Sperm Quality Assessment of White Shrimp (Litopenaeus Vannamei) Broodstock using Comet Assay
DOI:
https://doi.org/10.48048/tis.2023.4768Keywords:
Litopenaeus vanamei, Captive breeding, Comet assay, Sperm quality assessment, DNA damageAbstract
The quality of sperms is considered one of the factors determining fertilization success of white shrimp, Litopenaeus vannamei. Generally, conventional methods such as sperm count and viability are used for determining sperm quality. Occasionally, the fertilization rate does not correlate with what conventional parameters indicate. Presently, it has become increasingly evident that sperm DNA damage is a potential cause of an unexplained fertilization incompetence of the sperm. Therefore, the present study aimed to determine the feasible application of the comet assay for measuring sperm quality from different male broodstock and to analyze the correlation between sperm DNA integrity and fertilization efficacy of the shrimp. Male and female shrimp sampled from 4 different broodstock ponds were mated in experimental tanks (n = 5). Sperm were collected and subjected to alkaline comet assay. Sperm count, sperm viability, and Gonadosomatic index(GSI) were analyzed for comparison. Viability of the obtained nauplii were recorded and used as larvae production efficiency. The results revealed that nauplius numbers were significantly different among treatments (6.78±1.61×104, 12.18±1.17×104, 16.76±0.97×104 and 21.38±1.14×104 cells/spawn in treatment 1, 2, 3 and 4, respectively), indicating the different fertilization rate between treatments. The average body weights, lengths, and GSIs of broodstock were not significantly different, indicating that the morphological factors were not major causes of the different nauplii production among treatments. The results of comet measurements clearly showed that the levels of comet parameters correlated with the numbers of nauplii obtained from each treatment while the conventional sperm analyses were unable to detect the different quality of the sperm among treatments. This indicates that the degree of sperm DNA damage correlates with the fertilization rate of the shrimp and can be used as a sensitive indicator for determining sperm quality of broodstock shrimp.
HIGHLIGHTS
- Sperm DNA damage is one of the potential causes of low fertilization rate in L. vannamei captive breeding
- Comet assay is used to assess the degree of DNA damage in a single cell such as shrimp sperm cell using electrophoresis technique
- Comet assay is more sensitive to detect the sperm integrity among individual L. vannamei broodstock when compared to morphological methods
- Sperm DNA damage can be used as a sensitive indicator for determining the sperm quality of L. Vannamei
GRAPHICAL ABSTRACT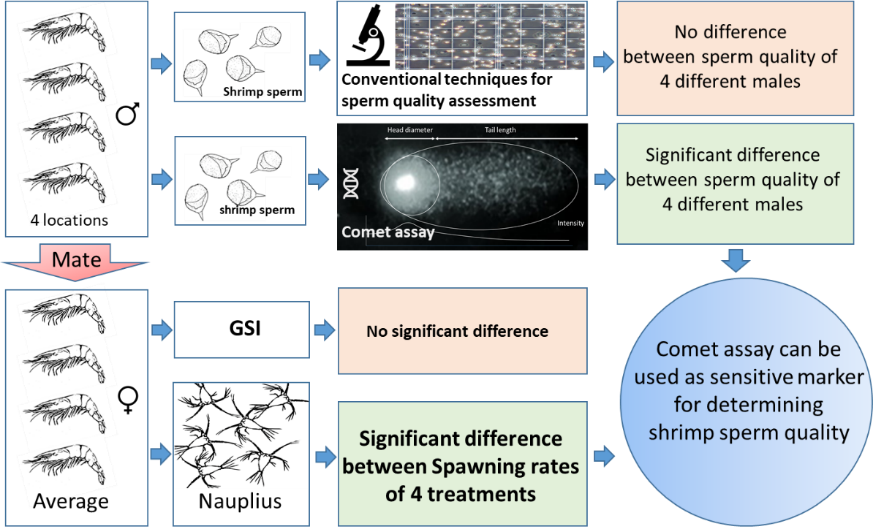
Downloads
References
A Albalat, S Zacarias, CJ Coates, DM Neil and SR Planellas. Welfare in farmed decapod crustaceans, with particular reference to Penaeus vannamei. Front. Mar. Sci. 2022; 9, 886024.
M Briggs, S Funge-Smith, R Subasinghe and M Phillips. Introductions and movement of Penaeus vannamei and Penaeus stylirostris in Asia and the Pacific. FAO Regional Office for Asia and the Pacific, Bangkok, Thailand, 2004, p. 1-12.
JC Sainz-Hernández, IS Racotta, S Dumas and J Hernández-López. Effect of unilateral and bilateral eyestalk ablation in Litopenaeus vannamei male and female on several metabolic and immunologic variables. Aquaculture 2008; 283, 188-93.
J Wyban. Global seafood alliance, Thailand’s white shrimp revolution, Available at: https://www.aquaculturealliance.org/advocate/thailands-white-shrimp-revolution, accessed June 2007.
V Alday-Sanz, J Brock, TW Flegel, R McIntosh, MG Bondad-Reantaso, M Salazar and R Subasinghe. Facts, truths and myths about SPF shrimp in aquaculture. Rev. Aquac. 2018; 12, 76-84.
GJ Coman, SJ Arnold, MJ Jones and NP Preston. Effect of rearing density on growth, survival and reproductive performance of domesticated Penaeus monodon. Aquaculture 2007; 264, 175 -83.
F Andriantahina, XL Liu, H Huang, JH Xiang and CM Yang. Comparison of reproductive performance and offspring quality of domesticated Pacific white shrimp, Litopenaeus vannamei. Aquaculture 2012; 324, 194-200.
J Alfaro-Montoya, A Braga and R Umana-Castro. Research frontiers in penaeid shrimp reproduction: Future trends to improve commercial production. Aquaculture 2019; 503, 70-87.
O Meunpol, S Iam-Pai, W Suthikrai and S Piyatiratitivorakul. Identification of progesterone and 17 alpha-hydroxyprogesterone in polychaetes (Perinereis sp.) and the effects of hormone extracts on penaeid oocyte development In vitro. Aquaculture 2007; 270, 485-92.
O Meunpol, E Duangjai, R Yoonpun and S Piyatiratitivorakul. Detection of prostaglandin E2 in polychaete Perinereis sp. and its effect on Penaeus monodon oocyte development in vitro. Fish. Sci. 2010; 76, 281-6.
I Yano, B Tsukimura, JN Sweeney and J Wyban. Induced ovarian maturation of Penaeus vannamei by implantation of lobster ganglion. J. World Aquac. Soc. 2007; 19, 204-9.
Y Tinikul, J Poljaroen, R Tinikul, P Anuracpreeda, C Chotwiwatthanakun, N Senin, T Poomtong, PJ Hanna and P Sobhon. Effects of gonadotropin-releasing hormones and dopamine on ovarian maturation in the Pacific white shrimp, Litopenaeus vannamei, and their presence in the ovary during ovarian development. Aquaculture 2014; 420-421, 79-88.
T Alnawafleh, BK Kim, HE Kang, TH Yoon and HW Kim. Stimulation of molting and ovarian maturation by methyl Farnesoate in the pacific white shrimp Litopenaeus vannamei (Boone, 1931). Fish. Aquatic Sci. 2014; 17, 115-21.
E Duangjai, W Suthikrai, N Puanglarp, P Srisapoome, P Unagul, O Meunpol. Effect of salinity on previtellogenic development of female pacific white shrimp (Litopenaeus vannamei Boone, 1931). KU. Fish. Res. Bull. 2015; 39, 15-29.
J Alfaro-Montoya. The reproductive conditions of male shrimps, genus Penaeus, sub-genus Litopenaeus (open thelyca penaeoid shrimps): A review. Aquaculture 2010; 300, 1-9.
J Alfaro-Montoya and L Hernández. The histological structure of the androgenic gland and cellular cord of the male reproductive system of adult Litopenaeus and Rimapenaeus byrdi. J. Crustac. Biol. 2012; 32, 351-7.
S Aungsuchawan, CL Browdy and B Withyachumnarnkul. Sperm capacitation of the shrimp Litopenaeus vannamei. Aquac. Res. 2011; 42, 188-95.
AL Braga, DLA Lopes, LH Poersch and W Wasielesky. Spermatophore and sperm quality of the pink shrimp Farfantepenaeus paulensis fed with fresh food supplemented with pollen and paprika. Aquaculture 2013; 380, 29-32.
V Fransozo, AB Fernandes, LS López-Greco, FJ Zara and DC Santos. Functional morphology of the male reproductive system of the white shrimp Litopenaeus schmitti (Burkenroad, 1936) (Crustacea, Penaeidea) compared to other Litopenaeus. Invertebr. Reprod. Dev. 2016; 60, 161-74.
MM Harlıoğlua, A Farhadia and S Gürb. Determination of sperm quality in decapod crustaceans Aquaculture 2018; 490, 185-93.
G Vázquez-Islas, IS Racotta, A Robles-Romo, R Campos-Ramos. Energy balance of spermatophores and sperm viability during the molt cycle in intact and bilaterally eyestalk ablated male pacific white shrimp Litopenaeus vannamei. Aquaculture 2013; 414-415, 1-8.
R Leelatanawit, U Uawisetwathana, J Khudet, A Klanchui, S Phomklad, S Wongtripop, P Angthoung, P Jiravanichpaisal, N Karoonuthaisiri. Effects of polychaetes (Perinereis nuntia) on sperm performance of the domesticated black tiger shrimp (Penaeus monodon). Aquaculture 2014; 433, 266-75.
J Alfaro-Montoya. The effect of Ibuprofen on female and male reproduction of the open thelyca marine shrimp, Litopenaeus. Aquac. Res. 2015; 46, 105-16.
JC Pérez‐Rodríguez, J Gómez‐Gutiérrez, LS López‐Greco and E Cortés‐Jacinto. Spermatophore production and sperm quality of the river prawn Macrobrachium americanum spence bate, 1868 fed with different diets. Aquac. Res. 2019; 50, 117-3129.
MF Uberti, FN Vieira, HR Salência, GS Vieira and LA Vinatea. Assessment of viability of sperm cells of Litopenaeus vannamei on cryopreservation. Braz. Arch. Biol. Tech. 2014; 57, 374-80.
MT Botelho, N Fuller, M Vannuci-Silva, G Yang, K Richardson and AT Ford. Unusual male size vs sperm count relationships in a coastal marine amphipod indicate reproductive impairment by unknown toxicants Aquat. Toxicol. 2021; 233, 105793.
M Lezcano, C Granja and M Salazar. The use of flow cytometry in the evaluation of cell viability of cryopreserved sperm of the marine shrimp (Litopenaeus vannamei). Cryobiology 2004; 48, 349-56.
MM Harlıoğlu, F Kutluyer and S Gür. An investigation on the sperm number and reproductive parameters of males in wild caught freshwater crayfish (Astacus leptodactylus, Eschscholtz). Anim. Biol. 2017; 62, 409-18.
B Pratoomchat, S Piyatiratitivorakul, P Menasveta and AW Fast. Sperm quality of pond-reared and wild-caught Penaeus monodon in Thailand. J. World Aquac. Soc. 1993; 24, 530-40.
BP Ceballos Vázquez, C Rosas and IS Racotta.Sperm quality in relation to age and weight of white shrimp Litopenaeus vannamei. Aquaculture 2003; 228, 141-51.
SG Jiang, JH Huang, FL Zhou, X Chen, QB Yang, WG Wen and ZM Ma. Observations of reproductive development and maturation of male Penaeus monodon reared in tidal and earthen ponds. Aquaculture 2009; 292, 121-8.
HW Bakos, JG Thompson, D Feil and M Lane. Sperm DNA damage is associated with assisted reproductive technology pregnancy. Int. J. Androl. 2008; 31, 518-26.
S Perez-Cerezales, S Martinez-Paramo, J Beirao and MP Herraez. Evaluation of DNA damage as a quality marker for rain trout sperm cryopreservation and use of LDL as cryoprotectant. Theriogenology 2010; 74, 282-9.
D Sakkas and JG Alvarez. Sperm DNA Fragmentation: Mechanisms of origin, impact on reproductive outcome, and analysis. Fertil. Steril. 2010; 93, 1027-36.
SJ Arnold, GJ Coman, C Burridge and M Rao. A novel approach to evaluate the relationship between measures of male fertility and egg fertilization in Penaeus monodon. Aquaculture 2012; 338-341, 181-9.
SJ Arnold, GJ Coman and M Emerenciano. Constraints on seed stock production in eighth generation domesticated Penaeus monodon broodstock. Aquaculture 2013; 410-411, 95-100.
T Feng, J Gosálvez, C Lopez-Fernandez, F Arroyo, B Paterson and S Johnston. Sperm chromatin dispersion test (SCDt) for the assessment of sperm DNA fragmentation in black tiger prawn, Penaeus monodon. Aquaculture 2018; 491, 281-8.
J Rank, K Jensen and PH Jespersen. Monitoring DNA damage in indigenous blue mussels (Mytilus edulis) sampled from coastal sites in Denmark. Genet. Toxicol. Environ. Mutagen. 2005; 585, 33-42.
AN Jha. Ecotoxicological applications and significance of the comet assay. Mutagenesis 2008; 23, 207-21.
A Shaliutina, M Hulak, I Gazo, P Linhartova and O Linhart. Effect of short-term storage on quality parameters, DNA integrity, and oxidative stress in Russian (Acipenser gueldenstaedtii) and Siberian (Acipenser baerii) sturgeon sperm. Anim. Reprod. Sci. 2013; 139, 127-35.
HC Reinardy, E Skippins, TB Henry and AN Jha. Assessment of DNA damage in sperm after repeated non-invasive sampling in zebrafish Danio rerio. J. Fish Biol. 2013; 82, 1074-81.
H Kobayashi, C Sugiyama, Y Morikawa, M Hayashi and T Sufuni. Comparison between manual microscopic analysis and computerised image analysis in single cell gel electrophoresis assay. MMS Commun. 1995; 3, 103-15.
TS Kumaravel, B Vilhar, SP Faux and AN Jha. Comet assay measurements: A perspective. Cell Biol. Toxicol. 2009; 25, 53-64.
C Lewis and T Galloway. Reproductive consequences of paternal genotoxin exposure in marine invertebrates. Environ. Sci. Tech. 2009; 43, 928-33.
E Lacaze, O Geffard, S Bony and A Devaux. Genotoxicity assessment in the amphipod Gammarus fossarum by use of the alkaline Comet assay. Mut. Res-Gen. Toxicol. Environ. Mutagen. 2010; 700, 32-8.
C Lewis and AT Ford. Infertility in male aquatic invertebrates: A review. Aquat. Toxicol. 2012; 120-121, 79-89.
AB Noor- Hidayati, MS Shahreza, AB Abol- Munafi and M Ikhwanuddin. Sperm quality assessment of banana shrimp Fenneropenaeus merguiensis (De Man, 1888) from ultraviolet irradiation for initial development of gynogenesis application. J. Fish. Aquat. Sci. 2014; 9, 187-96.
AP Pawar, SV Sanaye, S Shyama, RA Sreepada, J Bhagat, P Kumar and RDS Khandeparker. In vivo DNA damage in gill, haemolymph and muscle cells of whiteleg shrimp Litopenaeus vannamei on exposure to organophosphorus pesticide. Aqua. Environ. Int. 2019; 11, 75-86.
BP Ceballos-Vázquez, C Rosas and IS Racotta. Sperm quality in relation to age and weight of white shrimp Litopenaeus vannamei. Aquaculture 2003; 228, 141-51.
AB Bugnot and LS López-Greco. Sperm production in the red claw crayfish Cherax quadricarinatus (Decapoda, Parastacidae). Aquaculture 2009; 295, 292-9.
MM Harlıoğlu, K Köprücü, AG Harlıoğlu, SM Yonar, TC Duran, MN Cakmak, Ö Aksu, S Özcan, F Kutluyer and H Gündoğdu. Effect of dietary n-3 series fatty acids on sperm production in the freshwater crayfish, Astacus leptodactylus (Eschscholtz) (Astacidae). Aqua. Int. 2013; 21, 273-82.
NP Singh, MT McCoy, RR Tice and EL Schneider. A simple technique for quantitation of low levels of DNA damage in individual cells. Exp. Cell Res. 1988; 175, 184-91.
H Migaud, G Bell, E Cabrita, B McAndrew, A Davie, J Bobe, MP Herráez and M Carrillo. Gamete quality and broodstock management in temperate fish. Rev. Aqua. 5 2013; 5, S194-S223.
BE Felgenhauer and LG Abele. Morphological diversity of decapod spermatozoa. In: RT Bauer and JW Martin (Eds.). Crustacean sexual biology. Columbia University Press, New York, 1991, p. 322- 41.
BGM Jamieson. Ultrastructure and phylogeny of crustacean spermatozoa. Mem. Queensl. Mus. 1991; 31, 109-42.
BGM Jamieson and CC Tudge. Crustacea-decapoda. In: BGM Jamieson (Ed.). Reproductive biology of invertebrates. Volume IX. Progress in male gamete ultrastructure and phylogeny part C. John Wiley & Sons, Chichester, 2000, p. 1- 95.
CC Tudge. Spermatozoal morphology and its bearing on decapod phylogeny. In: JW Martin, KA Crandall and DL Felder (Eds.). Decapod crustacean phylogenetics. Taylor and Francis/CRC Press, Boca Raton, 2009, p. 101-19.
WH Clark, MC Kleve and AI Yudin. An acrosome reaction in natantian sperm. J. Exp. Zool. 1981; 218, 279-91.
FJ Griffin, K Shigekawa and WH Clark. Formation and structure of the acrosomal filament in the sperm of Sicyonia ingentis. J. Exp. Zool. 1988; 246, 94-102.
J Beirão, M Boulais, V Gallego, JK O’Brien, S Peixoto, TR Robeck and E Cabrita. Sperm handling in aquatic animals for artificial reproduction. Theriogenology 2019; 133, 161-78.
BP Ceballos Vázquez, B Aparicio-Simon, E Palacios and IS Racotta. Sperm quality over consecutive spermatophore regenerations in the pacific white shrimp Litopenaeus vannamei. J. World Aquac. Soc. 2004; 35, 178-88.
A Erraud, M Bonnard, A Duflot, A Geffard, JM Danger, J Forget-Leray and B Xuereb. Assessment of sperm quality in palaemonid prawns using Comet assay: methodological optimization. Environ. Sci. Pollut. Res. 2018; 25, 11226-37.
PL Olive. Cell proliferation as a requirement for development of contact effect in Chinese hamster V79 spheroids. Radiat. Res. 1989; 117, 79-92.
RR Tice, E Agurell, D Anderson, B Burlinson, A Hartmann, H Kobayashi, Y Miyamae, E Rojas, JC Ryu and YF Sasaki. The single cell gel /comet assay: Guidelines for in vitro and in vivo genetic toxicology testing. Environ. Mol. Mutagen. 2000; 35, 206-21.
G Speit and A Hartmann. The comet assay: A sensitive genotoxicity test for the detection of DNA damage and repair. Methods Mol. Biol. 2006; 314, 275-86.
AR Collins, AA Oscoz, G Brunborg, I Gaivão, L Giovannelli, M Kruszewski, CC Smith and R Stetina. The comet assay: Topical issues. Mutagenesis 2008; 23, 143-51.
B Burlinson, RR Tice, G Speit, E Agurell, SY Brendler-Schwaabe, AR Collins, P Escobar, M Honmah, TS Kumaravel, M Nakajima, YF Sasaki, V Thybaud, Y Uno, M Vasquez and A Hartmannoa. Fourth international workgroup on genotoxicity testing: Results of the in vivo comet assay workgroup. Mutat. Res. 2007; 627, 31-5.
Y Hong, HJ Han, H Lee, D Lee, J Ko, Z Hong, JY Lee, JH Seok, HS Lim, WC Son and I Sohn. Deep learning method for comet segmentation and comet assay image analysis. Sci. Rep. 2020; 10, 18915.
Y Lorenzo, S Costa, AR Collins and A Azqueta. The comet assay, DNA damage, DNA repair and cytotoxicity: Hedgehogs are not always dead. Mutagenesis 2013; 28, 427-32.
S Braafladt, V Reipa and DH Atha. The comet assay: Automated imaging methods for improved analysis and reproducibility. Sci. Rep. 2016; 6, 32162.
P Møller, A Azquet, E Boutet-Robinet, G Koppen, S Bonassi, M Milić, G Gajski, S Costa, JP Teixeira, CC Pereira, M Dusinska, R Godschalk, G Brunborg, KB Gutzkow, L Giovannelli, MS Cooke, E Richling, B Laffon, V Valdiglesias, N Basaran, CD Bo, B Zegura, M Novak, H Stopper, P Vodicka, S Vodenkova, VMD Andrade, M Sramkova, A Gabelova, A Collins and SAS Langie. Minimum information for reporting on the comet assay (MIRCA): Recommendations for describing comet assay procedures and results. Nat. Protoc. 2020; 15, 3817-26.
G Brunborg and A Collins. Guidance for publishing comet assay results. Genet. Toxicol. Environ. Mutagen. 2020; 854-855, 503146.
AR Collins, A Ai-guo and SJ Duthie. The kinetics of repair of oxidative DNA damage (strand breaks and oxidised pyrimidines) in human cells. Mutat. Res. 1995; 336, 69-77.
H Kobayashi, C Sugiyama, Y Morikawa, M Hayashi and T Sufuni. Comparison between manual microscopic analysis and computerised image analysis in single cell gel electrophoresis assay. MMS Commun. 1995; 3, 103-15.
TS Kumaravel, B Vilhar, SP Faux and AN Jha. Comet Assay measurements: A perspective. Cell Biol. Toxicol. 2009; 25, 53-64.
PL Olive and JP Banath. Sizing highly fragmented DNA in individual apoptotic cells using the comet assay and a DNA crosslinking agent. Exp.Cell Res.1995; 221, 19-26.
PL Olive, JP Banath and RE Durand. Heterogeneity in radiation induced DNA damage and repair in tumor and normal cells measured using the “Comet” assay. Radiat. Res. 1990; 122, 86-94.
AK Zhanataev, EA Anisina, ZV Chayka, IA Miroshkina and AD Durnev. The phenomenon of atypical DNA comets. Cell Tissue Biol. 2017; 11, 286-92.

Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2022 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






