Hydrogen Generation During Thermal Processes of Water Decomposition on the Surface of Nano-ZrO2+3mol.%Y2O3
DOI:
https://doi.org/10.48048/tis.2023.4684Keywords:
Nano-ZrO2 3mol.%Y2O3, Hydrogen generation, Kinetics, Thermal processes, Adsorption, desorptionAbstract
The study of physicochemical properties of nano-ZrO2+3mol.%Y2O3 was determined. The X-ray diffraction spectrum of the nano-ZrO2 and system nano-ZrO2+3 mol.%Y2O3 compound was drawn by the Ritveld method and the crystal structure was determined at different temperatures (T = 373, 473, 573, 673 K) and under normal conditions. The energy of the arrangement of H2 as a result of the decay of H2O, on the surface of nano-ZrO2+3mol.%Y2O3 was considered. Impacts of adsorption and desorption preparation of ZrO2+3mol.%Y2O3 nanoparticles were considered at diverse (T = 373, 473, 573, 673 K) temperatures. The thought of H2 in warm forms at nano-ZrO2+3mol.%Y2O3 framework expanded. This tells about hydrogen era by water part in close future. Nano-ZrO2 and system nano-ZrO2+3mol.%Y2O3 with the temperature range of T = 373, 473, 573, 673 K have been studied by SEM analysis. These results are promising of hydrogen generation by water splitting in near future.
HIGHLIGHTS
- First innovative research article in this area
- Importance of the thermal in water splitting for hydrogen generation
- Design and optimization of water splitting for hydrogen generation
- Future of hydrogen generation at a large scale economically
GRAPHICAL ABSTRACT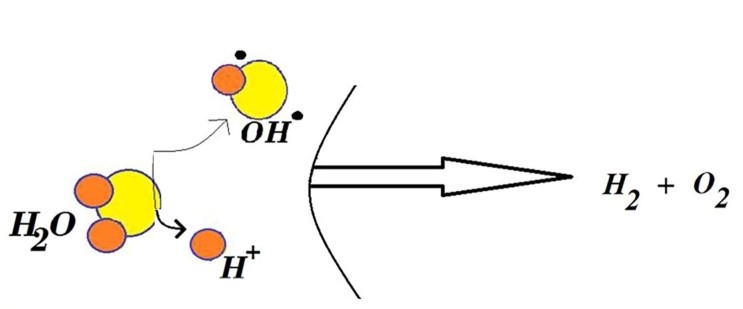
Downloads
References
A Basheer and I Ali. Water photo splitting for green hydrogen energy by green nanoparticles. Int. J. Hydro. Energ. 2019; 44, 11564-72.
G Imanova, T Agayev and S Jabarov. Investigation of structural and optical properties of zirconia dioxide nanoparticles by radiation and thermal methods. Mod. Phys. Lett. B 2021; 34, 2150050.
T Agayev, G Imanova and A Aliyev. Influence of gamma radiation on current density and volt-ampere characteristics of metallic zirconium. Int. J. Mod. Phys. B 2022; 36, 2250115.
I Ali, G Imanova, A Garibov, T Agayev, S Jabarov, A Almalki and A Alsubaie. Gamma rays mediated water splitting on nano-ZrO2 surface: Kinetics of molecular hydrogen formation. Radiat. Phys. Chem. 2021; 46, 109431.
T Agayev, S Musayeva and G Imanova. Studying the Kinetics of Formation of Molecular Hydrogen during the Radiolysis of Hexane and a Mixture of C6H14–H2O on a Surface of n-ZrO2. Russ. J. Phys. Chem. A 2021; 24, 270-2.
I Ali, G Imanova, X Mbianda and O Alharbid. Role of the radiations in water splitting for hydrogen generation. Sustain. Energ. Tech. Assess. 2022; 51, 101926.
G Imanova, T Agaev, A Garibov, S Melikova, S Jabarov and H Akhundzada. Radiation-thermocatalytic and thermocatalytic properties of n-ZrO2-n-SiO2 systems in the process of obtaining hydrogen from water at different temperatures. J. Mol. Struct. 2021; 36, 130651.
G Imanova. Kinetics of radiation-heterogeneous and catalytic processes of water in the presence of zirconia nanoparticles. Adv. Phys. Res. 2020; 2, 94-101.
G Imanova and M Kaya. Importance of the radiations in radiolysis processes for hydrogen generation. Generis publishing, Chișinău, Republic of Moldova, 2021.
I Ali, G Imanova, M Locatelli, HM Albishri, WH Alshitari, MN Siddiqui and AM Hameed. An ionic-liquid-imprinted nanocomposite adsorbent: Simulation, kinetics and thermodynamic studies of triclosan endocrine disturbing water contaminant removal. Molecules 2022; 27, 5358.
DE Karakaş, G Imanova, M Akdemir, HD Kıvrak, S Horoz and M Kaya. Biomass-based metal-free catalyst as a promising supercapacitor electrode for energy storage. J. Mater. Sci. Mater. Electron. 2022; 33, 18111-23.
T Agaev, G Imanova and S Melikova. Study of the radiation-thermal decomposition of water into nano-ZrO2 + H2O by IR spectroscopy. J. Chem. High Energ. 2014; 48, 281-5.
T Agayev, G Imanova and S Melikova. An IR spectroscopic study of the effect of gamma radiation on the n-ZrO2+n-SiO2+H2O systems. Surf. Physicochem. Protect. Mater. 2018; 54, 813-6.
G Imanova. Molecular hydrogen production by radiolysis of water on the surface of nano-ZrO2 under the influence of gamma rays. Synth. Sinter. 2022; 2, 9-13.
S Basahel, I Ali, M Mokhtar and K Narasimha. Influence of crystal structure of nanosized ZrO2 on photocatalytic degradation of methyl orange. Nanoscale Res. Lett. 2015; 10, 65-73.
S Mekhrdod, K Kholmirzo, D Aleksandr, A Elmar, Y Tomoyuki, L Andrei, A Valer and A Madadzada. Density functional theory calculations of the water interactions with ZrO2 nanoparticles Y2O3 doped. J. Phys. Conf. Ser. 2018; 994, 012013.
R Hashimov, N Ismayilova, F Mikailzade, A Dashdemirov, A Trukhanov, S Trukhanov, Y Aliyev, E Asgerov, S Jabarov amd N Dang. Electronic structure and density of states in hexagonal BaMnO3. Mod. Phys. Lett. B 2018; 32, 1850186.
M Yashima, S Sasaki, M Kakihana, Y Yamaguchi, H Arashi and M Yoshimura. Oxygen-induced structural change of the tetragonal phase around the tetragonal-cubic phase boundary in ZrO2-YO1.5 solid solutions. Acta Cryst. B 1994; 50, 663-72.
Y Han and J Zhu. Surface science studies on the zirconia-based model catalysts. Top. Catal. 2013; 56, 1525-41.
AS Doroshkevich, EB Asgerov, AV Shylo, AI Lyubchyk, AI Logunov, VA Glazunova, AK Islamov, VA Turchenko, V Almasan, D Lazar, M Balasoiu, VS Doroshkevich, AI Madadzada, KT Kholmurodov, VI Bodnarchuk and BL Oksengendler. Direct conversion of the water adsorption energy to electricity on the surface of zirconia nanoparticles. Appl. Nanosci. 2019; 9, 1603-9.
J LaVerne and L Tandon. H2 Production in the radiolysis of water on CeO2 and ZrO2. J. Phys. Chem. B 2002; 106, 380-6.
JL Verne. H2 Formation from the radiolysis of liquid water with zirconia. J. Phys. Chem. B 2005; 109, 5395-7.
S Caër and P Rotureau. Radiolysis of confined water: Hydrogen production at a high dose rate. ChemPhysChem 2005; 12, 2585-96.
H Mahmudov, T Suleymanov, Z Sabzaliyeva, G Imanova, H Akhundzada, K Azizova, S Hasanova and S Hasanov. Kinetic ınteraction of hexan conversion and oxidation on the surface of an Al2O3 nanocatalyzer at room temperature under the effect of gamma radiation. J. Chem. 2021; 2021, 9493765.
E Asgerov, A Doroshkevich, A Madadzada, A Beskrovnyy and E Popov. Martensitic phase transition in yttrium (0 %, 3 %, 8 %)- stabilized ZrO2 nanopowders by adsorption of water. In: Procedings of the 21st International Summer School on Vacuum, Electron and Ion Technologies, Bulgaria. 2019.
E Asgerov. Density functional theory calculations of the water interactions with ZrO2 nanoparticles Y2O3 doped. J. Phys. Conf. Ser. 2018; 994, 012013.
E Asgerov. The effect of percolation electrical properties in hydrated nanocomposite systems based on polymer sodium alginate with a filler in the form nanoparticles ZrO2 - 3mol%Y2O3. Adv. Phys. Res. 2019; 1, 70-80.
T Konstantinova, I Danilenko, V Glazunova, G Volkova and O Gorban. Mesoscopic phenomena in oxide nanoparticles systems: Processes of growth. J. Nanopart. Res. 2011; 13, 4015-23.
Z Tursunmetova, G Imanova, I Bekpulatov, Method For Low-Temperature Vacuum-Thermal Cleaning of Surface Single Crystals Si and GaAs, J. Polytec. 2022; 25, 921-927.
I Ali, G Imanova, T Agayev, A Aliyev, S Jabarov, HM Albishri, WH Alshitari, AM Hameed, A Alharbi. Seawater splitting for hydrogen generation using zirconium and its niobium alloy under gamma radiation. Molecules 2022; 27, 6325.
M Normuradov, I Bekpulatov, G Imanova, B Igamov, Structures for constructing devices from formed Mn4Si7 and CoSi films, Adva. Phys. Rese. 4; 2022, 142-154.
I Ali, T Agayev, GT Imanova, A Aliyev, OML Alharbi, A Alsubaie and ASA Almalki. A comparison of hydrogen production by water splitting on the surface of α-, - and γ-Al2O3. Chemistry Select 2022; 7, e202202618.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2022 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






