Development of Delayed-Release Matrix Tablets Comprising Solid Dispersion of Mefenamic Acid
DOI:
https://doi.org/10.48048/tis.2023.6078Keywords:
Mefenamic acid, Solid dispersion, Delayed-release, Matrix tablet, DissolutionAbstract
Mefenamic acid (MA), a member of nonselective nonsteroidal anti-inflammatory drugs (NSAIDs), has been widely use to relief pain and inflammation. Its medical uses are limited by poor aqueous solubility resulting in low bioavailability and gastric irritation. The aim of this study was to develop a mefenamic acid delayed-release matrix tablet formulation using solid dispersion (SD). Delayed-release drug delivery systems were designed to retard drug release in upper gastrointestinal tract avoiding gastrointestinal (GI) adverse reactions. SDs of MA were successfully prepared by solvent evaporation method employing methanol as a solvent. SDs incorporated surfactant and super disintegrant gave much higher rates of dissolution than SDs with combined carriers (PEG and surfactant), SD containing PEG and pure drug, respectively. The optimal SD containing MA:PEG4000:poloxamer188:crospovidone in the ratio 1:8:1:3 exhibited higher amount of drug release up to 8-fold compared with pure MA. FTIR and DSC were performed to identify the physicochemical interaction between drug and polymers. The resulting data justified that no change in the chemical structure of MA and the crystalline MA transformed into the amorphous state after preparation. The formulation F4 delayed-release tablet comprising SD of MA dissolved less than 4 % in artificial gastric fluid in the initial 2 h and released more than 95 % at 3 h in the artificial intestinal fluid. Accordingly, formulation F4 containing polyethylene oxide as a time-controlled matrix-forming polymer was a promising delayed-release solid dispersion system of MA.
HIGHLIGHTS
- The present study demonstrated a mefenamic acid delayed-release matrix tablet formulation using solid dispersion (SD)
- The optimal SD of mefenamic acid exhibited higher amount of drug release (8-fold) compared with that of the pure drug
- The tablet formulation F4 containing polyethylene oxide is capable of releasing mefenamic acid in a typical delayed-release profile
GRAPHICAL ABSTRACT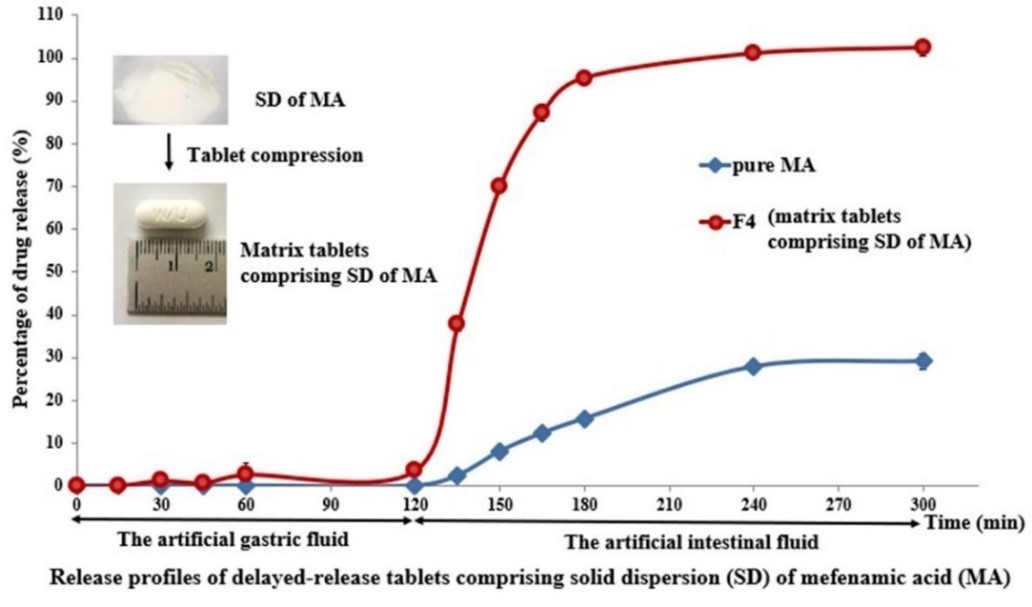
Downloads
References
N Cimolai. The potential and promise of mefenamic acid. Expert. Rev. Clin. Pharmacol. 2013; 6, 289-305.
M Vahedi, SB Hasanpoor-Azghady, L Amiri-Farahani and I Khaki. Comparison of effect of auriculotherapy and mefenamic acid on the severity and systemic symptoms of primary dysmenorrhea: A randomized clinical trial. Trials 2021; 22, 655.
C Cooper, R Chapurlat, N Al-Daghri, G Herrero-Beaumont, O Bruyère, F Rannou, R Roth, D Uebelhart and JY Reginster. Safety of oral non-selective non-steroidal anti-inflammatory drugs in osteoarthritis: What does the literature say? Drugs Aging 2019; 36, 15-24.
PB Patil, VRM Gupta, RH Udupi, K Srikanth and BSG Prasad. Development of dissolution medium for poorly water soluble drug mefenamic acid. Res. J. Pharmaceut. Biol. Chem. Sci. 2010; 1, 544-9.
B Baghernejad. Synthesis, analgesic and anti-inflammatory activities of aminobenzothiazole derivatives of mefenamic acid. Pharmaceut. Chem. J. 2021; 55, 355-60.
FL Lanza, FKL Chan and EMM Quigley. Guidelines for prevention of NSAID-related ulcer complications. Am. J. Gastroenterol. 2009; 104, 728-38.
R Smolková, V Zeleňák, R Gyepes and D Hudecová. Biological activity of two novel zinc(II) complexes with NSAID mefenamic acid. Chem. Paper. Chem. Zvesti 2020; 74, 1525-40.
D Sid, M Baitiche, Z Elbahri, F Djerboua, M Boutahala, Z Bouaziz and ML Borgne. Solubility enhancement of mefenamic acid by inclusion complex with β-cyclodextrin: In silico modelling, formulation, characterisation, and in vitro studies. J. Enzym. Inhib. Med. Chem. 2021; 36, 605-17.
P Sriamornsak, S Limmatvapirat, S Piriyaprasarth, P Mansukmanee and Z Huang. A new self-emulsifying formulation of mefenamic acid with enhanced drug dissolution. Asian J. Pharmaceut. Sci. 2015; 10, 121-7.
M Kumar, D Singh and N Bedi. Mefenamic acid-loaded solid SMEDDS: An innovative aspect for dose reduction and improved pharmacodynamic profile. Ther. Deliv. 2019; 10, 21-36.
K Bodnár, SP Hudson and ÅC Rasmuson. Stepwise use of additives for improved control over formation and stability of mefenamic acid nanocrystals produced by antisolvent precipitation. Cryst. Growth Des. 2017; 17, 454-66.
E Valkama, O Haluska, VP Lehto, O Korhonen and K Pajula. Production and stability of amorphous solid dispersions produced by a freeze-drying method from DMSO. Int. J. Pharmaceut. 2021; 606, 120902.
QB Jarrar, Z Ahmad, AA Kadir, ZA Zakaria and MN Hakim. Liposome encapsulation: A promising approach to enhanced and safe mefenamic acid therapy. Int. Res. J. Educ. Sci. 2017; 1, 32-9.
D Mudit, KP Keshavarao and P Selvam. Improvement of solubility and dissolution rate of mefenamic acid by solid dispersion in PEG4000. Int. Res. J. Pharm. 2011; 2, 207-10.
E Prasad, J Robertson and GW Halbert. Mefenamic acid solid dispersions: Impact of formulation composition on processing parameters, product properties and performance. Int. J. Pharmaceut. 2022; 616, 121505.
G Tambosi, PF Coelho, S Luciano, ICS Lenschow, M Zétola, HK Stulzer and BR Pezzini. Challenges to improve the biopharmaceutical properties of poorly water-soluble drugs and the application of the solid dispersion technology. Matéria 2018, 23, 13.
GP Andrews, H Zhai, S Tipping and DS Jones. Characterisation of the thermal, spectroscopic and drug dissolution properties of mefenamic acid and polyoxyethylene-polyoxypropylene solid dispersions. J. Pharmaceut. Sci. 2009; 98, 4545-56.
CHV Prasada Rao, MV Nagabhushanam and CH Prabhakar. Enhancement of dissolution rate of poorly soluble drug mefenamic acid by solid dispersion. Res. J. Pharmaceut. Biol. Chem. Sci. 2011; 2, 1025-35.
MV Nagabhushanam and AS Rani. Dissolution enhancement of mefenamic acid using solid dispersions in crospovidone. Int. J. Pharm. Pharmaceut. Sci. 2011; 3, 16-9.
WS Wong, CS Lee, HM Er and WH Lim. Preparation and evaluation of palm oil-based polyesteramide solid dispersion for obtaining improved and targeted dissolution of mefenamic acid. J. Pharmaceut. Innovat. 2017; 12, 76-89.
S Shariff, S Saleem, ANP Kumar, BA Kumar and P Madhusudhan. Formulation and evaluation of mefenamic acid solid dispersions using PEG-4000. Int. Res. J. Pharm. 2013; 4, 155-9.
W Putranti, L Widiyastuti and F Ulfani. Enhancing the dissolution rate of mefenamic acid with solid dispersion system using Avicel PH-101. Pharmaciana 2019; 9, 119-28.
RS Kumar and A Kumari. Formulation and evaluation of mefenamic acid solid dispersions employing starch citrate-a new solubility enhancer. J. Drug Deliv. Therapeut. 2019; 9, 48-52.
PJ Sheskey, WG Cook and CG Cable. Handbook of pharmaceutical excipient. 8th ed. Pharmaceutical Press, London, 2017, p. 705-6.
SP Chaudhari and RP Dugar. Application of surfactants in solid dispersion technology for improving solubility of poorly water soluble drug. J. Drug Deliv. Sci. Tech. 2017; 41, 68-77.
K Rajbanshi, R Bajracharya, A Shrestha and P Thapa. Dissolution enhancement of aceclofenac tablet by solid dispersion technique. Int. J. Pharmaceut. Sci. Res. 2014; 5, 127-39.
Y Qiu and PI Lee. Developing solid oral dosage forms (Second Edition). Pharmaceut. Theor. Pract. 2017; 2017, 519-54.
SH Song, BR Chae, SI Sohn, DW Yeom, HY Son, JH Kim, SR Kim, SG Lee and YW Choi. Formulation of controlled-release pelubiprofen tablet using Kollidon® SR. Int. J. Pharmaceut. 2016; 511, 864-75.
L Cimpoae, LL Rus and RI Iovanov. A study on the influence of the formulation factors on in vitro release of ketoprofen from sustained release tablets. Rom. J. Pharmaceut. Pract. 2021; 14, 41-8.
W Iffat, MH Shoaib, RI Yousuf, F Qazi, ZA Mahmood, IN Muhammad, K Ahmed, FR Ahmed and MS Imtiaz. Use of Eudragit RS PO, HPMC K100M, ethyl cellulose, and their combination for controlling nicorandil release from the bilayer tablets with atorvastatin as an immediate-release layer. J. Pharmaceut. Innovat. 2022; 17, 429-48.
K Patel, S Shah and J Patel. Solid dispersion technology as a formulation strategy for the fabrication of modified release dosage forms: A comprehensive review. Daru 2022; 30, 165-89.
Y Zhao, T Xin, T Ye, X Yang and W Pan. Solid dispersion in the development of a nimodipine delayed-release tablet formulation. Asian J. Pharmaceut. Sci. 2014; 9, 35-41.
A Avdeef. Solubility of sparingly-soluble ionizable drugs. Adv. Drug Deliv. Rev. 2007; 59, 568-90.
B Bhattacharya, A Mondal, SR Soni, S Das, S Bhunia, KB Raju, A Ghosh and CM Reddy. Multidrug salt forms of norfloxacin with non-steroidal anti-inflammatory drugs: Solubility and membrane permeability studies. CrystEngComm 2018; 20, 6420-9.
R Mehta, A Chawla, P Sharma and P Pawar. Formulation and in vitro evaluation of Eudragit S‑100 coated naproxen matrix tablets for colon‑targeted drug delivery system. J. Adv. Pharm. Technol. Res. 2013; 4, 31-41.

Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2022 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






