In vitro Studies of Antibacterial and Antifungal Activity of Novel Substituted Benzimidazole Derivatives
DOI:
https://doi.org/10.48048/tis.2023.6110Keywords:
Benzimidazole, Chloroacetyl chloride, Amines, Antibacterial and antifungal activityAbstract
A series of 2-substituted amino-1-(1H-benzo[d]imidazol-1-yl)ethanone was prepared by reacting with benzimidazole and chloroacetyl chloride by stirring method. The resultant compound was dissolved in ethanol and refluxed with aryl/aralkyl amine in the presence of anhydrous potassium carbonate. The tested compounds were evaluated against bacterial agents such as E. coli, P. aeruginosa, K. pneumonia, S. typhi, and S. aureus, amoxicillin has been used as a control drug, and also screened for antifungal activity against C. albicans and A. niger at 50 and 100 mg/mL level, griseofulvin used as control drug. The structure-activity relationship led to the conclusion compound PS3 showed equipotent potent, PS4 showed moderate, and PS2 showed less moderate activity. The aromatic/aliphatic chain containing compounds PS1 and PS5 showed the least activity against all bacteria. Compound PS3, PS4 and PS1 showed better activity against C. albicans at 100 µg/mL concentration. The tested compounds PS1, PS3, PS2 and PS4 showed good activity at 100 µg/mL concentration. The compound PS3 can serve as a lead molecule for further development as a new class of antimicrobial agent.
GRAPHICAL ABSTRACT 
Downloads
References
M Tonelli, G Paglietti, V Boido, F Sparatore, F Marongiu, E Marongiu, PL Colla and R Loddo. Antiviral activity of benzimidazole derivatives. Antiviral activity of 1-substituted-2-[(benzotriazol-1/2-yl)methyl]benzimidazoles. Chem. Biodivers. 2008; 5, 2386-401.
KCS Achar, KM Hosamani and HR Seetharamareddy. In-vivo analgesic and anti-inflammatory activities of newly synthesized benzimidazole derivatives. Eur. J. Med. Chem. 2010; 45, 2048-54.
G Serban, O Stanasel, E Serban and S Bota. 2-Amino-1,3,4-thiadiazole as a potential scaffold for promising antimicrobial agents. Drug Des. Devel. Ther. 2018; 12, 1545-66.
ZI Kirkwood, BC Millar, DG Downey and JE Moore. Antimicrobial effect of dimethyl sulfoxide and N, N-Dimethylformamide on Mycobacterium abscessus: Implications for antimicrobial susceptibility testing. Int. J. Mycobacteriol. 2018; 7, 134-6.
YK Yoon, MA Ali, TS Choon, R Ismail, AC Wei, RS Kumar, H Osman and F Beevi. Antituberculosis: Synthesis and antimycobacterial activity of novel benzimidazole derivatives. Biomed. Res. Int. 2013; 2013, 926309.
HM Refaat. Synthesis and anticancer activity of some novel 2-substituted benzimidazole derivatives. Eur. J. Med. Chem. 2010; 45, 2949-56.
B Bhrigu, N Siddiqui, D Pathak, MS Alam, R Ali and B Azad. Anticonvulsant evaluation of some newer benzimidazole derivatives: Design and synthesis. Acta Pol. Pharm. 2012; 69, 53-62.
NC Desai and GM Kotadiya. Microwave-assisted synthesis of benzimidazole bearing 1,3,4-oxadiazole derivatives: Screening for their in vitro antimicrobial activity. Med. Chem. Res. 2014; 3, 4021-33.
PNK Babu, B Ramadevi, Y Poornachandra and CG Kumar. Synthesis, antimicrobial, and anticancer evaluation of novel 2-(3-methylindolyl)benzimidazole derivatives. Med. Chem. Res. 2014; 23, 3970-8.
D Song and S Ma. Recent development of benzimidazole-containing antibacterial agents. Chem. Med. Chem. 2016; 11, 646-59.
K Kamanna. Synthesis and pharmacological profile of benzimidazoles. In: M Marinescu (Ed.). Chemistry and applications of benzimidazole and its derivatives. IntechOpen, London, 2019.
AN Khayyat, KO Mohamed, AM Malebari and A El-Malah. Design, synthesis, and antiproliferative activities of novel substituted imidazole-thione linked benzotriazole derivatives. Molecules 2021; 26, 5983.
N Vashist, SS Sambi, B Narasimhan, S Kumar, SM Lim, SAA Shah, K Ramasamy and V Mani. Synthesis and biological profile of substituted benzimidazoles. Chem. Cent. J. 2018; 12, 125.
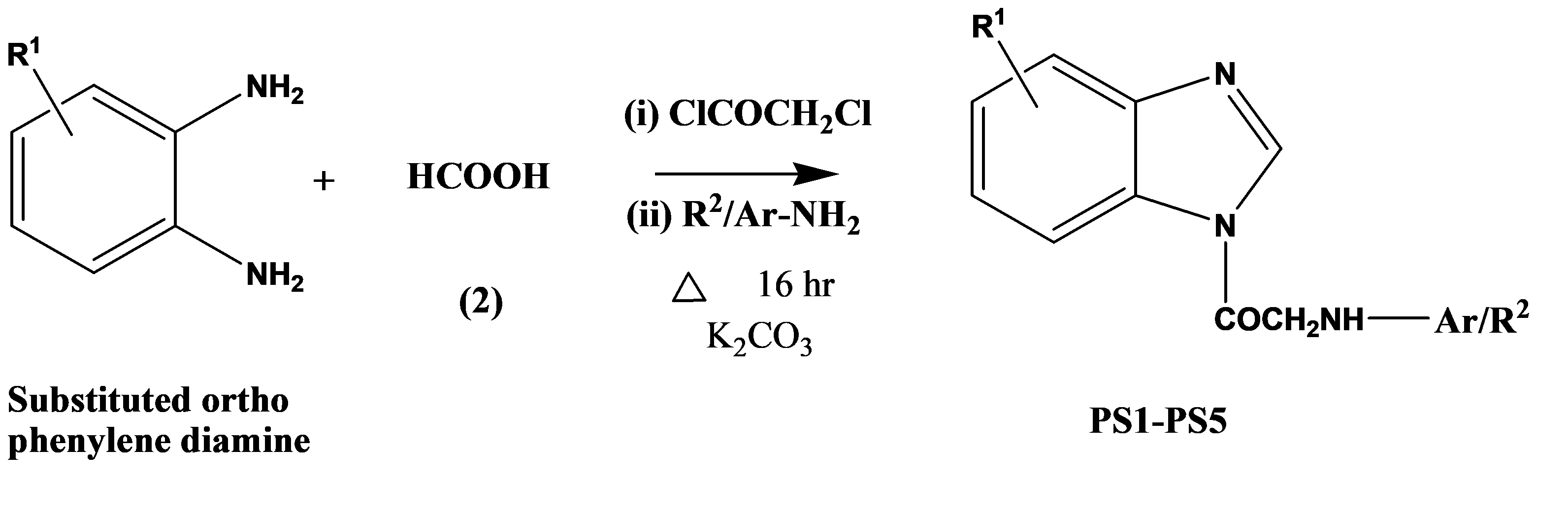
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2022 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






