Screening In silico Antidepressant Activity of Aqueous Extract of Leaves of Rumex acetosa L.
DOI:
https://doi.org/10.48048/tis.2023.4664Keywords:
Sorrel, FTIR, HPLC, GCMS, Docking study, Rhein, Physcion, EmodinAbstract
The main objective of this study is to obtain extracts of Rumex acetosa leaves using various solvents, characterize these extracts and finally evaluate in silico antidepressant activity. Rumex acetosa belongs to polygonaceae family and is often called sorrel. This plant’s leaves were gathered and extracted using a variety of solvents including petroleum ether, ethyl acetate, ethanol, chloroform, and ultimately water. All these extracts are explored to check for various phytoconstituents by commonly used phytochemical screening methods. It was found that aqueous extract of Rumex acetosa (AERA) contained many phytoconstituents like alkaloids, flavonoids, glycosides, and anthocyanins. This aqueous extract was further screened by FTIR, HPLC, and GCMS analysis techniques. In FTIR analysis, aqueous extract of Rumex acetosa showed peaks at 1163.11and 1060.88 cm−1corresponding to anthocyanins. HPLC analysis showed the presence of peaks with the retention time of 6.75, 18.94, 25.19, 28.04, 34.72 and 39.37 min corresponding to sennoside, aloe-emodin, rhein, emodin, chrysophanol, and physcion respectively. In GCMS analysis, peaks at 18.15, 18.50, 20.32, 20.48 and 24.99 correspond to rhein, dibutyl phthalate, emodin, octadecanoic acid, and chrysophanol respectively. Five phytoconstituents - aloe-emodin, chrysophanol, emodin, physcion, and rhein - are further assessed for in silico antidepressant effectiveness based on characterization data. As rhein has the highest negative value of glide energy, it indicates a stronger binding affinity for MAO-A than all other screened constituents. Thus, the study concludes regarding in silico anti-depressant activity against MAO-A. Further, present research puts forth a way to carry out the isolation of therapeutically proven phytoconstituents and to evaluate these constituents for their biological activity.
HIGHLIGHTS
- In FTIR analysis, absorption peaks belonging to C=C, C-H aromatic ring and O-H bonds were found at 1,467-1,431 cm−1, 3,049 and 3,162.71 cm−1 respectively in AERA
- HPLC analysis showed the presence of peaks with the retention time of 6.75, 18.94, 25.19, 28.04, 34.72 and 39.37 min corresponding to sennoside, aloe-emodin, rhein¸ emodin, chrysophanol, and physcion, respectively
- In GCMS analysis, peaks at 18.15, 18.50, 20.32, 20.48 and 24.99 correspond to rhein, dibutyl phthalate, emodin, octadecanoic acid, and chrysophanol, respectively
- Five phytoconstituents viz., aloe-emodin, chrysophanol, emodin, physcion, and rhein, are assessed for in silico antidepressant effectiveness and rhein has the highest negative value of glide energy, it indicates a stronger binding affinity for MAO-A than all other screened constituents
GRAPHICAL ABSTRACT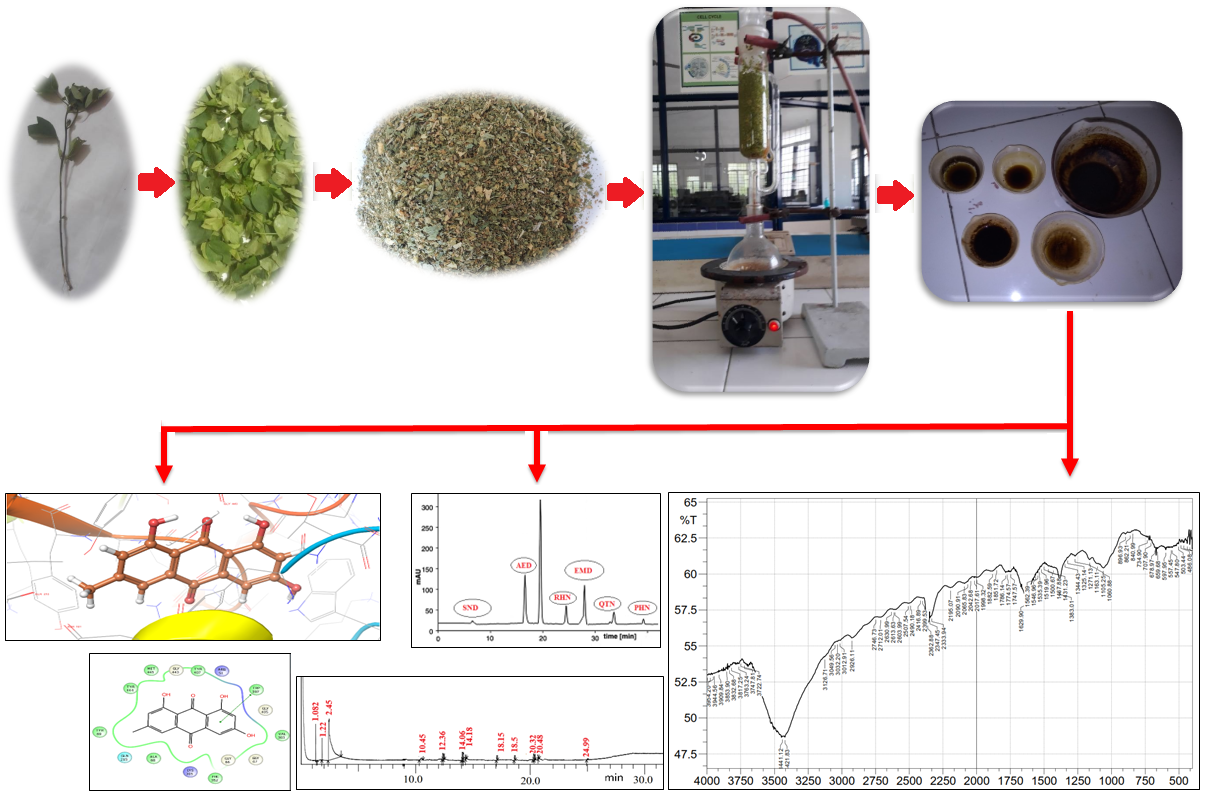
Downloads
References
CB Nemeroff. The burden of severe depression: A review of diagnostic challenges and treatment alternatives. J. Psychiatr. Res. 2007; 41, 189-206.
I Hindmarch. Beyond the monoamine hypothesis: Mechanisms, molecules and methods. Eur. Psychiatr. 2002; 17, 294-9.
SK Kulkarni, MK Bhutani and M Bishnoi. Antidepressant activity of curcumin: Involvement of serotonin and dopamine system. Psychopharmacology 2008; 201, 435-42.
JT Anthony, GK Bertram and BM Susan. Pharmacology examination and board review. McGraw-Hill Medical Publishing, New York.
M Wong and J Licinio. Research and treatment approaches to depression. Nat. Rev. Neurosci. 2001; 2, 343-51.
AS Elhwuegi. Central monoamines and their role in major depression. Progr. Neuro Psychopharmacol. Biol. Psychiatr. 2004; 28, 435-51.
Z Zhang. Therapeutic effects ofherbal extracts and constituents in animal models of psychiatric disorders. Life Sci. 2004; 75, 1659-99.
P Babulka. The Rumex, from ethobotany to modern phytotherapy (Rumex spp.), [Les rumex, de lethnobotanique ala phytotherapie modern (Rumex spp.)]. Phytotherapie 2004; 2, 153-6.
A Vasas, O Orban-Gyapai and J Hohmann. The genus Rumex: Review of traditional uses, phytochemistry, and pharmacology. J. Ethnopharmacol. 2015; 175, 198-228.
D Onstad. Whole foods companion: A guide for adventurous cooks, curious shoppers & lovers of natural foods. Chelsea Green Publishing, Vermont, 2004.
HX Liang, HQ Dai, HA Fu, XP Dong, AH Adebayo, LX Zhang and YX Cheng. Bioactive compounds from Rumex plants. Phytochem. Lett. 2010; 3, 181-4.
Z Kerem, I Bilkis, MA Flaishman and L Sivan. Antioxidant activity and inhibition of alpha-glucosidase by trans-resveratrol, piceid and a novel trans-stilbene from the roots of Israeli Rumex bucephalophorus L. J. Agr. Food Chem. 2006; 54,1243-7.
M Wegiera, HD Smolarz and A Bogucka-Kocka. Rumex L. species induce apoptosis in 1301, EOL-1 and H-9 cell lines. Acta Pol. Pharm. 2012; 69, 487-99.
BR Jeon, M Irfan, SE Lee, JH Lee and MH Rhee. Astilbechinensis modulates platelet function via impaired MAPK and PLCγ2 expression. Evid. Base. Compl. Alternative Med. 2018; 2018, 8.
J Bicker, F Petereit and A Hensel. Proanthocyanidins and a phloroglucinol derivative from Rumex acetosa L. Fitoterapia 2009; 80, 483-95.
A Derksen, A Hensel, W Hafezi, F Herrmann, TJ Schmidt, C Ehrhardt, S Ludwig and J Kühn. 3-O-galloylated procyanidins from Rumex acetosa L. inhibit the attachment of influenza A virus. PLoS One 2014; 9, 10.
SH Han, NRY Woo, SD Lee and MH Kang. Antioxidaitve and antibacterial activities of endemic plants extracts in Korea. Kor. J. Med. Crop Sci. 2006; 14, 49-55.
HMUD Qamar, R Qayyum, U Salma, S Khan, T Khan and AJ Shah. Vascular mechanisms underlying the hypotensive effect of Rumex acetosa. Pharmaceut. Biol. 2018; 56, 225-34.
K Gescher, A Hensel, W Hafezi, A Derksen and J Kuhn. Oligomericproanthocyanidins from Rumex acetosa L. inhibit the attachment of herpes simplex virus type-1. Antivir. Res. 2011; 89, 9-18.
NJ Lee, JH Choi, BS Koo, SY Ryu, YH Han, SI Lee and DU Lee. Antimutagenicity and cytotoxicity of the constituents from the aerial parts of Rumex acetosa. Biol. Pharmaceut. Bull. 2005; 28, 2158-61.
KR Khandelwal, J Khandelwal, SB Gokhle, CK Kokate and AP Pawar. Practical pharmacognosy: Techniques and experiments. Science Open, Berlin, Germany, 2008.
SE Mathew, SK Ramavarma, TD Babu and B Kuzhivelil. Preliminary assessment on phytochemical composition, cytotoxic and antitumor efficacy of Simaroubaglauca DC. leaf methanolic extract. Ann. Phytomed. 2019; 8, 121-6.
MDI Uddin, PVR Srikar, YK Preethi, C Rachanaand and R Deepika. Synthesis and characterization of chitosan nanoparticles loaded with 6-gingerol isolated from Zingiberofficinale Rosc. Ann. Phytomed. 2020; 9, 164-71.
Z Kucekova, J Mlcek, P Humpolicek, O Rrop, P Valasek and P Saha. phenolic compounds from allium schoenoprasum, tragopogonpratensis and rumexacetosa and their antiproliferative. Molecules 2011; 16, 9207-17.
AP Mohideen. In silico identification of novel immunostimulating phytochemicals with acetylcholinesterase inhibition activity from Piper betle L. and Vitexnegundo L. for the treatment of Alzheimer’s disease (AD). Ann. Phytomed. 2021; 10, 86-95.
A Hasan, I Ahmed, M Jayand and B Voirinn. Flavonoid glycosides and an Anthraquinone from Rumexchalepensis. Phytochemistry 1995; 39, 1211-3.
P Cos, N Hermans, TD Bruyne, S Apers, JB Sindambiwe, M Witvrouw, ED Clercq, BD Vanden, L Pieters and AJ Vlietinck. Antiviral activity of rwandan medicinal plants against human immunodeficiency virus type-1 (Hiv-1). Phytomedicine 2002; 9, 62-8.
NH Kapturaczak, V Thamilselvan and F Liu. Mechanism of heme oxygenase-1 gene induction by curcumin in human renal proximal tubule cells. Am. J. Physiol. Ren. Physiol. 2001; 281, 851-9.
JH Ahn, J Kim, NU Rehman, HJ Kim, MJ Ahn and HJ Chung. Effect of Rumex Acetosa extract, herbal drug, on the absorption of fexofenadine. Pharmaceutics 2020; 12, 547.
H Min, M Niu, W Zhang, J Yan, J Li, X Tan, B Li, M Su, B Di and F Yan. Emodin reverses leukemia multidrug resistance by competitive inhibition and downregulation of P-glycoprotein. PLoS One 2017; 12, e0187971.
Y Feng, SL Huang, W Dou, S Zhang, JH Chen, Y Shen, JH Shen and Y Leng. Emodin, a natural product, selectively inhibits 11beta-hydroxysteroid dehydrogenase type 1 and ameliorates metabolic disorder in diet-induced obese mice. Br. J. Pharmacol. 2010; 161, 113-26.
SC Hsu and JG Chung. Anticancer potential of emodin. Biomedicine 2012; 2, 108-16.
L Xie, H Tang, J Song, J Long, L Zhang and X Li. Chrysophanol: A review of its pharmacology, toxicity and pharmacokinetics. J. Pharm. Pharmacol. 2019; 71, 1475-87.
M Wegiera, HD Smolarz, D Wianowska and AL Dawidowicz. Anthracene derivatives in some species of Rumex L. genus. Acta Societatis Botanicorum Pol. 2007; 76, 103-8.
D Ramirez and J Caballero. Is it reliable to take the molecular docking top scoring position as the best solution without considering available structural data? Molecules 2018; 23, 1038.
M Ebadi, H Brown-Borg, J Ren, S Sharma, S Shavali, HE Refaey and EC Carlson. Therapeutic efficacy of selegiline in neurodegenerative disorders and neurological diseases. Curr. Drug Targets 2006; 7, 1513-29.
AS Kalgutkar, DK Dalvie, N Castagnoli and TJ Taylor. Interactions of nitrogen-containing xenobiotics with monoamine oxidase (MAO) isozymes A and B: SAR studies on MAO substrates and inhibitors. Chem. Res. Toxicol. 2001; 14, 1139-62.
S Carradori and R Silvestri. New frontiers in selective human MAO-B inhibitors: Miniperspective. J. Med. Chem. 2015; 58, 6717-32.
JH Meyer, AA Wilson, S Sagrati, L Miler, P Rusjan and PM Bloomfield. Brain monoamine oxidase A binding in major depressive disorder. Arch. Gen. Psychiatr. 2009; 66, 1304.
Schrodinger, Available at: https://www.schrodinger.com, accessed February 2022.

Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2022 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






