Corrosion Behavior of AISI 316L Stainless Steel in a Highly Sour Environment
DOI:
https://doi.org/10.48048/tis.2022.4642Keywords:
Sour corrosion, Localized corrosion, Partial pressure of H2S, 316L Stainless-steel, Passive filmAbstract
Sour corrosion in the presence of H2S gases can significantly affect the passive films deterioration of the 316L steel. 0 and 3 bar of H2S gases were chosen to identify how H2S gases could affect the corrosion rate on steels surface at normal and extremely sour conditions. Experiment was operated in an 7L autoclave for 7 days and was kept at 60 °C throughout the testing. The uniform corrosion rates were measured by weight loss while the pitting corrosion rates were determined by using a profilometer, respectively. The oxide layers formed under these conditions were determined by XPS. The results found that at 3 bar of H2S gas condition shows the formation of multi-layers consist of Fe3O4, (NiOH)2, NiO, MoO3, FeS, and NiS. Finally, corrosion products formation on 316L steels surface was then discussed.
HIGHLIGHTS
- The corrosion-resistant alloy (CRA) has been introduced to the pipeline industry due to its excellent properties of corrosion resistance but still remains unclear in a highly sour environment and needs to be further investigated
- 316L steel was selected because it is one of the austenitic alloys containing chromium, nickel and molybdenum, making it more corrosion resistant
- The effects of H2S gas pressure on the AISI 316L stainless-steel, with the emphasis on the formation and deterioration of passive films have been discussed
GRAPHICAL ABSTRACT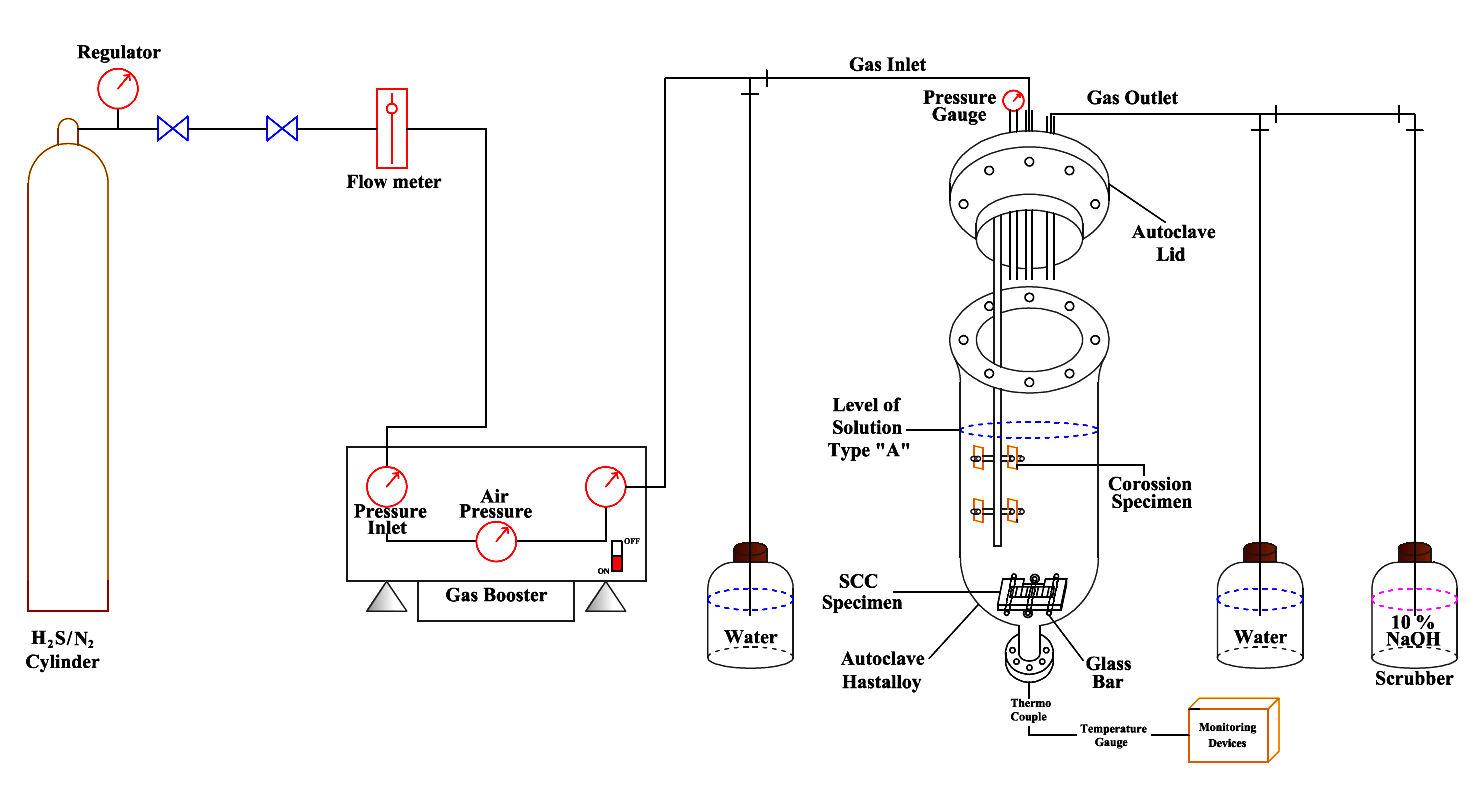
Downloads
References
RB Rebak and TE Perez. Effect of carbon dioxide and hydrogen sulfide on the localized corrosion of carbon steels and corrosion resistant alloys. In: Proceedings of the NACE International Corrosion 2017 Conference, Texas, United States. 2017, p. 1-15.
J Kvarekvål and J Moloney. Sour corrosion. In: AM El-Sherik (Ed.). Trends in oil and gas corrosion research and technologies: Production and transmission, Woodhead Publishing, Sawston, United Kingdom, 2017, p. 113-42.
M Muhammed, M Mustapha, TL Ginta, AM Ali, F Mustapha and CC Hampo. Statistical review of microstructure-property correlation of stainless steel: Implication for pre-and post-weld treatment. Processes 2020; 8, 811.
MA Rao, RS Babu and MV Pavan Kumar. Stress corrosion cracking failure of a SS 316L high pressure heater tube. Eng. Fail. Anal. 2018; 90, 14-22.
M Liu, S Bell, M Segarra, NH Steven, G Will, W Saman and F Bruno. A eutectic salt high temperature phase change material: Thermal stability and corrosion of SS316 with respect to thermal cycling. Sol. Energ. Mater. Sol. Cell. 2017; 170, 1-7.
S Zhang, C Sun, J Di and Y Tan. Corrosion behavior and morphology of passive films modified with zinc-aluminum simultaneous treatment on different metals. Materials 2020; 10, 986.
D Li, Q Liu, W Wang, L Jin and H Xiao. Corrosion behavior of AISI 316L stainless steel used as inner lining of bimetallic pipe in a seawater environment. Materials 2021; 14, 1539.
J Hesketh, EJF Dickinson, ML Martin, G Hinds and A Turnbull. Influence of H2S on the pitting corrosion of 316L stainless steel in oilfield brine. Corrosion Sci. 2021; 182, 109265.
NS Al-Mamun, W Haider and I Shabib. Corrosion resistance of additively manufactured 316L stainless steel in chloride-thiosulfate environment. Electrochim. Acta 2020; 362, 137039.
J Qiu, B Leng, H Liu, DD Macdonald, A Wu, Y Jia, W Xue, G Yu and X Zhou. Effect of SO42− on the corrosion of 316L stainless steel in molten FLiNaK salt. Corrosion Sci. 2018; 144, 224-9.
ASTM International. Standard practice for preparing, cleaning, and evaluating corrosion test specimens. ASTM International, Pennsylvania, United States, 2017.
M Monnot, RP Nogueira, V Roche, G Berthomé, E Chauveau, R Estevez and M Mantel. Sulfide stress corrosion study of a super martensitic stainless steel in H2S sour environments: Metallic sulfides formation and hydrogen embrittlement. Appl. Surf. Sci. 2017; 394, 132-41.
M Shah, MTM Ayob, R Rosdan, N Yaakob, Z Embong and NK Othman. The effect of H2S pressure on the formation of multiple corrosion products on 316L stainless steel surface. Sci. World J. 2020; 2020, 3989563.
RD Monteiro, JVD Wetering, B Krawczyk and DL Engelberg. Corrosion behaviour of type 316L stainless steel in hot caustic aqueous environments. Met. Mater. Int. 2020; 26, 630-40.
L Zhang, X Tang, Z Wang, T Li, Z Zhang and M Lu. The corrosion behavior of 316L stainless steel in H2S environment at high temperatures. Int. J. Electrochem. Sci. 2017; 12, 8806-19.
Z Wang, L Zhang, Z Zhang and M Lu. Combined effect of pH and H2S on the structure of passive film formed on type 316L stainless steel. Appl. Surf. Sci. 2018; 458, 686-99.
KL Cwalina, HM Ha, N Ott, P Reinke, N Birbilis and JR Scully. In operando analysis of passive film growth on Ni-Cr and Ni-Cr-Mo alloys in chloride solutions. J. Electrochem. Soc. 2019; 166, 3241-52.
Downloads
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.







