Effects of LED Lighting Technology on Morphology, Antioxidant activity, and the Bioactive Compounds Accumulation of Anoectochilus burmannicus in the Greenhouse System
DOI:
https://doi.org/10.48048/tis.2023.6296Keywords:
Anoectochilus burmannicus, Antioxidant activity, Bioactive compounds, Continuous lighting, LED lighting technologyAbstract
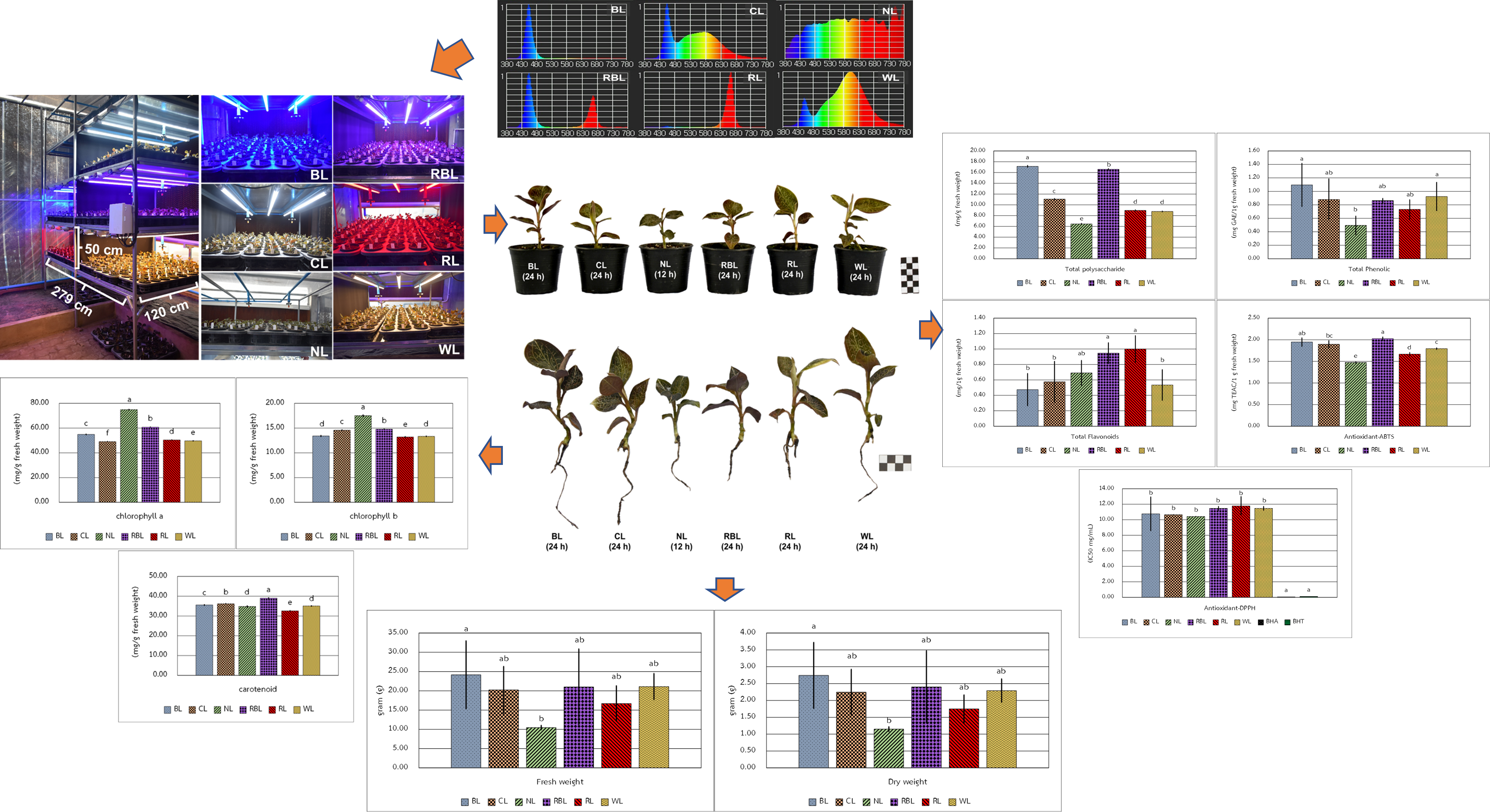
Anoectochilus burmannicus is valued for medicinal and ornamental purposes. It is characterized by beautiful foliage with attractively red-purple with bronze-colored veins blotches. Kinsenoside is an active compound in A. burmannicus which provided anti-inflammation properties, anti-obesity, and anti-diabetes properties. This study aimed to investigate the effects of light-emitting diode (LED) light quality on growth, photosynthetic pigment content, antioxidant activities, and the accumulation of bioactive compounds (polysaccharides, phenolics, and flavonoids) in A. burmannicus. Plants of A. burmannicus were grown under 6 different LED lighting in the greenhouse system for 60 days. The 6 treatments were blue light (BL, 24 h), cool light (CL, 24 h), natural light (NL, 12 h), red:blue 1:1 light (RBL, 24 h), red light (RL, 24 h), and warm light (WL, 24 h). The results showed that A. burmannicus plants have a positive effect on the growth and bioactive compounds under LED lights (24 h) technology compared to natural light (12 h). The WL treatment had the most positive effect on morphological parameters, while the BL treatment showed an enhancement in fresh weight and dry weight. NL treatment is increased the Chlorophyll a and Chlorophyll b content. The Carotenoid content increased in the RBL treatment phenolics content was considered tent were considered to be superior to the BL treatment. The total flavonoid content is increased in the RL and RBL treatments. The antioxidant activity of ABTS was significantly higher in the RBL treatment. This indicates that BL is required for the normal growth, biomass, and accumulation of polysaccharides and phenolic of A. burmannicus, while RBL is enhanced the accumulation of flavonoids and antioxidant activity. This technology is conducive to achieving large-scale sustainable production of high-quality medicinal plant materials.
GRAPHICAL ABSTRACT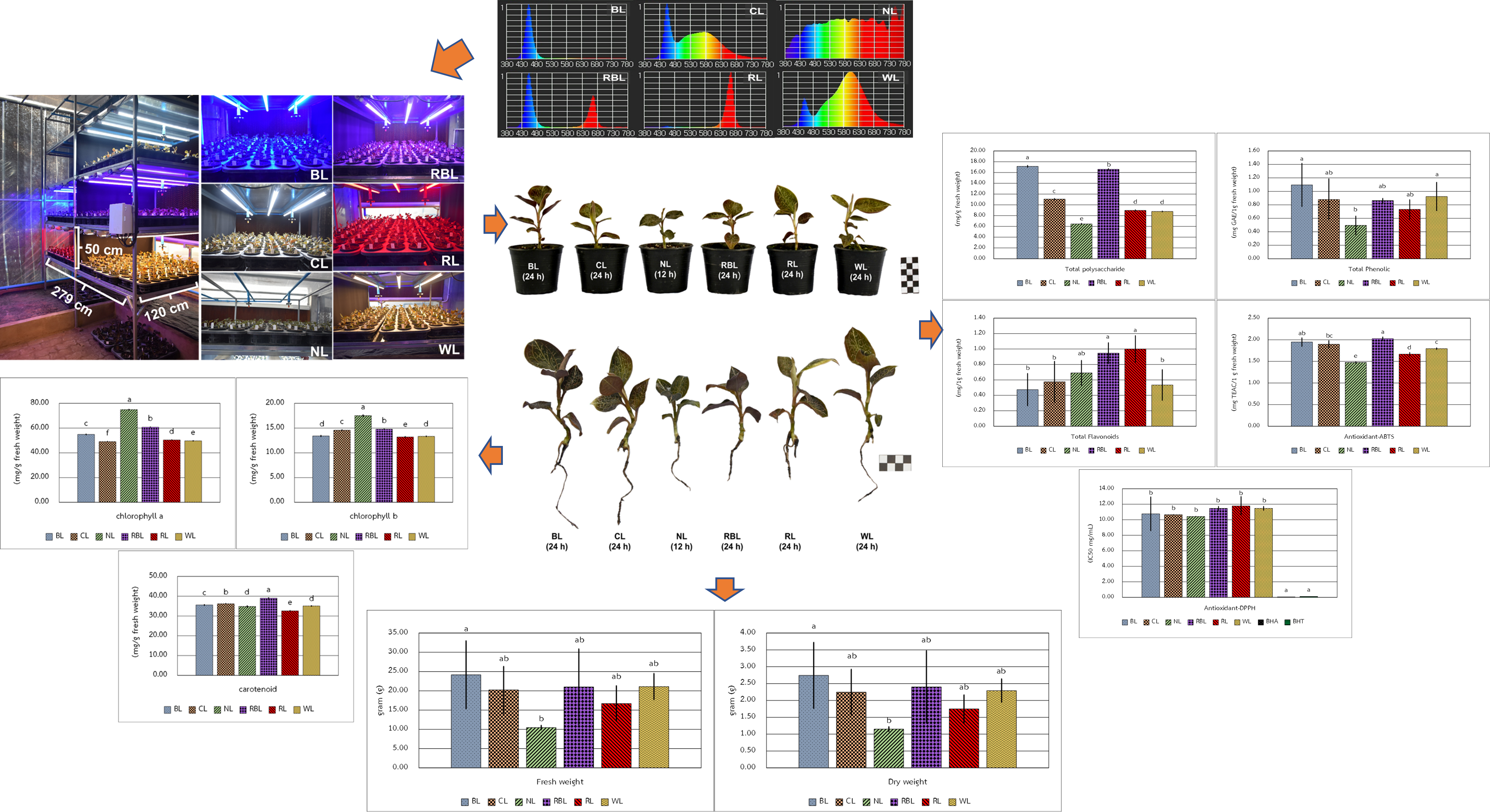
Downloads
References
XM Du, N Irino, N Furusho, J Hayashi and Y Shoyama. Pharmacologically active compounds in the Anoectochilus and Goodyera species. J. Nat. Med. 2008; 62, 132-48.
SY Wang, YH Kuo, HN Chang, PL Kang, HS Tsay, KF Lin, NS Yang and LF Shyur. Profiling and characterization antioxidant activities in Anoectochilus formosanus Hayata. J. Agr. Food Chem. 2002; 50, 1859-65.
F Zhang, Y Lv, H Dong and S Guo. Analysis of genetic stability through Intersimple sequence repeats molecular markers in micropropagated plantlets of Anoectochilus formosanus Hayata, a medicinal plant. Biol. Pharm. Bull. 2010; 33, 384-8.
S Takatsuki, JD Wang, T Narui and T Okuyama. Studies on the components of crude drug “Kim-soan-lian”. J. Jap. Botany. 1992; 67, 121-3.
HA Pedersen. Anoectochilus. In: T Santisuk and K Larsen (Eds.). Flora of Thailand, Bangkok, Thailand, 2011, p. 10-20.
S Sitthisatchatham. Orchids handbook (in Thai). Sarakadee press, Nonthaburi, Thailand. 2015.
P Pitchakan. Research on propagation and study of biological activities of Anoectochilus burmannicus and development as functional products to serve the royal initiative the plant genetic conservation project under the royal initiative of her royal highness princess Maha Chakri Sirindhorn (in Thai). Chiang Mai University, Chiang Mai, Thailand, 2016.
P Kaewthongprakum, T Tangtragoon, P Sutigoolabud and A Kanpington. Environmental factors affecting the growth of Anoectochilus burmannicus Rolfe, Ban Pong Krai, Pong Yang Sub District, Mae Rim District, Chiang Mai Province (in Thai). Rajamangala Univ. Tech. Srivijaya Res. J. 2019; 11, 181-96.
R Malkin and K Niyogi. Photosynthesis. In: B Buchanan, W Gruissem and R Jones (Eds.). Biochemistry and molecular biology of plants. 2nd eds. American Society of Plant Physiologists, Rockville, Maryland, 2000, p. 568-628.
T Ouzounis, E Rosenqvist and CO Ottosen. Spectral effects of artificial light on plant physiology and secondary metabolism: A review. HortScience 2015; 50, 1128-35.
F Bantis, S Smirnakou, T Ouzounis, A Koukounaras, N Ntagkas and K Radoglou. Current status and recent achievements in the field of horticulture with the use of light-emitting diodes (LEDs). Sci. Hortic. 2018; 235, 437-51.
F Abidi, T Girault, O Douillet, G Guillemain, G Sintes, M Laffaire, HB Ahmed, S Smiti, L Huché-Thélier and N Leduc. Blue light effects on rose photosynthesis and photomorphogenesis. Plant Biol. 2013; 15, 67-74.
AI Velez-Ramirez, WV Ieperen, D Vreugdenhil and FF Millenaar. Plants under continuous light. Trends Plant Sci. 2011; 16, 310-8.
MS Haque, KH Kjaer, E Rosenqvist and CO Ottosen. Continuous light increases growth, daily carbon gain, antioxidants, and alters carbohydrate metabolism in a cultivated and a wild tomato species. Front. Plant Sci. 2015; 6, 522.
N Ohtake, M Ishikura, H Suzuki, W Yamori and E Goto. Continuous irradiation with alternating red and blue light enhances plant growth while keeping nutritional quality in lettuce. HortScience 2018; 53, 1804-9.
J Lanoue, J Zheng, C Little, A Thibodeau, B Grodzinski and X Hao. Alternating red and blue light-emitting diodes allows for injury-free tomato production with continuous lighting. Front. Plant Sci. 2019; 10, 1114.
T Kozai, G Niu and M Takagaki. Plant factory: An indoor vertical farming system for efficient quality food production. Academic Press, Cambridge, 2015.
WV Ieperen. Plant morphological and developmental responses to light quality in a horticultural context. In: Proceedings of the 7th International Symposium on Light in Horticulture, Wageningen, Netherlands. 2012, p. 131-9.
O Alrifai, X Hao, MF Marcone and R Tsao. Current review of the modulatory effects of LED lights on photosynthesis of secondary metabolites and future perspectives of microgreen vegetables. J. Agr. Food Chem. 2019; 67, 6075-90.
P Benincasa, B Falcinelli, S Lutts, F Stagnari and A Galieni. Sprouted grains: A comprehensive review. Nutrients 2019; 11, 421.
Z Ma, S Li, M Zhang, S Jiang and Y Xiao. Light intensity affects growth, photosynthetic capability, and total flavonoid accumulation of Anoectochilus plants. HortScience 2010; 45, 863-7.
Q Shao, H Wang, H Guo, A Zhou, Y Huang, Y Sun and M Li. Effects of shade treatments on photosynthetic characteristics, chloroplast ultrastructure, and physiology of Anoectochilus roxburghii. PLoS ONE 2014; 9, e85996.
W Wang, M Su, H Li, B Zeng, Q Chang and Z Lai. Effects of supplemental lighting with different light qualities on growth and secondary metabolite content of Anoectochilus roxburghii. PeerJ 2018; 6, e5274.
F Thoma, A Somborn-Schulz, D Schlehuber, V Keuter and G Deerberg. Effects of light on secondary metabolites in selected leafy greens: A review. Front. Plant Sci. 2020; 11, 497.
RJ Porra. The chequered history of the development and use of simultaneous equations for the accurate determination of chlorophylls a and b. Photosynth. Res. 2002; 73, 149-56.
S Ye, Q Shao, M Xu, S Li, M Wu, X Tan and L Su. Effects of light quality on morphology, enzyme activities, and bioactive compound contents in Anoectochilus roxburghii. Front. Plant Sci. 2017; 8, 857.
J Liu, Y Li, W Liu, Q Qi, X Hu, S Li, J Lei and L Rong. Extraction of polysaccharide from Dendrobium nobile Lindl. by subcritical water extraction. ACS Omega 2019; 4, 20586-94.
H Zhu, Y Wang, Y Liu, Y Xia and T Tang. Analysis of flavonoids in Portulaca oleracea L. by UV-vis spectrophotometry with comparative study on different extraction technologies. Food Anal. Meth. 2010; 3, 90-7.
G Cao, HM Alessio and RG Cutler. Oxygen-radical absorbance capacity assay for antioxidants. Free Radical Bio. Med. 1993; 14, 303-11.
T Hatano, H Kagawa, T Yasuhara and T Okuda. Two new flavonoids and other constituents in licorice root: Their relative astringency and radical scavenging effects. Chem. Pharm. Bull. 1988; 36, 2090-7.
M Simlat, P Ślęzak, M Moś, M Warchoł, E Skrzypek and A Ptak. The effect of light quality on seed germination, seedling growth and selected biochemical properties of Stevia rebaudiana Bertoni. Sci. Hortic. 2016; 211, 295-304.
K Abe, S Kido, T Maeda, D Kami, H Matsuura, H Shimura and T Suzuki. Glucosinolate profiles in Cardamine fauriei and effect of light quality on glucosinolate concentration. Sci. Hortic. 2015; 189, 12-6.
A Manivannan, P Soundararajan, N Halimah, CH Ko and BR Jeong. Blue LED light enhances growth, phytochemical contents, and antioxidant enzyme activities of Rehmannia glutinosa cultured in vitro. Hortic. Environ. Biotechnol. 2015; 56, 105-13.
SW Lee, JM Seo, MK Lee, JH Chun, P Antonisamy, MV Arasu, T Suzuki, NA Al-Dhabi and SJ Kim. Influence of different LED lamps on the production of phenolic compounds in common and Tartary buckwheat sprouts. Ind. Crop Prod. 2014; 54, 320-6.
JBA Bautista, A Cruz, JDLLopez, MJS Macale and RSC Sevilla. Growing plant using four different lighting system: an experimental study. La Consolacion University Philippines, Malolos, Philippines, 2020, p. 39.
ML Bartucca, M Guiducci, B Falcinelli, DD Buono and P Benincasa. Blue: Red LED light proportion affects vegetative parameters, pigment content, and oxidative status of einkorn (Triticum monococcum L. ssp. monococcum) wheatgrass. J. Agr. Food Chem. 2020; 68, 8757-63.
T Lei, S Gao, X Lin, K Zhang, Y Liu, W Li, D Hu, X Yu, M Jiang, Q Chen and Y Sun. Calcium signaling mediated the regulation of growth and polysaccharide accumulation by light quality in Dendrobium officinale protocorms. Hortic. Environ. Biotechnol. 2021; 62, 287-97.
XP Lin and ZX Lai. Effect light quality on the proliferation of protocorm and active ingredient contents of Dendrobium officinale. Chin. J. Trop. Crops 2015; 36, 1796-801.
J Ren, JP Liang, R Zhou, QJ Feng, ML Jia and XX Wang. Effects of different light qualities on growth and accumulation of medicinal components in Astragalus membranaceus. J. Shanxi Agr. Sci. 2014; 42, 1078-81.
EH Kim, SH Kim, JI Chung, HY Chi, JA Kim and IM Chung. Analysis of phenolic compounds and isoflavones in soybean seeds (Glycine max (L.) Merill) and sprouts grown under different conditions. Eur. Food Res. Tech. 2006; 222, 201-8.
M Johkan, K Shoji, F Goto, SN Hashida and T Yoshihara. Blue light-emitting diode light irradiation of seedlings improves seedling quality and growth after transplanting in red leaf lettuce. HortScience 2010; 45, 1809-14.
K Shoji, E Goto, S Hashida, F Goto and T Yoshihara. Effect of light quality on the polyphenol content and antioxidant activity of sweet basil (Ocimum basilicum L.). In: Proceedings of the 6th International Symposium on Light in Horticulture, Tsukuba, Japan. 2009. p. 95-9.
JZ Lin. 2009, Functional analysis of Arabidopsis 4CL3 gene in flavonoid biosynthesis. Ph. D. Dissertation. Hunan University, Changsha, China.
A Žukauskas, Z Bliznikas, K Breivė, A Novičkovas, G Samuolienė, A Urbonavičiūtė, A Brazaitytė, J Jankauskienė and P Duchovskis. Effect of supplementary pre-harvest LED lighting on the antioxidant properties of lettuce cultivars. In: Proceedings of the 6th International Symposium on Light in Horticulture, Tsukuba, Japan. 2009. p. 87-90.
ES Jung, S Lee, SH Lim, SH Ha, KH Liu and CH Lee. Metabolite profiling of the short-term responses of rice leaves (Oryza sativa cv. Ilmi) cultivated under different LED lights and its correlations with antioxidant activities. Plant Sci. 2013; 210, 61-9.
T Ahmadi, L Shabani and MR Sabzalian. LED light sources improved the essential oil components and antioxidant activity of two genotypes of lemon balm (Melissa officinalis L.). Bot. Stud. 2021; 62, 1-13.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2023 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






