Schiff Bases Ligands Derived from o-Phthalaldehyde and Their Metal Complexes with Cu2+ and Ni2+: Synthesis, Anti-Breast Cancer and Molecular Docking Study
DOI:
https://doi.org/10.48048/tis.2023.5675Keywords:
Schiff base, Metal complexes, Molecular docking, Breast cancer, o-Phthalaldehyde, MTT, Breast cancer, MOE programAbstract
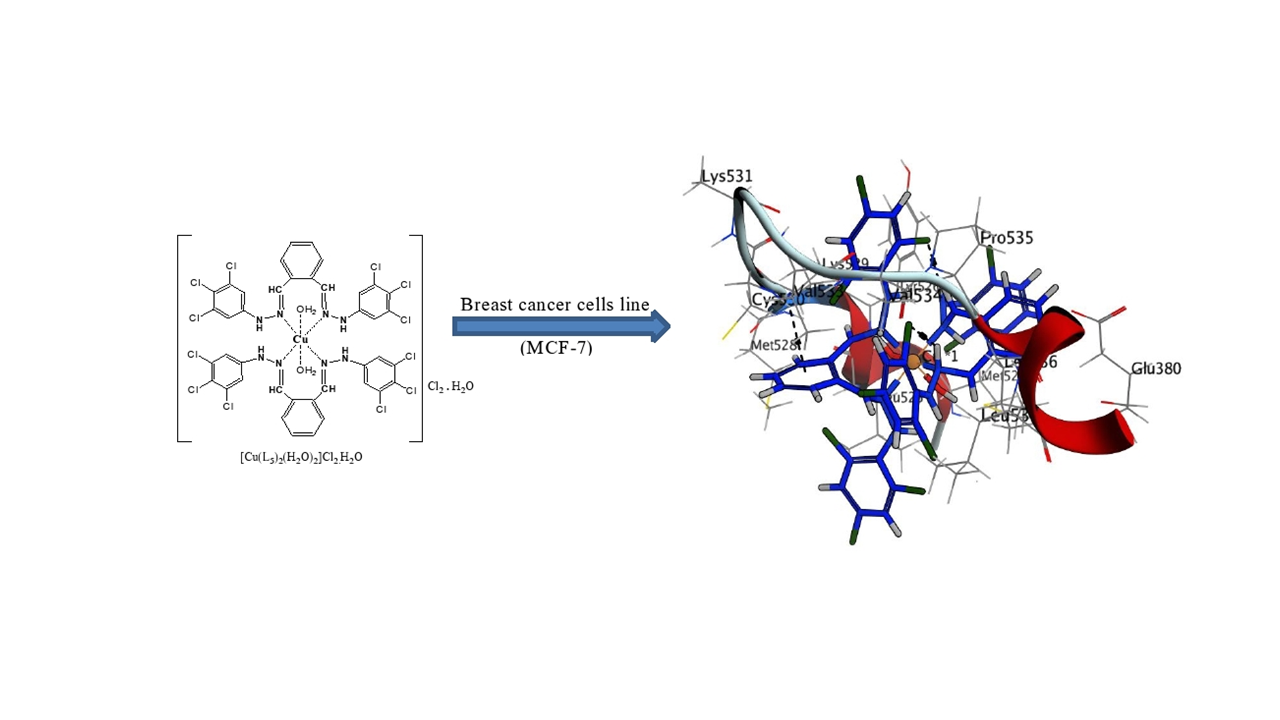
The Schiff bases and their complexes have an observed biological efficacy, so in the current study it has been prepared, characterized and evaluated the biological activity of some Schiff bases and their metal complexes with copper and nickel and based on ortho-phathaldehyde as a primary compound. A new Schiff bases ligands and metal complexes {[Cu(L)n(H2O)2]Cl2.mH2O and [Ni(L)n].mH2O, where L=N',N'''-((1E,1'E)-1,2-phenylenebis(methanylylidene)) di(benzohydrazide) L1, N',N'''-((1E,1'E)-1,2-phenylenebis (methanylylidene)) di(isonicotinohydrazide) L2, 1,2-bis((E)-(2-(2,4,6-trichlorophenyl)hydrazono)methyl) benzene L3 ; n = 1, 2 ; m = 0, 1, 3/2, 5/2} have been synthesized and elucidated by mass, FT-IR, 13C and 1HNMR, molar conductivity, flame-atomic absorption, magnetic susceptibility, Powder-XRD and TG analysis. The results showed that the L1 and L2 ligands behave as a tetra-dentate donor and were associated with metal ions in a molar ratio of 1:1, while L3 ligand was bi-dentate donor and associated with metal ions in 1:2 molar ratio. In addition, the geometric shapes of the prepared complexes were tetrahedral and square planar for Ni2+ and Zn2+ complexes, and octahedral for Cu2+ complexes. The effect of cellular toxicity in the laboratory has been examined by MTT assay for all compounds against the MCF-7 cancer breast cell line and found to have low efficacy except [Cu(L3)2(H2O)2] Cl2.H2O (5). The copper complex's molecular docking has been performed with breast cancer proteins using the MOE program, and found to target ERα, CDK6 and EGFR proteins by binding to hydrogen bonds and pi-interactions.
HIGHLIGHTS
- Synthesis new chemical compounds, which are some Schiff bases and their metal complexes with copper and nickel ions
- Characterization of chemical structure of synthesized compounds by mass, FT-IR, 13C and 1HNMR, molar conductivity, flame-atomic absorption, magnetic susceptibility, Powder-XRD and TG analysis
- Study the biological activity of synthesized compounds as anti-breast cancer
- Study of molecular docking of compounds that showed biological efficacy with proteins responsible for breast cancer cells
GRAPHICAL ABSTRACT
Downloads
References
FM Ahmed, SA Sadeek and WH El-Shwiniy. Synthesis, spectroscopic studies, and biological activity of some new N2O2 tetradentate schiff base metal complexes. Russ. J. Gen. Chem. 2019; 89, 1847-83.
AM Abu-Dief and IMA Mohamed. A review on versatile applications of transition metal complexes incorporating schiff bases. Beni Suef Univ. J. Basic Appl. Sci. 2015; 4, 119-33.
RH Al-Asadi, MK Mohammed and HK Dhaef. Mercuration and telluration of 2-fluoro-5-nitroaniline: Synthesis, antibacterial, and computational study. Russ. J. Gen. Chem. 2020; 90, 703-9.
WH El-Shwiniy, LM Abbass, SA Sadeek and WA Zordok. Synthesis, structure, and biological activity of some transition metal complexes with the mixed ligand of metformin and 1,4-diacetylbenzene. Russ. J. Gen. Chem. 2021; 91, 1774-82.
RH Al-Asadi, TA Fahad, BA Saeed and WA Al-Masoudi. Synthesis, characterization and antitumor activity of some new oganotellurium compounds containing azo group, part II. J. Adv. Chem. 2014; 8, 1464-71.
A Arunadevi and N Raman. Biological response of schiff base metal complexes incorporating amino acids - a short review. J. Coordination Chem. 2020; 73, 2095-116.
RH Al-Asadi Synthesis, DFT Calculation and Biological Activity of Some Organotellurium Compounds Containing Azomethine Group. Orbital Electron. J. Chem. 2019; 11, 402-10.
RG Deghadi, AE Elsharkawy, AM Ashmawy and GG Mohamed. Can one novel series of transition metal complexes of oxy-dianiline schiff base afford advances in both biological inorganic chemistry and materials science? Comm. Inorgan. Chem. 2022; 42, 1-46.
P Deveci, B Taner, E Özcan, Z Kılıç, M Karakaya and A Karakas. Structural characterization and second-order nonlinear optical behavior of metal complexes of ferrocene derivative. Russian J. Gen. Chem. 2019; 89, 330-8.
HA Abdullmajed, HA Sultan, RH Al-Asadi, QM Hassan, AA Ali and CA Emshary. Synthesis, DFT calculations and optical nonlinear properties of two derived Schiff base compounds from ethyl-4-amino benzoate. Phys. Scripta 2022; 97, 025809.
BM Ahmed, NA Rudell, I Soto and G Mezei. Reaction of amines with aldehydes and ketones revisited: Access to a class of non-scorpion ate tris(pyrazolyl)methane and related ligands. J. Org. Chem. 2017; 82, 10549-62.
OE Sherif and NS Abdel-Kader. DFT calculations, spectroscopic studies, thermal analysis and biological activity of supramolecular schiff base complexes. Arab. J. Chem. 2018; 11, 700-13.
AA Jambol, MSA Hamid, AH Mirza, MS Islam and MR Karim. Some novel schiff bases from pyruvic acid with amines containing N & S donor atoms: Synthesis, spectral studies and x-ray crystal structures. Int. J. Org. Chem. 2017; 7, 42-56.
T Sujeshwari, E Akila and P Maheswaran. DNA cleavage, antioxidant and antibacterial activity studies of new Schiff base Co(II), Ni(II), Cu(II) and Mn(II) complexes derived from o-phthalaldehyde. Eur. J. Mol. Clin. Med. 2020; 7, 3335-47.
H Köksal, M Dolaz, M Tümer and S Serin. Copper (II), cobalt (III), nickel (II), palladium (II), and zinc (II) complexes of the schiff base ligands derived from 2, 6-diacetylpyridine and phthaldialdehyde. Synth. Reactivity Inorg. Met. Org. Chem. 2001; 31, 1141-62.
Manju, D Kishore and D Kumar. Cadmium and tin complexes of schiff-base ligands. J. Coordination Chem. 2011; 64, 2130-56.
RK Mohapatra, PK Das, MK Pradhan, AA Maihub and MM El ajaily. Biological aspects of schiff base-metal complexes derived from benzaldehydes: An overview. J. Iran. Chem. Soc. 2018; 15, 2193-227.
N Harbeck, F Penault-Llorca, J Cortes, M Gnant, N Houssami, P Poortmans, K Ruddy, J Tsang and F Cardoso. Breast cancer. Nat. Rev. Dis. Primers 2019; 5, 66.
RH Al-Asadi, WA Al-Masoudi and KSA Al-Rassol. Synthesis, biological activity and computational study of some new unsymmetrical organotellurium compounds derived from (2-amino-5-carboxyphenyl) mercury(II) chloride. Asian J. Chem. 2016; 28, 1171-6.
SA Attique, M Hassan, M Usman, RA Muhammad, S Mahboob, KA Al-Ghanim, M Bilal and MZ Nawaz. A molecular docking approach to evaluate the pharmacological properties of natural and synthetic treatment candidates for use against hypertension. Int. J. Environ. Res. Public Health 2019; 16, 923.
FA Nawar, RH AL-Asadi and DS Abid. Synthesis, antibacterial activity and DFT calculations of some Thiazolidine-4-Carboxylic acid derivatives and their complexes with Cu(II), Fe(II) and VO(II). Egyptian J. Chem. 2020; 63, 349-62.
L Mitu, M Ilis, N Raman, M Imran and S Ravichandran. Transition metal complexes of Isonicotinoyl-hydrazone-4-diphenylaminobenzaldehyde: Synthesis, characterization and antimicrobial studies. J. Chem. 2012; 9, 298175.
S Velumani, X Mathew and PJ Sebastian. Structural and optical characterization of hot wall deposited CdSexTe1−x films. Sol. Energ. Mater. Sol. Cells 2003; 76, 359-68.
S Fani, B Kamalidehghan, KM Lo, NM Hashim, KM Chow and F Ahmadipour. Synthesis, structural characterization, and anticancer activity of a monobenzyltin compound against MCF-7 breast cancer cells. Drug Des. Dev. Ther. 2015; 23, 6191-201.
DH Fagan, LM Fettig, S Avdulov, H Beckwith, MS Peterson, YY Ho, F Wang, VA Polunovsky and D Yee. Acquired tamoxifen resistance in MCF-7 breast cancer cells requires hyperactivation of eIF4F-mediated translation. Hormones cancer 2017; 8, 219-29.
K Venugopal, H Ahmad, E Manikandan, KA Thanigai, K Kavitha, M Moodley, K Rajagopal, KR Balabhaskar and M Bhaskar. The impact of anticancer activity upon Beta vulgaris extract mediated biosynthesized silver nanoparticles (ag-NPs) against human breast (MCF-7), lung (A549) and pharynx (Hep-2) cancer cell lines. J. Photochemistry Photobiology B Biol. 2017; 173, 99-107.
C Dong, J Wu, Y Chen, J Nie and C Chen. Activation of PI3K/AKT/mTOR pathway causes drug resistance in breast cancer. Front. Pharmacol. 2021; 12, 628690.
DZ Mutlaq, AA Al-Shawi and RH Al-Asadi. Synthesis, characterization, anticancer activity, and molecular docking of novel maleimide-succinimide derivatives. Egyptian Pharmaceut. J. 2021; 20, 303-12.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2022 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






