Synthesis, Spectroscopic Evaluations and UV-Vis Titration Studies of New Symmetrical Amide Compounds Derived from N-6-[(4-pyridylmethylamino)carbonyl]-pyridine-2-carboxylic Acid Methyl Ester
DOI:
https://doi.org/10.48048/tis.2024.7405Keywords:
Tetraamide, Symmetry, Conformation, Flexible, Anion, Pre-organizedAbstract
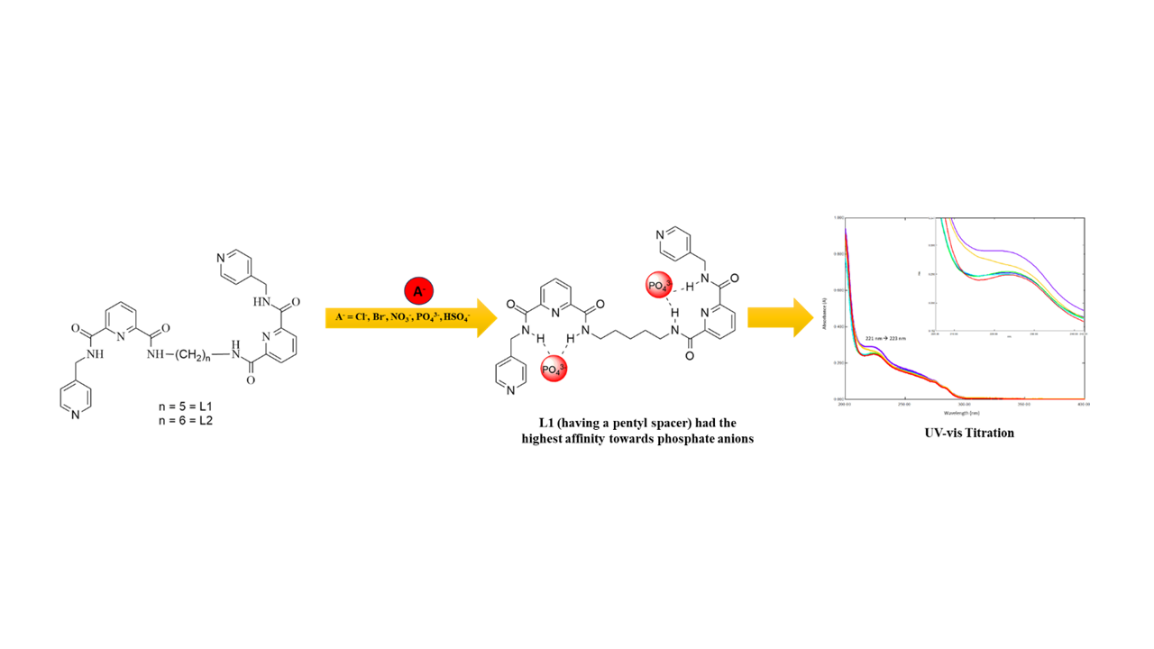
In this study, 2 flexible and pre-organized tetraamide compounds derived from N-6-[(4-pyridylmethylamino)carbonyl]-pyridine-2-carboxylic acid methyl ester namely 1,2-bis[N,N’-6-(4-pyridylmethylamido)pyridyl-2-carboxyamido]pentane (L1) and 1,2-bis[N,N’-6-(4-pyridylmethylamido)pyridyl-2-carboxyamido]hexane (L2) have been successfully synthesized when reacted with diamines in 1:2 ratio. These new compounds were built from combination of 3 main components, as a trend requires for anion receptor which are (i) 2,6-pyridine dicarboxamide moieties as targeted anion binding host, (ii) amino methyl pyridine pendants arms as the flexible moieties and (iii) pentyl (-C5H10-) and hexyl (-C6H12-) unit as the spacer. Compounds L1-L2 were fully characterized by using elemental analyzer, Fourier transform infrared (FTIR) spectroscopy, gas chromatography-mass spectroscopy (GC-MS), 1H, 2D NOESY and 13C Nuclear Magnetic Resonance (NMR) spectroscopies, and Ultraviolet-visible (UV-Vis) spectroscopies. In this study, anion titration methods were used to identify the affinity towards selected anions. The results showed that L1 (having a pentyl spacer) had the highest affinity towards phosphate anions as compared to L2, where the red shift changes were observed in the UV-vis spectrum at the amide region.
HIGHLIGHTS
- Two novel amide compounds with flexible and pre-organized structure are designed for use as potential anion receptors
- The flexible moieties at the amide and the linker allow hydrogen bonding interaction of the molecules with variety anions with different geometries
- Anion titration studies revealed good affinities towards phosphate anions as suggested in Hofmeister trend
GRAPHICAL ABSTRACT

Downloads
References
S Kundu, TK Egboluche, Z Yousuf and MA Hossain. Spectroscopic and colorimetric studies for anions with a new urea-based molecular cleft. Chemosensors 2021; 9, 287.
S Khan and J Ali. Chemical analysis of air and water. In: DP Häder and GS Erzinger (Eds.). Bioassays: Advanced methods and applications. Elsevier, Amsterdam, Netherlands, 2018.
X Jia, D Chen, L Bin, H Lu, R Zhang and Y Zheng. Highly selective and sensitive phosphate anion sensors based on AlGaN/GaN high electron mobility transistors functionalized by ion imprinted polymer. Sci. Rep. 2016; 6, 27728.
T Phuong, TD Cong, VT Ta and TH Nguyen. Study on leaching of phosphate from municipal wastewater treatment plant’s sewage sludge and followed by adsorption on Mg-Al layered double hydroxide. J. Nanomaterials 2022; 2022, 1777187.
T Guchhait, L Panda, SP Rout and M Giri. Fluoride-ion-mediated 1H/2D exchange in anion receptors: A 19F NMR probe. J. Mol. Struct. 2023; 1283, 135280.
V Suryanti, M Bhadbhade, DSC Black and N Kumar. N-acetylglyoxylic amide bearing a nitrophenyl group as anion receptors: NMR and X-ray investigations on anion binding and selectivity. J. Mol. Struct. 2017; 1146, 571-6.
A Dorazco-González, H Höpfl, F Medrano and AK Yatsimirsky. Recognition of anions and neutral guests by dicationic pyridine-2,6-dicarboxamide receptors. J. Org. Chem. 2010; 75, 2259-73.
MA Martínez-Aguirre, DM Otero, ML Álvarez-Hernández, T Torres-Blancas, A Dorazco-González and AK Yatsimirsky. Anion and sugar recognition by 2,6-pyridinedicarboxamide bis-boronic acid derivatives. Heterocycl. Comm. 2017; 23, 171-80.
CJ Sumby and LR Hanton. Syntheses and studies of flexible amide ligands: A toolkit for studying metallo-supramolecular assemblies for anion binding. Tetrahedron 2009; 65, 4681-91.
U Jakob, TO Petersen and W Bannwarth. Unusual metal complexes of N,N-bis(2-picolyl)amides - A comparative study of structure and reactivity. J. Organomet. Chem. 2015; 797, 46-51.
S Dalapati, S Jana and N Guchhait. Anion recognition by simple chromogenic and chromo-fluorogenic salicylidene Schiff base or reduced-Schiff base receptors. Spectrochim. Acta Mol. Biomol. Spectros. 2014; 129, 499-508.
SSR Namashivaya, AS Oshchepkov, H Ding, S Forster, VN Khrustalev and EA Katae. Anthracene-based receptors with a turn-on fluorescence response for nitrate. Org. Lett. 2019; 21, 8746-50.
N Chakraborty, S Bhuiya, A Chakraborty and S Das. Coumarin based azo dyes as anion sensors: A spectroscopic study. Indian J. Chem. 2018. 57A, 59-65.
ME Khansari, MH Hasan, CR Johnson, NA Williams, BM Wong, DR Powell, R Tandon and MA Hossain. Anion complexation studies of 3-nitrophenyl-substituted tripodal thiourea receptor: A naked-eye detection of sulfate via fluoride displacement assay. ACS Omega 2017; 2, 9057-66.
LM Lopez-Martinez, J Garcia-Elias, A Ochoa-Teran, HS Ortega, K Ochoa-Lara, CU Montano-Medina, AK Yatsimirsky, JZ Ramirez, V Labastida-Galvan and M Ordonez. Synthesis, characterization and anion recognition studies of new fluorescent alkyl bis(naphthylureylbenzamide)-based receptors. Tetrahedron 2017; 76, 130815.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2023 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






