In-vitro Study of Trachyspermum ammi extract against Diabetes Mellitus
DOI:
https://doi.org/10.48048/tis.2022.5698Keywords:
Antidiabetic screening, Starch plate assay, Yeast assay, In vitro antidiabetic assays, Glucose uptake assayAbstract
Diabetes resulting from the low secretion of insulin and insulin resistance, is a chronic metabolic disorder. Medication from plant materials for treatment of Diabetes Mellitus is advised as good source for novel drug or leading to preparation of novel drug. We investigated the antidiabetic property of fractions and subfractions obtained from the ethyl acetate extract of Trachyspermum ammi seeds. TLC analysis of crude extract was done. Then column chromatography was used to isolate the fractions of T. ammi. Primary screening of fractions was done by Starch-Agar Gel Diffusion assay for identification of Alpha-amylase inhibitors. Then after chromatographic separation of subfractions, their secondary screening was carried out by Glucose-Uptake assay in Yeast cells. Finally the in vitro Anti-diabetic activity of crude and screened subfraction of ethyl acetate extract of T. ammi was evaluated in 3T3-L1 cell lines. The results presented the antidiabetic activity of T. ammi and their fractions along with subfractions. Further in vitro cell line assay also proved the antidiabetic activity of T. ammi. It is suggested that the T. ammi had antidiabetic activity by inhibiting the α-amylase enzyme, also by facilitating the glucose utilization in adipose tissues and skeletal muscles.
HIGHLIGHTS
- TLC analysis of crude extract of Trachyspermum ammi was performed
- Starch-Agar Gel Diffusion assay for identification of Alpha-amylase inhibitors was done
- Glucose-Uptake assay in Yeast cells was carried out
- In-vitro Anti-diabetic activity of ethyl acetate extract of ammi was evaluated in 3T3-L1 cell lines
GRAPHICAL ABSTRACT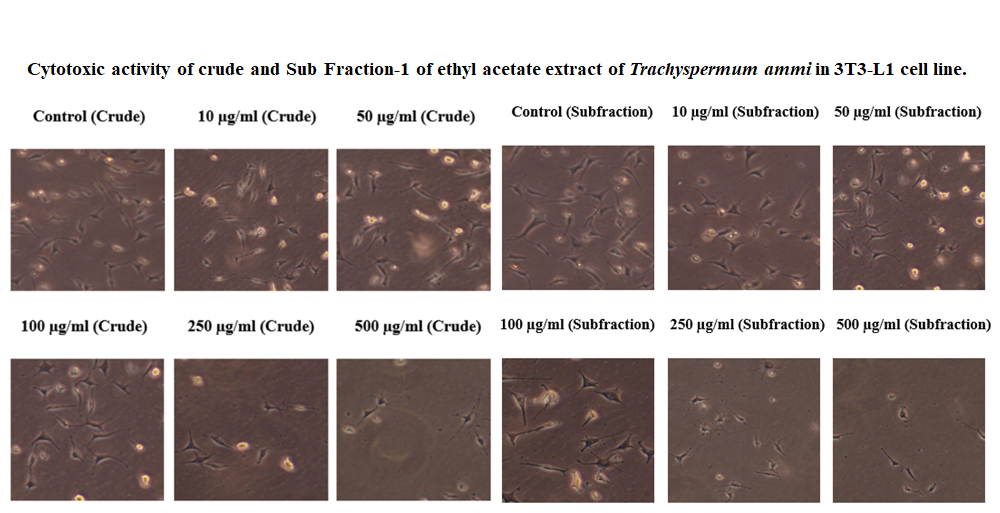
Downloads
References
K Mangesh, G Vidyasagar and K Ashwini. Diabetes and complications at a glance for pharmacy researchers. Int. J. Pharmaceut. Sci. Rev. Res. 2014; 25, 281-9.
A Chauhan, PK Sharma, P Srivastava, N Kumar, R Dudhe and R Dudhe. Plants having potential antidiabetic activity: A review. Scholars Res. Libr. 2010; 2, 369-87.
M Badran and I Laher. Type II diabetes mellitus in arabic-speaking countries. Int. J. Endocrinol. 2012; 2012, 902873.
ME Tucker. IDF Atlas: About 415 million adults worldwide have diabetes. In: Proceedings of the International Diabetes Federation 2015 World Congress, Columbia, Canada. 2015.
DC Whitcomb and ME Lowe. Human pancreatic digestive enzymes. Dig. Dis. Sci. 2007; 52, 1-17.
J Iulek, OL Franco, M Silva, CT Slivinski, C Bloch, DJ Rigden and MFGD Sá. Purification, biochemical characterisation and partial primary structure of a new α-amylase inhibitor from Secale cereale (rye). Int. J. Biochem. Cell. Biol. 2000; 32, 1195-204.
L Kandra. α-Amylases of medical and industrial importance. J. Mol. Struct. 2003; 666-667, 487-98.
GC Brayer, Y Luo and SG Withers. The structure of human pancreatic a-amylase at 1.8 A resolution and comparisons with related enzymes. Protein Sci. 1995; 4, 1730-42.
S Tangphatsornruang, M Naconsie, C Thammarongtham and J Narangajavana. Isolation and characterization of an α-amylase gene in cassava (Manihot esculenta). Plant. Physiol. Biochem. 2005; 43, 821-7.
KJ Naquvi, J Ahamad, SR Mir, M Ali and M Shuaib. Review on role of natural alpha-glucosidase inhibitors for management of diabetes mellitus. Int. J. Biomed. Res. 2011; 2, 374‐80.
K Hullatti and M Telagari. In vitro α-amylase and α-glucosidase inhibitory activity of Adiantum caudatum Linn. and Celosia argentea Linn. extracts and fractions. Indian J. Pharmacol. 2015; 47, 425-9.
AYY Cheng and IG Fantus. Oral antihyperglycemic therapy for type 2 diabetes mellitus. Can. Med. Assoc. J. 2005; 172, 213-26.
T Fujisawa, H Ikegami, K Inoue, Y Kawabata and T Ogihara. Effect of two α-glucosidase inhibitors, voglibose and acarbose, on postprandial hyperglycemia correlates with subjective abdominal symptoms. Metabolism 2005; 54, 387-90.
AY Oubré, TJ Carlson, SR King and GM Reaven. From plant to patient: an ethanomedical approach to the identification of new drugs for the treatment of NIDDM. Diabetologia 1997; 40, 614-7.
A Saxena and NK Vikram. Role of selected Indian plants in management of Type 2 Diabetes: A Review. J. Alternative Compl. Med. 2004; 10, 369-78.
RJ Marles and NR Farnsworth. Antidiabetic plants and their active constituents. Phytomedicine 1995; 2, 137-89.
MH Boskabady, S Alitaneh and A Alavinezhad. Carum copticum Linn: A herbal medicine with various pharmacological effects. BioMed Res. Int. 2014; 2014, 569087.
MR Moein, K Zomorodian, K Pakshir, F Yavari, M Motamedi and MM Zarshenas. Trachyspermum ammi (L.) sprague: Chemical composition of essential oil and antimicrobial activities of respective fractions. J. Evid. Base. Integr. Med. 2014; 20, 50-6.
M Lateef, Z Iqbal, U Rauf and A Jabbar. Anthelmintic activity of Carum copticum seeds against gastrointestinal nematodes of sheep. J. Anim. Plant Sci. 2006; 16, 34-7.
B Ranjan, S Manmohan, SR Singh and RB Singh. Medicinal uses of Trachyspermum ammi: A Review. Pharmacologyonline 2012; 5, 247-58.
S Javed, AA Shahid, MS Haider, A Umeera, R Ahmad and M Sobia. Nutritional, phytochemical potential and pharmacological evaluation of Nigella sativa (Kalonji) and Trachyspermum ammi (Ajwain). J. Med. Plants Res. 2012; 6, 768-75.
E Stahl. Thin layer chromatography. A Laboratory hand book. 2nd ed. Springer Berlin, Heidelberg, Germany, 1969.
DM Steinberg, LJ Sokoll, KC Bowels, JH Nichols, R Roberts, SK Schultheis CM O,Donnell. Clinical evaluation of toxi-prepTM: A semiautomated solid-phase extraction system for screening of drugs in urine. Clin. Chem. 1997; 43, 2099-105.
MM Parvez, MA Rahman, MK Molla and A Akter. Compound isolation and purification by chromatographic method of stem bark of Anisoptera scaphula (Roxb.). Int. J. Pharmaceut. Sci. Rev. Res. 2012; 1, 1-6.
RS Alm, RJP Williams and A Tiselius. Gradient elution analyses I. A General Treatment. Acta Chem. Scand. 1952; 6, 826-36.
K Fossum and JR Whitaker. Simple method for detecting amylase inhibitors in biological materials. J. Nutr. 1974; 104, 930-36.
VP Cirillo. Mechanism of glucose transport across the yeast cell membrane. J. Bacteriol. 1962; 84, 485-91.
T Mosmann. Rapid colorimeter assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Meth. 1983; 65, 55-63.
S Sarasvathy. Antioxidant and insulin-like properties of extracts from wheat grains fermented by selected indigenous ganoderma spp / Sarasvathy Subramaniam. Master Thesis. University of Malaya, Kuala Lumpur, Malaysia.
JK Grover, S Yadav and V Vats. Medicinal plants of India with anti-diabetic potential. J. Ethnopharmacol. 2002; 81, 81-100.
M Jung, M Park, HC Lee, K Yoon-Ho, ES Kang and SK Kim. Antidiabetic agents from medicinal plants. Curr. Med. Chem. 2006; 13, 1203-18.
K Vijayalakshmi, C Immanuel Selvaraj, S Sundhu and P Arumugam. In vitro investigation of antidiabetic potential of selected traditional medicinal plants. Int. J. Phcog. Phytochemical Res. 2014; 6, 856-61.
N Anbu, V Musthafa and Velpandian. Anti-diabetic activity of polyherbal formulation aavaraiyathichurnam in alloxan induced diabetic rats. Int. J. Toxicol. Pharmacol. Res. 2012; 4, 77-80.
IV Asharani, M Gowtham, M Paridhavi and D Thirumalai. A comprehensive review on folklore antidiabetic plants. Int. J. Pharmaceut. Sci. Rev. Res. 2016; 39, 213-5.
FAVD Laar. Alpha-glucosidase inhibitors in the early treatment of type 2 diabetes. Vas. Heal. Risk. Managt. 2008; 4, 1189-95.
N Chiniwala and S Jabbour. Management of diabetes mellitus in the elderly. Curr. Opin. Endocrinol. Diabetes. Obes. 2011; 18, 148-52.
SB Gaikwad, GK Mohan and MS Rani. Phytochemicals for diabetes management. Pharmaceut. Crop. 2014; 5, 11-28.
G Brahmachari. Bio-flavonoids with promising antidiabetic potentials: A critical survey. In: VK Tiwari and BB Mishra (Eds.). Opportunity, Challenge and Scope of Natural Products in Medicinal Chemistry. 1st ed. Research Signpost, Ahmedabad, India, 2011, p. 187-212.
S Kumar, R Malhotra and D Kumar. Antidiabetic and free radicals scavenging potential of Euphorbia hirta Flower Extract. Indian J. Pharmaceut. Sci. 2010; 72, 533-7.
M Mosaddegh, F Naghibi, H Moazzenia, A Pirania and S Esmaeili. Ethnobotanical survey of herbal remedies traditionally used in Kohghiluyeh va Boyer Ahmad province of Iran. J. Ethnopharmacol. 2012; 141, 80-95.
A Gholamhoseinian, H Fallah, F Sharifi-far and M Mirtajeddini. The inhibitory effect of some Iranian plants extracts on the alpha glucosidase. Iranian J. Basic Med. Sci. 2008; 11, 1-9.
AAF Sima and S Chakrabarti. Long-term suppression of postprandial hyperglycaemia with acarbose retards the development of neuropathies in the BB/W-rat. Diabetologia 1992; 35, 325-30.
JM Carrascosa, JC Molero, Y Fermin, C Martinez, A Andres and J Satrustegui. Effects of chronic treatment with acarbose on glucose and lipid metabolism in obese diabetic wistar rats. Diabetes Obes. Metabol. 2001; 3, 240-8.
P McCue, YI Kwon and K Shetty. Anti-diabetic and anti-hypertensive potential of sprouted and solid-state bioprocessed soybean. Asia Pac. J. Clin. Nutr. 2005; 14, 145-52.
H Wagner and S Bladt. Plant drug analysis. A thin layer chromatography atlas. 2nd ed. Springer Berlin, Heidelberg, Germany, 1996.
SR Bale and AE Gawade. Screening of aqueous bark extracts of some plant species for α-amylase inhibitory activity. Bionano Frontier 2014; 7, 75-7.
G Daksha, Chandrashekar, R Lobo, Y Nayak and G Nilesh. In-vitro Antidiabetic activity of stem bark of Bauhinia purpurea Linn. Der Pharmacia Lettre 2012; 4, 614-9.
F Ahmed, S Sairam, A Urooj. Effect of various ayurvedic formulations and medicinal plants on carbohydrate hydrolyzing enzymes and glucose uptake by yeast cells-an in vitro study. J. Pharm. Res. 2009; 2, 563-8.
TG Scharff. Evidence for hexose transport in acetone-dried yeast. Arch. Biochem. Biophys. 1961; 95, 329-35.
MA Bhutkar, SD Bhinge, DS Randive and GH Wadkar. Hypoglycemic effects of berberis aristata and Tamarindus indica extracts in vitro. Bull. Facul. Pharm. Cairo Univ. 2017; 55, 91-4.
P Heera, L Inbathamizh and J Ramachandran. An invitro study on antidiabetic activity of different solvent extract from Alpinia galanga. Int. J. Ethnomedicine Pharmacol. Res. 2014; 2, 1-10.
DS Reddy, SR Avanapu and PH Ananth. In vitro antioxidant and glucose uptake effect of Trichodesma indicum in L-6 cell lines. Int. J. Pharma Bio Sci. 2012; 3, 810-9.
BS Ko, SB Choi, SK Park, JS Jang, YE Kim and S Park. Insulin sensitizing and insulinotropic action of Berberine from Cortidis Rhizoma. Biol. Pharmaceut. Bull. 2005; 28, 1431-7.
H Shittu, A Gray, B Furman and L Young. Glucose uptake stimulatory effect of akuammicine from Picralima nitida (Apocynaceae). Phytochemistry Lett. 2010; 3, 53-5.
BD Kumar, K Krishnakumar, SK Jaganathan and M Mandal. Effect of Mangiferin and Mahanimbine on glucose utilization in 3T3-L1 cells. Phcog. Mag. 2013; 9, 72-5.
Downloads
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.







