Influences of Organic Additives on Asymbiotic Seed Germination of Dendrobium cruentum Rchb. f. for In Vitro Micropropagation
DOI:
https://doi.org/10.48048/tis.2023.4181Keywords:
Dendrobium cruentum Rchb. f., Asymbiotic seed germination, Micropropagation, Organic additives, Greenhouse acclimatizationAbstract
Dendrobium cruentum Rchb. f., a commercial orchid in Thailand, faces the risk of extinction and should be conserved, and propagated for commercial. Therefore, the aim of this research was to identify a suitable VW medium supplemented with organic additives for the micropropagation of D. cruentum through asymbiotic seed germination. The VW media supplemented with separate organic additives (coconut water (CW) (100 mL/L), ‘Hom Thong’ banana homogenate (BH) (1000 mg/L), and potato homogenate (PH) (1000 mg/L) or its combination was determined, and the seed germination rate (%), protocorm formation, and plantlet development were observed. The results showed that a combination of BH and PH resulted in the highest seed germination (94.69 ± 1.55 %) when compared to basal VW (control). Protocorm formation was highest with CW alone (47.38 ± 7.17 %) and a combination of CW and PH (52.13 ± 6.20 %). Additionally, after 10 weeks of culture, supplementation with BH yielded the highest number of plantlets. However, after 24 weeks of culture, each organic additives gave the different developments of healthy plantlets. To investigate suitable potting media for acclimatization of well-grown plantlets in greenhouse conditions, 2 pieces of foam, dried coconut husk, and a combination of peat moss: Sphagnum moss: Pumice: Perlite (3:1:1:1) were investigated. The highest survival rate percentage was found in foam-planted plantlets (80 %). Ultimately, the effectively simple mediums containing organic additives and a one-step protocol were provided for the micropropagation of D. cruentum via seeds, and the derived information could be used for D. cruentum conservation and production.
HIGHLIGHTS
- The effects of organic additives on asymbiotic seed germination for in vitro micropropagation of Dendrobium cruentum Rchb. f. was firstly reported
- The media supplemented with organic additives yield significantly the highest seed germination, protocorm formation, and plantlet formation and development of D. cruentum when compared to basal media
- In greenhouse conditions, 2 pieces of foam gave the highest survival rate percentage of plantlets (80 %)
- The practical and beneficial media supplemented with organic additives for micropropagation of D. cruentum was provided
GRAPHICAL ABSTRACT 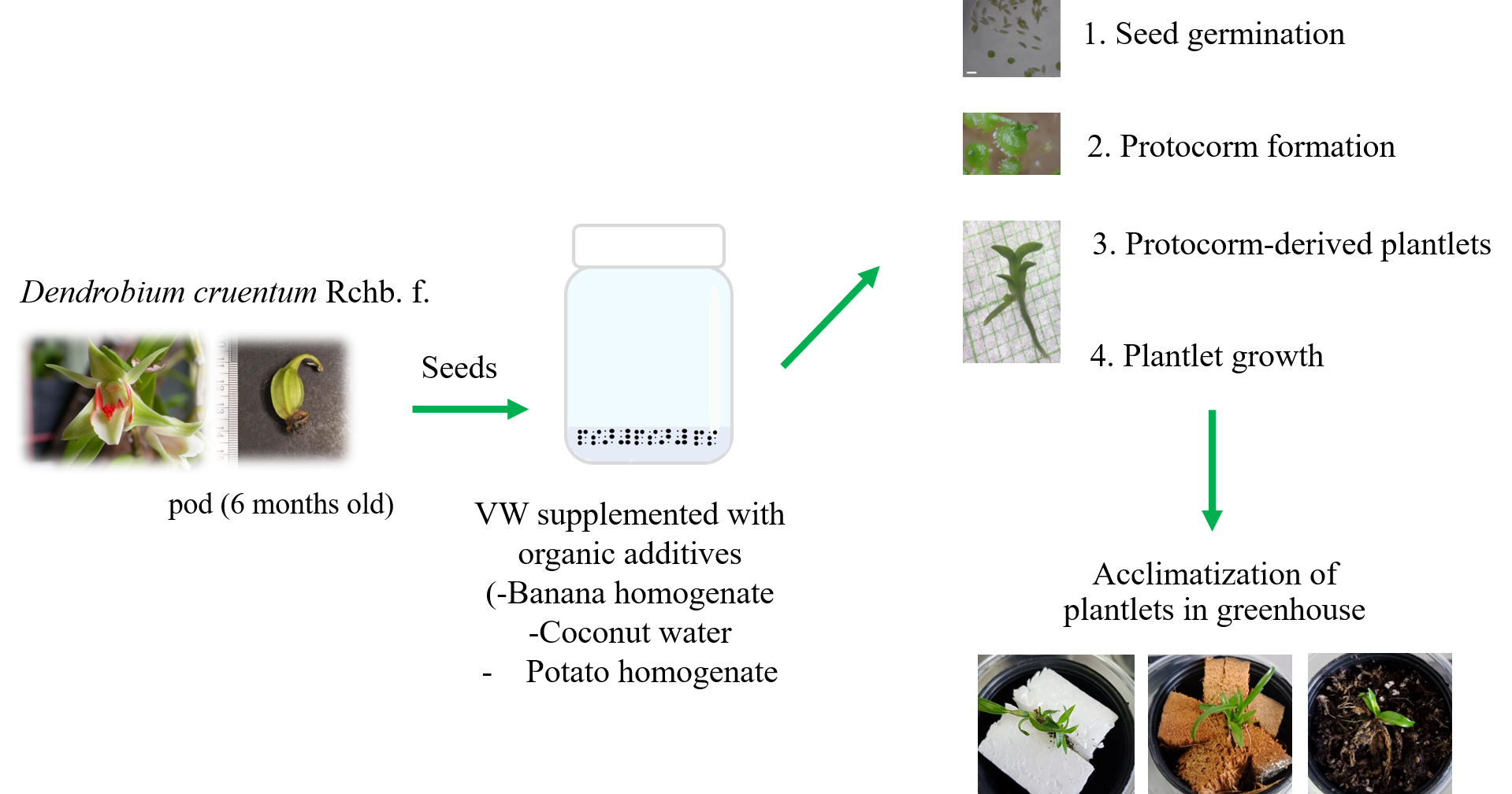
Downloads
References
C Hotrawaisaya, W Chandraprakaikul and N Suthikarnarunai. Performance improvement by logistics collaboration management model for orchid flower industry in Thailand. Eng. Manag. Res. 2014; 3, 52-68.
P Shekarriz, M Kafi, S Dianati Deilamy and M Mirmasoumi. Coconut water and peptone improve seed germination and protocorm like body formation of hybrid Phalaenopsis. Agr. Sci. Dev. 2014; 3, 317-22.
B Long, AX Niemiera, Z Cheng and C Long L. In vitro propagation of four threatened Paphiopedilum species (Orchidaceae). Plant Cell Tiss. Org. 2010; 101, 151-62.
S Zhang, Y Yang, J Li, J Qin, W Zhang, W Huang and H Hu. Physiological diversity of orchids. Plant Divers. 2018; 40, 196-208.
S Sangdum, K Thammasiri, N Chuenboonngarm and K Sopalun. Micropropagation of Dendrobium cruentum Rchb. f., a rare Thai orchid species. Acta Hortic. 2017; 1167, 69-74.
JATD Silva, GB Kerbauy, S Zeng, Z Chen and J Duan. In vitro flowering of orchids. Crit. Rev. Biotechnol. 2014; 34, 56-76.
ESW Utami and S Hariyanto. Organic compounds: Contents and their role in improving seed germination and protocorm development in orchids. Int. J. Agron. 2020; 2, 1-12.
TD Thomas and A Michael. High-frequency plantlet regeneration and multiple shoot induction from cultured immature seeds of Rhynchostylis retusa Blume, an exquisite orchid. Plant Biotechnol. Rep. 2007; 1, 243-9.
H Piri, P Pathak and RK Bhanwra. Asymbiotic germination of immature embryos of a medicinally important epiphytic orchid Acampe papillosa (Lindl.) Lindl. Afr J Biotechnol. 2013; 12, 162-7.
YS Huh, JK Lee, SY Nam, KY Paek and GU Suh. Improvement of asymbiotic seed germination and seedling development of Cypripedium macranthos Sw. with organic additives. J. Plant Biotechnol. 2016; 43, 138-45.
S Zeng, K Wu, JATD Silva, J Zhang, Z Chen, N Xia and J Duan. Asymbiotic seed germination, seedling development and reintroduction of Paphiopedilum wardii Sumerh, an endangered terrestrial orchid. Sci Hortic. 2012; 138, 198-209.
MO Islam, M Akter and AKMA Prodhan. Effect of potato extract on in vitro seed germination and seedling growth of local Vanda roxburgii orchid. J. Bangladesh Agril. Univ. 2011; 9, 211-5.
B Bakar, MA Latip and JA Gansau. Asymbiotic germination and seedling development of Dimorphics lowii (Orchidaceae). Asian J. Plant Biol. 2014; 2, 28-33.
G Pereira, V Albornoz, C Romero, S Lara, M Sanchezolate, D Rios and C Atala. Asymbiotic germination in three Chloraea species. (Orchidaceae) from Chile. Gayana Bot. 2017, 74, 131-9.
ESW Utami, S Hariyanto and YSW Manuhara. In vitro propagation of the endangered medicinal orchid, Dendrobium lasianthera J.J.Sm through mature seed culture. Asian Pac. J. Trop. Biomed. 2017; 7, 406-10.
MM Hossain. Asymbiotic seed germination and in vitro seedling development of Epidendrum ibaguense Kunth. (Orchidaceae). Afr. J. Biotechnol. 2008; 7, 3614-9.
MM Hossain and R Dey. Multiple regeneration pathways in Spathoglottis plicata Blume-a study in vitro. S. Afr. J. Bot. 2013; 85, 56-62.
L Buyun, A Lavrentyeva, L Kovalska and R Ivannikov. In vitro germination of seeds of some rare tropical orchids. Acta Univ. Latviensis Biol. 2004; 676, 159-62.
D Srivastava, MC Gayatri and SK Sarangi. In vitro seed germination and plant regeneration of an epiphytic orchid Aerides ringens (Lindl.) Fischer. Indian J. Biotechnol. 2015; 14, 574-80.
N Kananont, R Pichyangkura, S Chanprame, S Chadchawan and P Limpanavech. Chitosan specificity for the in vitro seed germination of two Dendrobium orchids (Asparagales: Orchidaceae). Sci. Hortic. 2010; 124, 239-47.
D David, R Jawan, H Marbawi and JA Gansau. Organic additives improves the in vitro growth of native orchid Vanda helvola Blume. Not. Sci. Biol. 2015; 7, 192-7.
S Muthukrishnan, TS Kumar and MV Rao. Effects of different media and organic additives on seed germination of Geodorum densiflorum (Lam) Schltr.: An endangered orchid. Int. J. Sci. Res. 2013; 2, 23-6.
AG Jualang, D Devina, M Hartinie, JS Sharon and J Roslina. Asymbiotic seed germination and seedling development of Vanda dearei. Malays. Appl. Biol. 2014; 43, 25-33.
SF Lo, SM Nalawade, CL Kuo, CL Chen and HS Tsay. Asymbiotic germination of immature seeds, plantlet development and ex vitro establishment of plants of Dendrobium tosaense Makino - a medicinally important orchid. Vitro Cell. Dev. Biol. Plant 2004; 40, 528-35.
N Nambiar, CS Tee and M Maziah. Effects of organic additives and different carbohydrate sources on proliferation of protocorm-like bodies in Dendrobium Alya Pink. Plant Omics. 2012; 5, 10-8.
MO Islam, MS Islam and MA Saleh. Effect of banana extract on growth and development of protocorm like bodies in Dendrobium sp. orchid. Agriculturists 2015; 13, 101-8.
PA Tantasawat, A Khairum, K Arsakit, O Poolsawat, P Pornbungkerd and C Kativat. Effects of different culture media on growth and proliferation of Dendrobium ‘Earsakul’ protocorm-like bodies. HortTechnology 2015; 25, 681-6.
B Mala, K Kuegkong, N Sa-ngiaemsri and S Nontachaiyapoom. Effect of germination media on in vitro symbiotic seed germination of three Dendrobium orchids. S. Afr. J. Bot. 2017; 112, 521-6.
RLM Pierik, PA Sprenkels, BVD Harst and QGVD Mers. Seed germination and further development of plantlets of Paphiopedilum ciliolare Pfitz. in vitro. Sci. Hortic. 1988; 34, 139-53.
JATD Silva, M Chan, T Sanjaya, ML Chai and M Tanaka. Priming abiotic factors for optimal hybrid Cymbidium (Orchidaceae) PLB and callus induction, plantlet formation, and their subsequent cytogenetic stability analysis. Sci. Hortic. 2006; 109, 368-78.
MO Islam, M Akter and AKMA Prodhan. Effect of potato extract on in vitro seed germination and seedling growth of local Vanda roxburgii orchid. J. Bangladesh Agril. Univ. 2011; 9, 211-5.
D Nicomrat and J Anantasaran. A reliable homemade tissue culture protocol for Dendrobium orchid cultivation. Appl. Mech. Mater. 2015; 804, 227-30.
JWH Yong, L Ge, YF Ng and SN Tan. chemical composition and biological properties of coconut (Cocos nucifera L.) water. Molecules 2009; 14, 5144-64.
P Gnasekaran, R Xavier, RS Uma and S Sreeramanan. A study on the use of organic additives on the protocorm-like bodies (PLBs) growth of Phalaenopsis violacea orchid. J. Phytol. 2010; 2, 29-33.
U Santoso, K Kubo, T Ota, T Tadokoro and A Maekawa. Nutrient composition of kopyor coconuts (Cocos nucifera L.). Food Chem. 1996; 57, 299-304.
RA Khalifah. Gibberellin-like substances from the developing banana. Plant Physiol. 1966; 41, 771-3.
RA Khalifah. Indolyl-3-acetic acid from the developing banana. Nature 1996; 212, 1471-2.
AA Al-Khateeb. Regulation of in vitro bud formation of date palm (Phoenix dactylifera L.) cv. Khanezi by different carbon sources. Bioresource Tech. 2008; 99, 6550-5.
D Mohapatra, S Mishra and N Sutar. Banana and its by product utilisation: An overview. J. Sci. Ind. Res. 2010; 69, 323-9.
K Sittisuanjik and K Sriruksa. Comparative study on antioxidant activity, total phenolic contents and physicochemical properties of banana flours (in Thai). Agr. Sci. J. 2013; 44, 213-6.
HRP Fernando, V Srilaong, N Pongprasert, P Boonyaritthongchai and P Jitareerat. Changes in antioxidant properties and chemical composition during ripening in banana variety ‘Hom Thong’ (AAA group) and ‘Khai’ (AA group). Int. Food Res. J. 2014; 21, 749-54.
P Gnasekaran, R Poobathy, M Mahmood, MR Samian and S Subramaniam. Effects of complex organic additives on improving the growth of PLBs of Vanda Kasem’s delight. Aust. J. Crop Sci. 2012; 6, 1245-8
V Bartova and J Barta. Chemical composition and nutritional value of protein concentrates isolated from potato (Solanum tuberosum L.) fruit juice by precipitation with ethanol or ferric chloride. J. Agr. Food Chem. 2009; 57, 9028-34.
Z Molnar, E Virag and V Ordog. Natural substances in tissue culture media of higher plants. Acta Biol. Szeged. 2011; 55, 123-7.
JATD Silva, MM Hossain, M Sharma, J Dobránszki, JC Cardoso and Z Songjun. Acclima tization of in vitro-derived Dendrobium. Hortic. Plant J. 2017; 3, 110-24.
MR Evans, JN Smith and RA Cloyd. Fungus gnat population development in coconut coir and Sphagnum peat-based substrates. Hortechnology 1998; 8, 406-9.
B Winarto and F Rachmawati. In vitro propagation protocol of Dendrobium ‘Gradita 31’ via protocorm like bodies. Thammasat Int. J. Sci. Tech. 2013; 18, 54-68.
NR Nayak, S Sahoo, S Patnaik and SP Rath. Establishment of thin cross section (TCS) culture method for rapid micropropagation of Cymbidium aloifolium (L.) Sw. and Dendrobium nobile Lindl. (Orchidaceae). Sci. Hortic. 2002; 94, 107-16.
KP Martin and J Madassery. Rapid in-vitro propagation of Dendrobium hybrids through direct shoot formation from foliar explants, and protocorm-like bodies. Sci. Hortic. 2006; 108, 95-9.
S Dohling, S Kumaria and P Tandon. Multiple shoot induction from axillary bud cultures of the medicinal orchid, Dendrobium longicornu. AoB Plants 2012; 2012, pls032.

Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2022 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






