Antiproliferative and Cytotoxic Efficacy of 10-Hydroxy-2-Decenoic Acid, Compared to Doxorubicin, on MCF-7 Breast Cancer Cells
DOI:
https://doi.org/10.48048/tis.2021.409Keywords:
Apoptosis, Cell cycle arrest, C-MYC, 10-hydroxy-2-decenoic acid, NRF2, HO-1Abstract
Exploration of effective chemotherapy is needed for cancer treatment. 10-hydroxy-2-decenoic acid (10-H2DA), a unique fatty acid from royal jelly (RJ), is reported to have antitumor activities. However, its mechanisms remain under-examined. This study investigated the antiproliferative and cytotoxic efficacy of 10-H2DA treatments and their underlying mechanisms, compared to doxorubicin (DXR), on MCF-7 breast cancer cells. The antiproliferative effect was determined using the MTS tetrazolium assay. Cytotoxic activity was performed using a modified MTS assay. Cell cycle progression and cell apoptosis were analyzed by flow cytometry. Pivotal protein expressions were detected by Western blot. Results revealed that 125 µg/mL 10-H2DA treatment significantly inhibited cancer cell growth by 65 %, better than 0.54 µg/mL DXR treatment (48 %), compared to the medium control (p<0.05). The 50 % lethal concentration (LC50) values of 10-H2DA were 190 µg/mL, representing cytotoxic activity. The underlying antiproliferative and cytotoxic mechanisms of 125 µg/mL 10-H2DA treatment demonstrated that it extensively suppressed c-MYC/BAX and slightly activated p53/BAX, leading to G0/G1 cell cycle arrest (decreased cyclin D1 and CDK4) and cell apoptosis (decreased BCL2/BAX). It slightly limited lifespan extension (decreased hTERT/BAX). Nevertheless, it strongly activated HO-1/BAX and NRF2/BAX, possibly inducing chemoresistance and cell invasion later on. Our findings suggested that 10-H2DA treatments induced antiproliferative effects on MCF-7 breast cancer cells via suppression of c-MYC, CDK4, and cyclin D1, leading to cell cycle arrest and cell apoptosis. However, long-term treatment may increase chemoresistance and cell invasion due to induction of antioxidative power, NRF2/BAX, and HO-1/BAX. Therefore, aggressive treatment for a short period would be recommended for using 10-H2DA as a chemotherapeutic compound to prevent chemoresistance and cell invasion. Further long-term in vitro and in vivo studies are necessary to confirm its strength and weakness.
HIGHLIGHTS
- 10-hydroxy-2-decenoic acid (10-H2DA), a marker royal jelly acid, effectively inhibited MCF-7 breast cancer cells proliferation and induced cytotoxicity
- The inhibitory mechanisms involved the high suppression of c-MYC, cyclin D1, and CDK4, which induced cell cycle arrest and cell apoptosis
- 10-H2DA treatment at proper dose induced high antioxidative potency via activation of NRF2/BAX and HO-1/BAX
- Limitation of 10-H2DA treatment is that it might induce chemoresistance
GRAPHICAL ABSTRACT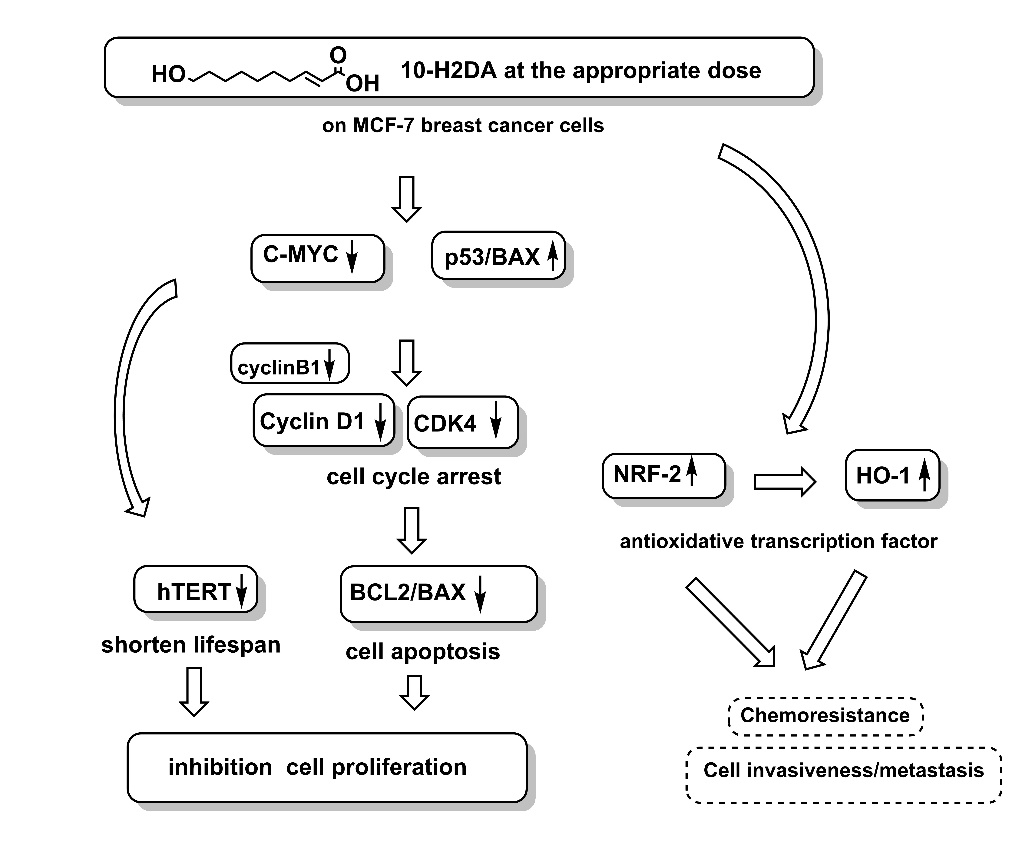
Downloads
References
F Bray, J Ferlay, I Soerjomataram, RL Siegel, LA Torre and A Jemal. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Canc. J. Clin. 2018; 68, 394-424.
D Hanahan and RA Weinberg. The hallmarks of cancer. Cell 2000; 100, 57-70.
ML Tan, PFM Choong and CR Dass. Review: Doxorubicin delivery systems based on chitosan for cancer therapy. J. Pharm. Pharmacol. 2009; 61, 131-42.
GF Townsend, JF Morgan, S Tolnai, B Hazlett, HJ Morton and RW Shuel. Studies on the in vitro antitumor activity of fatty acids. I. 10-Hydroxy-2-decenoic acid from royal jelly. Canc. Res. 1960; 20, 503-10.
F Fratini, G Cilia, S Mancini and A Felicioli. Royal Jelly: An ancient remedy with remarkable antibacterial properties. Microbiol. Res. 2016; 192, 130-41.
YC Yang, WM Chou, DA Widowati, IP Lin and CC Peng. 10-hydroxy-2-decenoic acid of royal jelly exhibits bactericide and anti-inflammatory activity in human colon cancer cells. BMC Compl. Alternative Med. 2018; 18, 202.
Y Honda, Y Fujita, H Maruyama, Y Araki, K Ichihara, A Sato, T Kojima, M Tanaka, Y Nozawa, M Ito and S Honda. Lifespan-extending effects of royal jelly and its related substances on the nematode Caenorhabditis elegans. PLoS One 2011; 6, e23527.
C Bincoletto, S Eberlin, CAV Figueiredo, MB Luengo and MLS Queiroz. Effects produced by royal jelly on haematopoiesis: relation with host resistance against Ehrlich ascites tumour challenge. Int. Immunopharmacol. 2005; 5, 679-88.
Y Miyata, K Araki, K Ohba, T Mastuo, Y Nakamura, T Yuno, Y Mukai, A Otsubo, K Mitsunari, Y Mochizuki and H Sakai. Oral intake of royal jelly improves anti-cancer effects and suppresses adverse events of molecular targeted therapy by regulating TNF-alpha and TGF-beta in renal cell carcinoma: A preliminary study based on a randomized double-blind clinical trial. Mol. Clin. Oncol. 2020; 13, 29.
S Pengpanich, D Srisupabh and WU Tanechpongtamb. Potential role of royal jelly and 10-hydroxy-2-decenoic acid as metastasis inhibitors in triple-negative breast cancer cells. J. Med. Assoc. Thailand 2019; 102, 7-24.
M Nakaya, H Onda, K Sasaki, A Yukiyoshi, H Tachibana and K Yamada. Effect of royal jelly on bisphenol A-induced proliferation of human breast cancer cells. Biosci. Biotechnol. Biochem. 2007; 71, 253-5.
DA Gewirtz. A critical evaluation of the mechanisms of action proposed for the antitumor effects of the anthracycline antibiotics adriamycin and daunorubicin. Biochem. Pharmacol. 1999; 57, 727-41.
DM Miller, SD Thomas, A Islam, D Muench and K Sedoris. c-Myc and cancer metabolism. Clin. Canc. Res. 2012; 18, 5546-53.
KJ Wu, C Grandori, M Amacker, N Simon-Vermot, A Polack, J Lingner and R Dalla-Favera. Direct activation of TERT transcription by c-MYC. Nat. Genet. 1999; 21, 220-4.
JI Daksis, RY Lu, LM Facchini, WW Marhin and LJ. Penn. Myc induces cyclin D1 expression in the absence of de novo protein synthesis and links mitogen-stimulated signal transduction to the cell cycle. Oncogene 1994; 9, 3635-45.
CW Greider. Telomerase activation. One step on the road to cancer? Trends Genet. 1999; 15,109-12.
L Jiang, N Kon, T Li, SJ Wang, T Su, H Hibshoosh, R Baer and W Gu. Ferroptosis as a p53-mediated activity during tumour suppression. Nature 2015; 520, 57-62.
J Yuan, H Lan, X Jiang, D Zeng and S Xiao. Bcl2 family: Novel insight into individualized therapy for ovarian cancer (Review). Int. J. Mol. Med. 2020; 46, 1255-65.
ME Maes, CL Schlamp, RW Nickells. Live-cell imaging to measure BAX recruitment kinetics to mitochondria during apoptosis. PLoS One 2017; 12, e0184434.
SJ Korsmeyer, JR Shutter, DJ Veis, DE Merry and ZN Oltvai. Bcl-2/Bax: A rheostat that regulates an anti-oxidant pathway and cell death. Semin. Canc. Biol. 1993; 4, 327-32.
JD Hayes and AT Dinkova-Kostova. The Nrf2 regulatory network provides an interface between redox and intermediary metabolism. Trends Biochem. Sci. 2014; 39, 199-218.
MC Jaramillo and DD Zhang. The emerging role of the Nrf2-Keap1 signaling pathway in cancer. Gene. Dev. 2013; 27, 2179-91.
N Tracey, H Creedon, AJ Kemp, J Culley, M Muir, T Klinowska and VG Brunton. HO-1 drives autophagy as a mechanism of resistance against HER2-targeted therapies. Breast Canc. Res. Treat. 2020; 179, 543-55.
H Shen, Y Yang, S Xia, B Rao, J Zhang and J Wang. Blockage of Nrf2 suppresses the migration and invasion of esophageal squamous cell carcinoma cells in hypoxic microenvironment. Dis. Esophagus 2014; 27, 685-92.
P Dong, C Zhang, BT Parker, L You and B Mathey-Prevot. Cyclin D/CDK4/6 activity controls G1 length in mammalian cells. PLoS One 2018; 13, e0185637.
G Tchakarska and B Sola. The double dealing of cyclin D1. Cell Cycle 2020; 19, 163-78.
W Jiang, H Huang, L Ding, P Zhu, H Saiyin, G Ji, J Zuo, D Han, Y Pan, D Ding, X Ma, Y Zhang, J Wu, Q Yi, JO Liu, H Huang, Y Dang and L Yu. Regulation of cell cycle of hepatocellular carcinoma by NF90 through modulation of cyclin E1 mRNA stability. Oncogene 2015; 34, 4460-70.
B Strauss, A Harrison, PA Coelho, K Yata, M Zernicka-Goetz and J Pines. Cyclin B1 is essential for mitosis in mouse embryos, and its nuclear export sets the time for mitosis. J. Cell. Biol. 2018; 217, 179-93.
AL Niles, RA Moravec and TL Riss. Update on in vitro cytotoxicity assays for drug development. Expet. Opin. Drug Discov. 2008; 3, 655-69.
SL Holbeck. Update on NCI in vitro drug screen utilities. Eur. J. Canc. 2004; 40, 785-93.
M Lenzi, M Malaguti, V Cocchi, S Hrelia and P. Hrelia. Castanea sativa Mill. bark extract exhibits chemopreventive properties triggering extrinsic apoptotic pathway in Jurkat cells. BMC Compl. Alternative Med. 2017; 17, 251.
SNSA Rahman, NAWahab and SNA Malek. In Vitro Morphological Assessment of Apoptosis Induced by Antiproliferative Constituents from the Rhizomes of Curcuma zedoaria. Evid. Base. Compl. Alternative Med. 2013; 2013, 257108.
W Jenkhetkan, S Thitiorul, C Jansom and T Ratanavalachai. Molecular and cytogenetic effects of Thai royal jelly: modulation through c-MYC, h-TERT, NRF2, HO-1, BCL2, BAX and cyclins in human lymphocytes in vitro. Mutagenesis 2017; 32, 525-31.
W Jenkhetkan, S Thitiorul, C Jansom and T Ratanavalachai. Genoprotective effects of Thai royal jelly against doxorubicin in human lymphocytes in vitro. Nat. Product Comm. 2018; 13, 79-84.
XJ Wang, Z Sun, NF Villeneuve, S Zhang, F Zhao, Y Li, W Chen, X Yi, W Zheng, GT Wondrak, PK Wong and DD Zhang. Nrf2 enhances resistance of cancer cells to chemotherapeutic drugs, the dark side of Nrf2. Carcinogenesis 2008; 29, 1235-43.
Downloads
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.







