eur. J. Lipid Sci. Technol. 2018; 120, 1800191.
A Kurinjery and A Kulanthaiyesu. Anti-hyaluronidase and cytotoxic activities of fucoxanthin cis/trans isomers extracted and characterized from 13 brown seaweeds. Process Biochem. 2022; 122, 53-68.
A Kawee-Ai, A Kuntiya and SM Kim. Anticholinesterase and antioxidant activities of fucoxanthin purified from the microalga Phaeodactylum tricornutum. Nat. Prod. Commun. 2013; 8, 1381-6.
S Xia, K Wang, L Wan, A Li, Q Hu and C Zhang. Production, characterization, and antioxidant activity of fucoxanthin from the marine diatom Odontella aurita. Mar. Drugs 2013; 11, 2667-81.
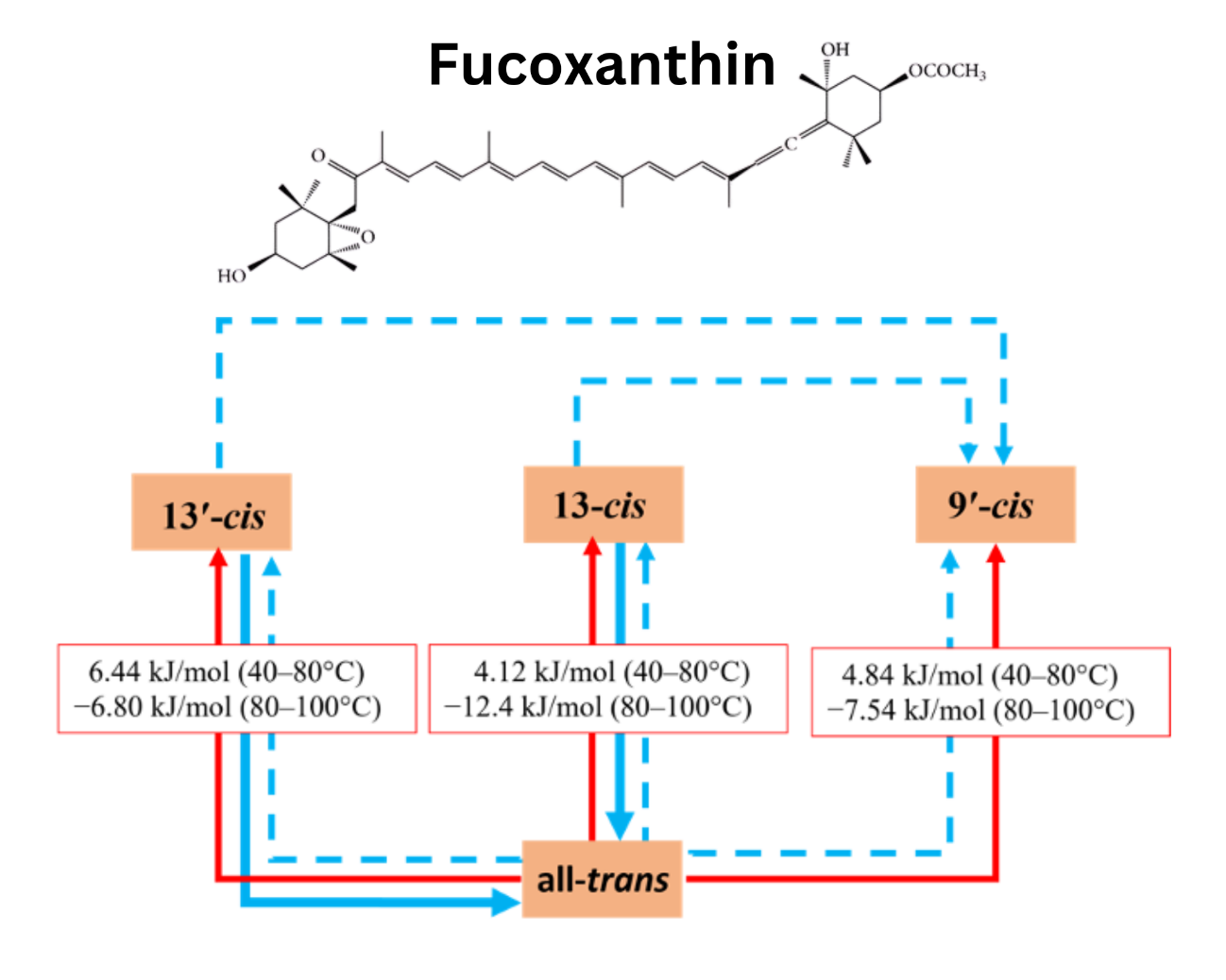
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






