Fe3+ Selective Pyrimidine Based Chromogenic and Fluorogenic Chemosensor
DOI:
https://doi.org/10.48048/tis.2022.4487Keywords:
Chemosensor, Pyrimidine based chromogenic, UV-Vis, Fluorescence, Fe3Abstract
By using UV-Visible and fluorescence measurements, a new chemosensor N-(pyrimidin-2-yl) thiophene-2-carboxamide (PTC) was introduced for the discriminating and responsive detection of Fe3+ in aqueous methanolic medium. Since the PTC-Fe3+ complexation occurred in 1:1 binding stoichiometry, the receptor PTC preferentially revealed a charge transfer band between 310 to 475 nm in the UV-Vis technique, with the absorption peak at 351 nm. Furthermore, the fluorescence technique, which dampened PTC's fluorescence emission band at 325 nm, showed similar selectivity. The Stern-Volmer graphic shows that fluorescence quenching occurs in a static manner. The series of examined metal ions (Na+, K+, Li+, Cs+, Fe2+, Ni2+, Sr2+, Cu2+, Cd2+, Pb2+, Co2+, Hg2+, Ba2+, Mg2+, Mn2+, Ca2+, Zn2+, Cr3+, Ag+ and Al3+) failed to perturb the UV-Vis and fluorescence of PTC. With PTC, the concentration of Fe3+ can be detected down to 2.03 and 1.24 µM by UV-Vis and fluorescence methods, respectively.
HIGHLIGHTS
- An easy-to-synthesize pyrimidine-based receptor PTC was developed for the detection of Fe3+
- PTC showed Fe3+selective turn-off fluorescence and a new charge transfer absorption band at 351 nm
- LOD of PTC is 2.03 and 1.24 µM, respectively by UV-Vis and fluorescence methods
- Fluorescence quenching is static in nature
GRAPHICAL ABSTRACT 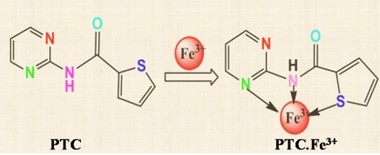
Downloads
References
CD Geddes and JR Lakowicz. Advanced concepts in fluorescence sensing part A: Small molecule sensing. Springer, New York, 2005.
APD Silva, HQ Gunaratne, N Gunnlaugsson, AJ M Huxley, CP McCoy, JT Rademacher and TE Rice. Signaling recognition events with fluorescent sensors and switches. Chem. Rev. 1997; 97, 1515-66.
APD Silva, DB Fox, AJM Huxley and TS Moody. Combining luminescence, coordination and electron transfer for signaling purposes. Coord. Chem. Rev. 2000; 205, 41-57.
C Bargossi, MC Fiorini, M Montalti, L Prodi and N Zaccheroni. Recent developments in transition metal ion detection by luminescent chemosensors. Coord. Chem. Rev. 2000; 208, 17-32.
R Martinez-Manez and F Sancenon. Fluorogenic and chromogenic chemosensors and reagents for anions. Chem. Rev. 2003; 103, 4419-76.
HLJ Fan and X Peng. Colorimetric and fluorescent probes for the optical detection of palladium ions. Chem. Soc. Rev. 2013; 42, 7943-62.
H Dai, Y Yan, Y Guo, L Fan, Z Che and H Xu. A selective and sensitive “Turn‐on” fluorescent chemosensor for recognition of Hg2+ ions in water. Chem. Eur. J. 2012; 18, 11188.
PG Georgopoulos, A Roy, MJ Yonone-Lioy, RE Opiekun and PJ Lioy. Environmental copper: Its dynamics and human exposure issues. J. Toxicol. Environ. Health, Part B 2001; 4, 341-94.
SK Sahoo, D Sharma, RK Bera, G Crisponi and JF Callan. Iron (III) selective molecular and supramolecular fluorescent probes. Chem. Soc. Rev. 2012; 41, 7195-227.
RH Crabtree. Metal ions at work. Science 1994; 266, 1591-2.
MB Zimmermann. The influence of iron status on iodine utilization and thyroid function. Annu. Rev. Nutr. 2006; 26, 367-89.
S Toyokuni, Iron-induced carcinogenesis: The role of redox regulation. Free Radical Biol. Med. 1996; 20, 553-66.
JR Burdo and JR Connor. Brain iron uptake and homeostatic mechanisms: An overview. Bio. Metals 2003; 16, 63-75.
E Beutler. Iron storage disease: Facts, fictions and progress. Blood Cells Mol. Dis. 2007; 39, 140-7.
SK Sahoo and G Crisponi. Recent advances on iron (III) selective fluorescent probes with possible applications in bioimaging. Molecules 2019; 24, 3267.
P Patil, S Sehlangia, A Patil, C Pradeep, SK Sahoo and U Patil. A new phthalimide based chemosensor for selective spectrophotometric detection of Cu(II) from aqueous medium. Spectrochim. Acta A 2019; 220, 117129.
Y Upadhyay, LT Babu, P Paira, G Crisponi, SK Ashok Kumar, R Kumar and SK Sahoo. Three-in-one type fluorescent sensor based on pyrene pyridoxal cascade for the selective detection of Zn (II), hydrogen phosphate and cysteine. Dalton Trans. 2018; 47, 742-9.
NB Patil, UD Patil, PA Patil, S Bothra, SK Sahoo, S Sehlangia, CP Pradeep, AA Patil and SR Patil. A fused benzothiazolo pyrimidine based chemosensor for selective optical detection of Fe3+ and I− ions in aqueous media. ChemistrySelect 2019, 4, 4185-9.
N Kshirsagar, R Sonawane, P Patil, J Nandre, P Sultan, S Sehlangia, CP Pradeep, Y Wang, L Chen and SK Sahoo. Fluorescent chemosensor for Al(III) based on chelation-induced fluorescence enhancement and its application in live cells imaging, Inorg. Chim. Acta 2020; 511, 119805.
JS Chib, MF Stempien, JJT Cecil, GD Ruggieri and RF Nigrelli. Physiologically active substances from marine sponges IV: Heterocyclic compounds. J. Pharma. Sci. 1977; 66, 1052-4.
P Job. Formation and stability of inorganic complexes in solution. Ann. Chim. Appl. 1928; 9, 113-203.
SK Sahoo, D Sharma, A Moirangthem, A Kuba, R Thomas, R Kumar, A Kuwar, C Heung-Jin and A Basu. Pyridoxal derived chemosensor for chromogenic sensing of Cu2+ and fluorogenic sensing of Fe3+ in semi-aqueous medium. J. Luminescence 2016; 172, 297-303.
D Das, R Alu and M Ali. Rhodamine 6G-based efficient chemosensor for trivalent metal ions (Al3+, Cr3+ and Fe3+) upon single excitation with applications in combinational logic circuits and memory devices. Analyst 2022; 147, 471-9.
Y Upadhyay, T Anand, L TilakBabu, P Paira. SKA Kumar, R Kumar and SK Sahoo. Combined use of spectrophotometer and smartphone for the optical detection of Fe3+ using a vitamin B6 cofactor conjugated pyrene derivative and its application in live cells imaging, J. Photochem. 2018; 361, 34-40.
O Stern and M Volmer. Über die Abklingzeit der fluoreszenz. Phys. J. 1919; 20, 183-8.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2022 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.