Assessment of Cultivation Parameters Influencing Polygalacturonase Production by Aspergillus sp. SO5 in Submerged Fermentation
DOI:
https://doi.org/10.48048/tis.2023.4191Keywords:
Polygalacturonase, Aspergillus sp., Physiochemical parameters, Submerged fermentationAbstract
Polygalacturonase represents an important enzyme of the pectinase consortium in the food and beverage industry. The present study aims to isolate, screen, and enhance physicochemical parameters for enzyme production under submerged fermentation. A total of 34 fungal strains were obtained from rotten fruits and 70.6 % of them are able to produce polygalacturonase by degrading pectin in pectin screening agar. The fungal isolate (SO5) that exhibited the greatest diameter of the hydrolysis zone (45.3 ± 0.04 mm) was selected for further study and later identified as Aspergillus sp. SO5, based on morphological characteristics using light microscopic observation. Several physicochemical parameters improved for pectinase production including incubation temperature, initial pH, agitation speed, inoculum density, and carbon and nitrogen sources. Under improved conditions, the polygalacturonase activity and fungal biomass increased up to 74.63 and 69.44 %, respectively. The finding also revealed that the improved physicochemical conditions enhance enzyme production and shorten the incubation period from 6 days to 5 days.
HIGHLIGHTS
- This research paper highlighted the capability of indigenous fungus (Aspergillus) to produce polygalacturonase. The finding revealed that the improved physicochemical conditions including agitation speed, inoculum size, the concentration of pectin, and incorporation of yeast extract in the culture medium could enhance polygalacturonase production by up to 74.63 % (34.21 ± 0.49 U/mL). The improved physicochemical conditions not only enhance the polygalacturonase production but shorten the incubation period to obtain optimum enzyme production.
GRAPHICAL ABSTRACT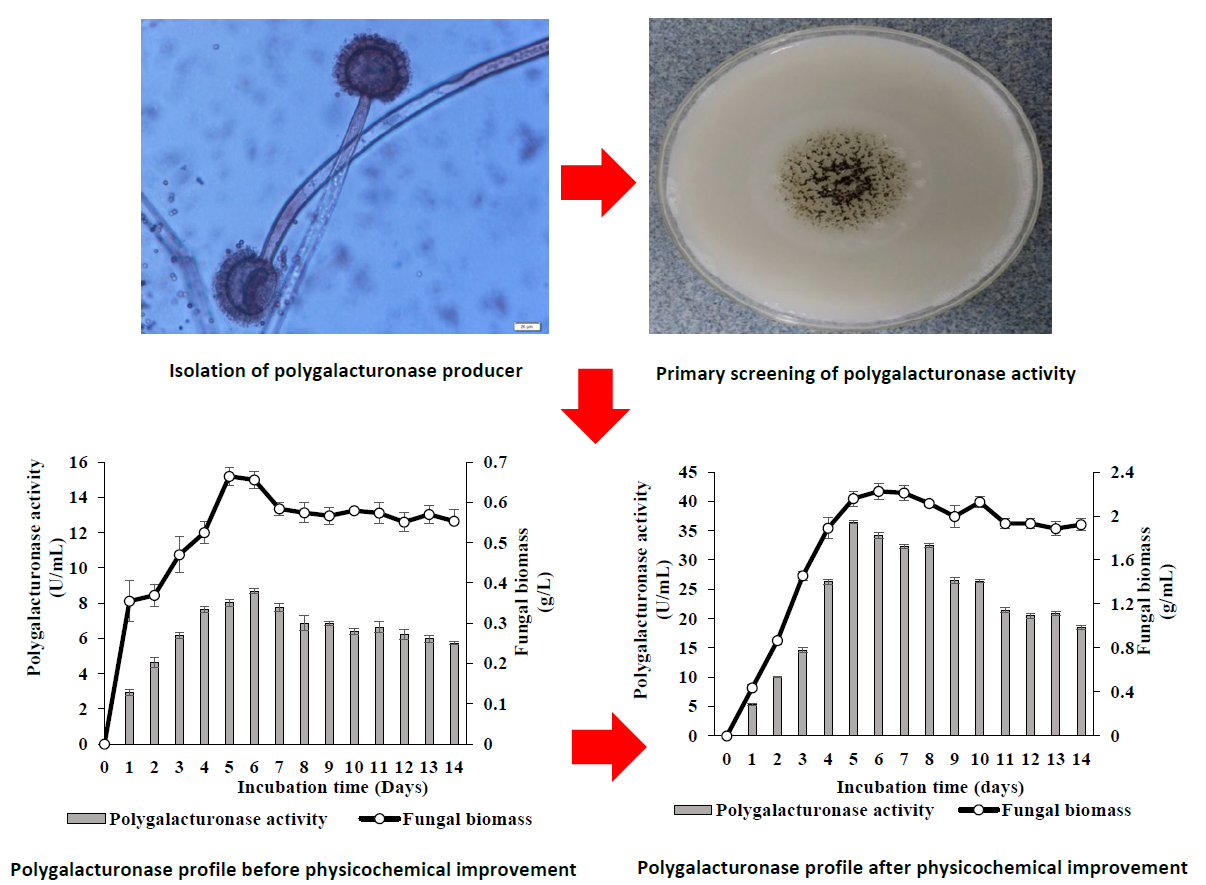
Downloads
References
P Samreen, M Mangipudi, S Grover, C Rajan and G Sibi. Production of pectinases and pectinolytic enzymes: Microorganisms, cultural conditions and substrates. Appl. Biotechnol. Microbiol. 2019; 14, 555884.
KC Rohit, KM Sudipta, B Purushotham and MK Swamy. Isolation, production, and characterization of extracellular pectinase from Aspergillus niger K3. Int. J. Pharma Bio. Sci. 2013; 4, 667-75.
A Blandino, T Iqbalsyah, SS Pandiella, D Cantero and C Webb. Polygalacturonase production by Aspergillus awamori on wheat in solid-state fermentation. Appl. Microbiol. Biotechnol. 2002; 58, 164-9.
G Anand, S Yadav and D Yadav. Production, purification, and biochemical characterization of an exo-polygalacturonase from Aspergillus niger MTCC 478 suitable for clarification of orange juice. 3 Biotech 2017; 7, 122.
N Deshmukh, R Talkal, K Jha, PG Singh and DC Prajapati. Production, purification, characterization and comparison of polygalacturonase from various strains of Aspergillus. Int. J. Sci. Tech. Res. 2012; 1, 85-91.
E Karahalil, F Demirel, E Evcan, M Germec, C Tari and I Turhan. Microparticle-enhanced polygalacturonase production by wild type Aspergillus sojae. 3 Biotech 2017; 7, 361.
QY Almulaiky, AA Albishri, NM Khalil, Y Algamal, M Aldhahri, AS AlHarbi, AH Al-Talhi and R Alhadi. Polygalacturonase by Aspergillus niger using seaweed waste under submerged fermentation: Production, purification and characterization. Biomed. J. Sci. Tech. Res. 2020; 25, 004249.
AL Ezugwu, TC Ezike, AN Ibeawuchi, CA Nsude, DI Udenwobele, SOO Eze, CU Anyawu and FC Chilaka. Comparative studies on pectinases obtained from Aspergillus fumigatus and Aspergillus niger in submerged fermentation system using pectin extracted from mango, orange and pineapple peels as carbon sources. Niger. J. Biotechnol. 2014; 28, 26-34.
BK Sethi, PK Nanda and S Sahoo. Enhanced production of pectinase by Aspergillus terreus NCFT 4269.10 using banana peels as substrate. 3 Biotech 2016; 6, 36.
R Ketipally and MR Ram. Optimization of pectinase production by Aspergillus oryzae RR 103. Curr. Agr. Res. J. 2018; 6, 37-44.
I Darah, MMJ Taufiq and S Lim. Pectinase production by Aspergillus niger LFP-1 using pomelo peels as substrate: An optimization study using shallow tray system. Ind. J. Biotechnol. 2015; 14, 552-8.
R Daoud, MK Tahla and MF Azmeh. Optimization of polygalacturonase production by Trichoderma harzianum on orange peels in submerged fermentation. Int. J. Chem. Tech. Res. 2016; 9, 359-65.
MA Klinch. Identification of common Aspergillus species. Centraalbureau voor Schimmelcultures, Utrecht, Netherlands, 2002.
I Darah, W Haritharan and SH Lim. Involvement of physicochemical parameters on pectinase production by Aspergillus niger HFD5A-1. J. Pure Appl. Microbiol. 2013; 7, 2541-9.
NP Patil and BL Chaudhari. Production and purification of pectinase by soil isolate Penicillium sp. and search for better agro-residue for its SSF. Recent Res. Sci. Tech. 2010; 2, 36-42.
GL Miller. Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal. Chem. 1959; 31, 426-8.
P Srivastava and R Malviya. Sources of pectin, extraction, and its applications in pharmaceutical industry - An overview. Indian J. Nat. Prod. Res. 2011; 2, 10-8.
JH Doughari and GC Onyebarachi. Production, purification and characterization of polygalacturonase from Aspergillus flavus grown on orange peel. Appl. Microbiol. Open Access 2019; 4, 155.
D Ibrahim, N Salikin, LS Hong, R Ahmad and H Weloosamy. Pomelo peels as alternative substrate for extracellular pectinase production by Aspergillus niger HFM-8. Malays. J. Microbiol. 2013; 9, 308-16.
FS Ire and EG Vinking. Production, purification and characterization of polygalacturonase from Aspergillus niger in solid state and submerged fermentation using banana peels. J. Adv. Biol. Biotechnol. 2016; 10, 1-15.
V Sharma and P Rishishwar. Effect of pH on commercially applicable pectinase enzyme derived from strain improved Aspergillus niger using agro-industrial waste by solid state fermentation process. Res. J. Sci. IT Manag. 2015; 5, 11-4.
RC Fontana and MM Silveira. Influence of pectin, glucose, and pH on the production of endo- and exo-polygalacturonase by Aspergillus oryzae in liquid medium. Braz. J. Chem. Eng. 2012; 29, 683-90.
K Anuradha, PN Padma, S Venkateshwar and G Reddy. Effect of physical factors on pellet morphology of Aspergillus awamori MTCC 9166 and polygalacturonase production. Biocatalysis Agr Biotechnol. 2014; 3, 271-4.
M Palaniyappan, V Vijayagopal, R Viswanathan and T Viruthagiri. Screening of natural substrates and optimization of operating variables on the production of pectinase by submerged fermentation using Aspergillus niger MTCC 281. Afr. J. Biotechnol. 2009; 8, 682-6.
I Darah, MMJ Taufiq and S Lim. Pectinase production by Aspergillus niger LFP-1 using pomelo peels as substrate: An optimization study using shallow tray system. Ind. J. Biotechnol. 2015; 14, 552-8.
I Darah, M Nisha and SH Lim. Enhancement of polygalacturonase production from Enterobacter aerogenes NB02 by submerged fermentation. Adv. Stud. Biol. 2013; 5, 173-89.
S Batool, MJ Asad, SMS Naqvi, RT Mahmood, A Guffar, M Gulfraz and SH Hadri. Production and partial purification of pectin lyase by Aspergillus niger grown on orange peels. Afr. J. Microbiol. Res. 2013; 7, 1144-9.
R Abdullah, A Jafer, K Nisar, A Kaleem, M Iqtedar, T Iftikhar, F Saleem and S Naz. Process optimization for pectinase production by locally isolated fungal strain using submerged fermentation. Biosci. J. 2018; 34, 1025-32.
I Ahmed, MA Zia, MA Hussain, Z Akram, MT Naveed and A Nowrouzi. Bioprocessing of citrus waste peel for induced pectinase production by Aspergillus niger; its purification and characterization. J. Radiat. Res. Appl. Sci. 2016; 9, 148-54.
IG Sandri, RC Fontana and MM Da-Silveira. Influence of pH and temperature on the production of polygalacturonases by Aspergillus fumigatus. LWT Food Sci. Tech. 2015; 61, 430-6.
RS Jayani, SK Shukla and R Gupta. Screening of bacterial strains for polygalacturonase activity: Its production by Bacillus sphaericus (MTCC 7542). Enzym. Res. 2010; 2010, 306785.
DR Kashyap, S Chandra, A Kaul and R Tewari. Production, purification and characterization of pectinase from a Bacillus sp. DT7. World J. Microbiol. Biotechnol. 2000; 16, 277-82.
KC Sudeep, J Upadhyaya, DR Joshi, B Lekhak, DK Chaudhary, BR Pant, TR Bajgai, R Dhital, S Khanal, N Koirala and VV Raghavan. Production, characterization, and industrial application of pectinase enzyme isolated from fungal strains. Fermentation 2020; 6, 59.
N, Ogbonnaya and EI Chukwuemeka. Production of polygalacturonase by Aspergillus niger BC23 isolated from Irvingia gabonensis (African mango) fruit. Hemijska Industrija 2016; 70, 717-24.
YP Paudel, C Lin, Z Shen and W Qin. Characterization of pectin depolymerising exo polygalacturonase by Bacillus sp. HD2 isolated from the gut of Apis mellifera L. Microbiol. Discov. 2015; 3, 2.
S Koser, Z Anwar, Z Iqbal, A Anjum, TT Aqil, S Mehmood and M Irshad. Utilization of Aspergillus oryzae to produce pectin lyase from various agro-industrial residues. J. Rad. Res. Appl. Sci. 2014; 7, 327-32.

Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2022 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






