The Behavior of Nanovesicles of Physicochemical Profiles: Compose of β-Cyclodextrin and Cisplatin for Anti-Tumor Activity and Decrease Toxicity Affected Normal Surrounding Tumor Tissues
DOI:
https://doi.org/10.48048/tis.2023.4293Keywords:
Cisplatin, Nanovesicles, Hydroxypropyl β-cyclodextrin, HeLa Cells, SRB Assay, Liposomes, Anti-Tumor activityAbstract
CP (cisplatin) was entrapped in lipid bilayer and β-CD-CP (β-cyclodextrin-CP) was entrapped in hydrophilic bilayer both were prepared by chloroform film method with sonication. The entrapment efficiency of CP-LPS (CP-Liposome) and β-CD-CP-LPS were 83.44 and 82.20 %, respectively, two-tailed t-test and no significant difference at the confidence interval, p < 0.05. Size distribution of CP-LPS and β-CD-CP-LPS were investigated by DLS were in range 322 and 1033 nm, respectively. CP release from CP-LPS or β-CD-CP-LPS exposed to physiological conditions as PBS at 37 °C. CP from both LPS formulations have showed an initial burst release around < 17 and < 10 %, respectively. CP-LPS and β-CD-CP-LPS maintained as plateau phase of both for four days and seven days, respectively. CP-LPS showed % cumulative controlled release CP of CP-LPS and β-CD-CP-LPS were about 15 - 16 and 10 - 11 %, respectively. CP-LPS and β-CD-CP-LPS released until 83.6 % for six days and 82 % for eight days, respectively. CP released from CP-LPS and β-CD-CP-LPS were in a controlled deportment for 10 days. β-CD-CP-LPS, CP molecules bound β-CD was exhibited in dynamic equilibrium with free drug molecules which ascribes the controlled release behavior of β-CD-CP-LPS to quietly release and prolong action, diminish dose of toxicity of cancer drug that effected to normal tissues that surrounding tumor tissue. β-CD was oligosaccharides,an attracted tumor, tumor snatch themselves as nutrient consume more than the normal tissues. Thus, β-CD-CP-LPS and low dose of normal dose of CP can damage cancer cells. This synergistic effect was saved for normal cells which surrounding tumor tissue.
HIGHLIGHTS
- Nanovesicles (NVs) are a particularly attractive delivery system because of their NVs’ size, which permits carrying across biological membranes and exact target sites by a probable surface adjustment, sustained release delivers, and diminished drug dose to treat. The experiment found that β-CD-CP-LPS transported to HeLa cells in a sustained release to prolonged action. β-CD-CP-LPS, CP molecules bound within the β-CD are in dynamic equilibrium with free drug molecules, which ascribes the controlled release behavior of β-CD-CP-LPS to quietly released and long term which diminished toxicity that effected to normal tissues which surrounding tumor tissue. Moreover, β-CD was an oligosaccharide, an attracted tumor. Tumors snatch themselves as nutrient consume more than the normal Thus, β-CD-CP-LPS and a low dose of normal dose of CP can damage cancer cells. This cooperative effect was saved for normal cells which surround tumor tissue.
GRAPHICAL ABSTRACT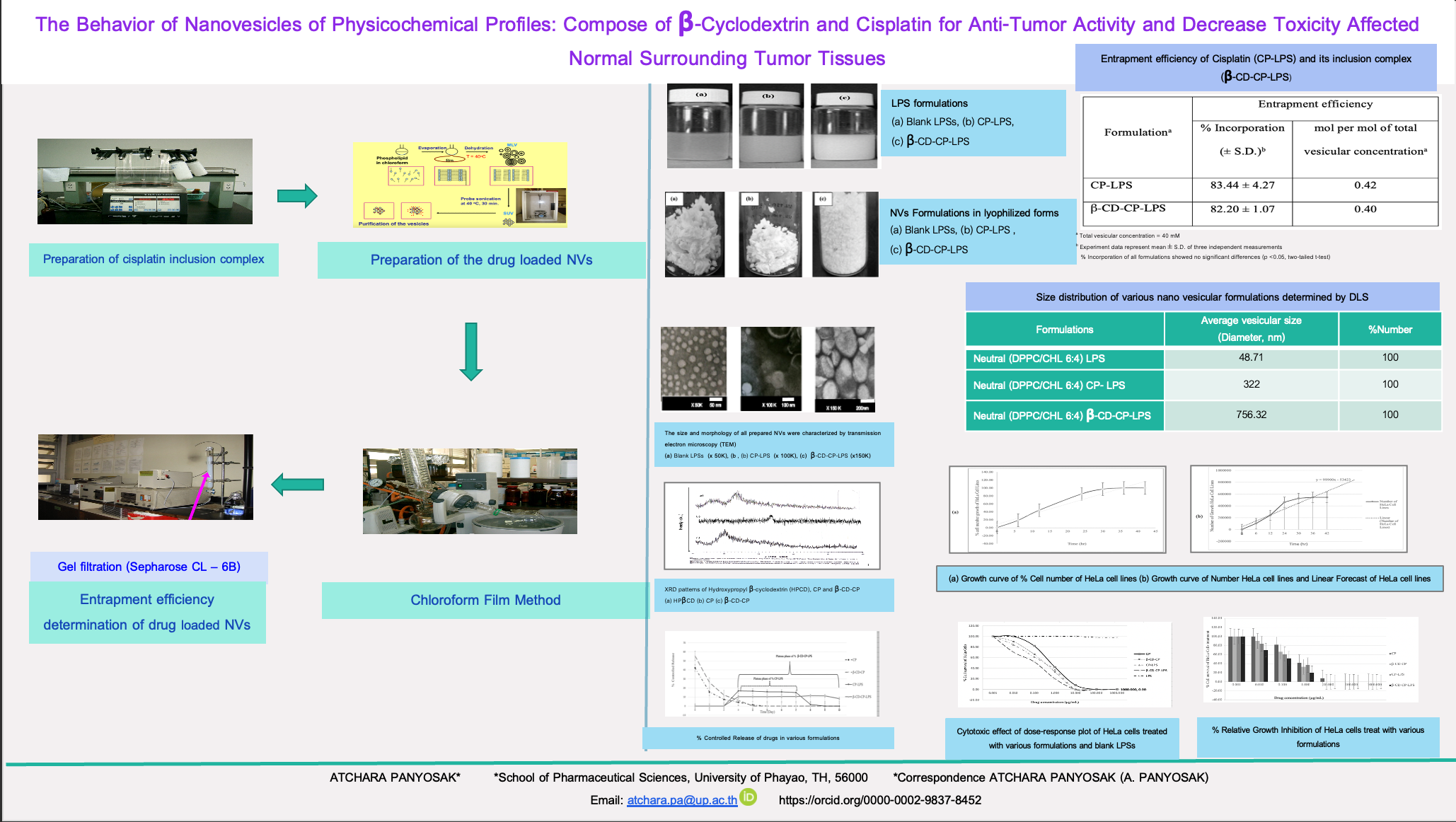
Downloads
References
KS Kim, DH Kim and DH Kim. Recent advances to augment NK cell cancer immunotherapy using nanoparticles. Pharmaceutics 2021; 13, 525.
Y Gao, K Yang, AN Shelling and Z Wu. Nanotechnology-enabled COVID-19 mRNA vaccines. Encyclopedia 2021; 1, 773-80.
A Panyosak, J Manosroi, Y Rojanasakul and A Manosroi. Safety assessment of azelaic acid and its derivatives entrapped in nanovesicles. Hum. Exp. Toxicol. 2009; 28, 387-92.
Understanding Nanotechnology. Nanotechnology in medicine - nanotechnology in medicine application, Available at: https://www.understandingnano.com/medicine.html, accessed October 2021.
Nanotechnology in medicine: Technology trends, medical device network, Available at: network.com/comment/nanotechnology-medicine-technology, accessed October 2021.
Y Dong, X Wu, X Chen, P Zhou, F Xu and W Liang. Nanotechnology shaping stem cell therapy: Recent advances, application, challenges, and future outlook. Biomed. Pharmacother. 2021; 137, 111236.
E Katz. Magnetic nanoparticles. Magnetochemistry 2020; 6, 6.
RN Alauddin, VE Petrov, K Langer, A Berthold, DA Kharkevich and J Kreuter. Delivery of loperamide across the blood-brain barrier with polysorbate 80-coated poly butyl cyanoacrylate nanoparticles. Pharm. Res. 1997; 14, 325-8.
J Martin, EJ Díaz-Montaña and AG Asuero. Cyclodextrins: Past and present. In: P Arora and N Dhingra (Eds.). Cyclodextrin. IntechOpen, London, 2018, p. 1-43.
A Rasheed. Cyclodextrins as drug carrier molecule: A review. Sci. Pharm. 2018; 76, 567-98.
G Tiwari, R Tiwari and AK Rai. Cyclodextrins in delivery systems: Applications. J. Pharm. Bioallied Sci. 2010; 2, 72-9.
B Gidwani and A Vyas. A comprehensive review on cyclodextrin-based carriers for delivery of chemotherapeutic cytotoxic anticancer drugs. Biomed. Res. Int. 2015; 2015, 198268.
E Raymond, S Faivre, S Chaney, J Woynarowski and E Cvitkovic. Cellular and molecular pharmacology of oxaliplatin. Mol. Canc. Ther. 2002; 3, 227-35.
I Zaki, RAI Abou-Elkhair, AHA Almaaty, OAA Ali, E Fayad, AGA Gaafar and MY Zakaria. Design and synthesis of newly synthesized acrylamide derivatives as potential chemotherapeutic agents against MCF-7 breast cancer cell line lodged on PEGylated bilosomal nano-vesicles for improving cytotoxic activity. Pharmaceuticals 2021; 14, 1021.
R Kadokawa, T Fujie, G Sharma, K Ishibashi, K Ninomiya, K Takahashi, E Hirata and K Kuroda. High loading of trimethylglycine promotes the aqueous solubility of poorly water‐soluble cisplatin. Sci. Rep. 2021; 11, 9770.
CM Riley and LA Sternson. Cisplatin. In: K Florey (Ed.). Analytical profiles of drug substances. Academic Press, New York, 1985, p. 78-105.
K Czarnobaj and J Lukasia. In vitro release of cisplatin from sol-gel processed porous silica xerogels. Drug Deliver. 2004; 11, 341-4.
B De Spiegeleer, G Slegers, C Vandecasteele, WVD Bossche, K Schelstraete, A Claeys and PD Moerloose. Microscale synthesis of nitrogen-13-labeled cisplatin. J. Nucl. Med. 1986; 27, 399-403.
World Health Organization. Annex 10. Stability testing of active pharmaceutical ingredients and finished products: WHO expert committee on specifications for pharmaceutical preparations. World Health Organizatio, Geneva, Switzerland, 2009.
D Moreno, S Zalba, H Colom, IF Trocóniz, CT de Ilarduya and MJ Garrido. Biopharmaceutic and pharmacodynamic modeling of the in vitro Antiproliferative effect of new controlled delivery systems of cisplatin. Eur. J. Pharm. Sci. 2009; 37, 341-50.
K Taylor and M Aulton. Aulton’s pharmaceutics - the design and manufacture of medicines. Elsevier, Edinburgh, United Kingdom, 2017.
AT Florence and D Attwood. FASTtrack: Physicochemical principles of pharmacy. Pharmaceutical Press, London, 2008.
D Jones. FASTtrack: Pharmaceutics dosage form and design. Pharmaceutical Press, London, 2008.
L Kelland. The resurgence of platinum-based cancer chemotherapy. Nat. Rev. Canc. 2007; 7, 573-84.
Z Gu. Liposome-based drug delivery systems in cancer immunotherapy. Pharmaceutics 2020; 12, 1054.
TN Tozer and M Rowland. Introduction of pharmacokinetics and pharmacodynamics: The quantitative basis of drug therapy. Lippincott Williams & Wilkins, Baltimore, United States, 2006.
A Matencio, F Caldera, C Cecone, JM López-Nicolás and F Trotta. Cyclic oligosaccharides as active drugs, an updated review. Pharmaceuticals 2020; 13, 281.
AR Daniel. Cyclodextrin-modified nanomaterials for drug delivery: Classification and advances in controlled release and bioavailability. Pharmaceutics 2021; 13, 2131.

Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2022 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






