The Effect of Particle Size on the Physical Characteristics and Drug-release Behavior of Mini-tablets
DOI:
https://doi.org/10.48048/tis.2021.43Keywords:
Drug release profile, Mini-tablets, Particle size, Physical characterizationAbstract
This study aimed to investigate the influence of particle size of the excipients on the behavior of mini-tablets in terms of physical characteristics and drug release profiles. Material and methods: All of the mini-tablets based on HPMC, PEG 6K and PEO 8M PVP produced high quality mini-tablets from both sieve fractions while 90F was very fragile. Results: Crushing strength values were the most pronounced difference between the mini-tablets from all formulations. The PEO 8M-based mini-tablets showed the highest values and deformation behavior instead of fracture. Water absorption and drug release profiles also showed intriguing and different results between the various formulations. Conclusion: The results suggested that even though different particle size has limited effect on the physical characteristic of the product, it may have a significant effect on its drug dissolution profile.
HIGHLIGHTS
- The particle size of powders or granules can influence the physical properties and release profiles of tablets, particularly mini-tablets
- PEG 6K and PEO 8M mini-tablets from the larger sieve fraction variation absorbed less moisture and released drugs faster than their counterparts, whereas HPMC and PVP 90F mini-tablets from both sieve fractions exhibited a similar pattern in terms of moisture uptake and drug release profiles
- PEO 8M mini-tablets of both sieve fractions displayed almost linear release profiles with the highest crushing strength than the rest of the formulations
GRAPHICAL ABSTRACT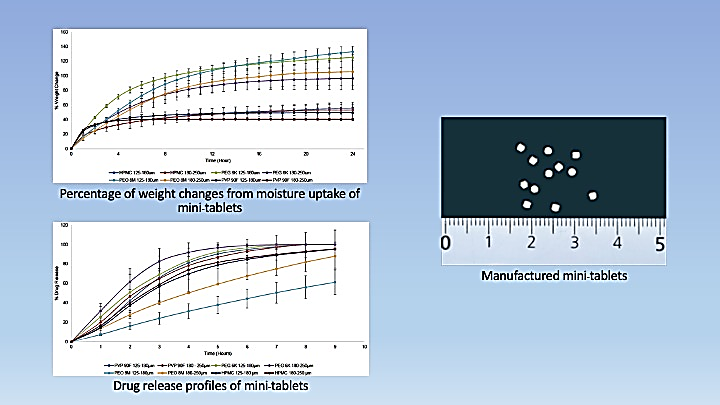
Downloads
References
P Lennartz and JB Mielck. Minitabletting: Improving the compactability of paracetamol powder mixtures. Int. J. Pharm. 1998; 173, 75-85.
SM Majeed, MK Al-Shaheen, RN Al-Zidan and SM Mahmood. Multiple Unite Pellet Systems (MUPS) as drug delivery model. J. Drug Deliv. Ther. 2020; 10, 231-5.
H Kotlowska, J Krotka, M Szymanska, B Kubiak, M Sznitowska and BN Nalluri. the use of novel tools for the assessment of powders and granules flow properties and for the analysis of minitablets compression process. Drug Dev. Ind. Pharm. 2020; 46, 547-56.
C Tissen, K Woertz, J Breitkreutz and P Kleinebudde. Development of mini-tablets with 1 mm and 2 mm diameter. Int. J. Pharm. 2011; 416, 164-70.
BP Commission. British pharmacopoeia 2020. The Stationery Office, London, 2019.
AA El-Gawad, OA Soliman, SA Barker and GNS Girgis. Formulation and Evaluation of gel forming ocular minitablets containing piroxicam. Br. J. Pharm. Res. 2012; 2, 141-67.
MRC Marques, R Loebenberg and M Almukainzi. Simulated biological fluids with possible application in dissolution testing. Dissolution Tech. 2011; 18, 15-28.
W Weyenberg, A Vermeire, JP Remon and A Ludwig. Characterization and in vivo evaluation of ocular bioadhesive minitablets compressed at different forces. J. Contr. Release 2003; 89, 329-40.
W Weyenberg, A Vermeire, J Vandervoort, JP Remon and A Ludwig. Effects of roller compaction settings on the preparation of bioadhesive granules and ocular minitablets. Eur. J. Pharm. Biopharm. 2005 ; 59, 527-36.
M Sacchetti, R Teerakapibal, K Kim and EJ Elder. Role of water sorption in tablet crushing strength, disintegration, and dissolution. AAPS Pharm. Sci. Tech. 2017; 18, 2214-26.
M Aulton and KMG Taylor. Aulton’s Pharmaceutics: The design and manufacture of medicines. 5th ed. Churchill Livingstone, Edinburgh, Scotland, 2017.
CK Brown, HD Friedel, AR Barker, LF Buhse, S Keitel, TL Cecil, J Kraemer, JM Morris, C Reppas, MP Stickelmeyer C Yomota and VP Shah. Meeting report: FIP/AAPS joint workshop report: Dissolution/in vitro release testing of novel/special dosage forms. Dissolution Tech. 2011; 73, 338-53.
Downloads
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.







