Probe Ultrasonic-Assisted Extraction of Andrographolide Rich Extract from Andrographis paniculata: Processing Parameters Optimization Using Response Surface Methodology
DOI:
https://doi.org/10.48048/tis.2022.385Keywords:
Andrographis paniculata, Andrographolide, Probe ultrasonic-assisted extraction, Response surface methodologyAbstract
The conventional extraction method has low extraction efficiency. This study aims to optimize the ultrasonic-assisted extraction conditions of andrographolide from A. paniculata. A ½ inch ultrasonic probe sonicator was used to extract A. paniculata. The effects of extraction time (X1), duty cycle (X2) and amplitude (X3) on the yield of andrographolide (Y) were investigated by using response surface methodology. Experiments were designed according to 3-level 3-factor Box Behnken Design. The concentrations of andrographolide were quantified by high-performance liquid chromatography. Results of analysis of variance showed the data were well fitted with the 2nd-order polynomial equation with a R2 value of 0.9969 and p-value less than 0.005. Optimum extraction conditions were found to be at 5 min, 11 % duty cycle, and 66 A. A checkpoint experiment was carried out based on the optimum conditions, validated highest andrographolide was 3.50 ± 0.17 w/w%. Response surface methodology was successfully applied to optimize the extraction processing parameters for yielding the andrographolide-rich A. paniculata extract. This study could be relevant for future ultrasonic-assisted extraction scale-up of andrographolide.
HIGHLIGHTS
- Andrographis paniculata was extracted using ultrasonic probe sonicator
- Variables studied were time, duty cycle, and amplitude
- Extraction parameters were sucessfully optimized by response surface methodology
- 50 ± 0.17 w/w% of andrographolide was extracted.
GRAPHICAL ABSTRACT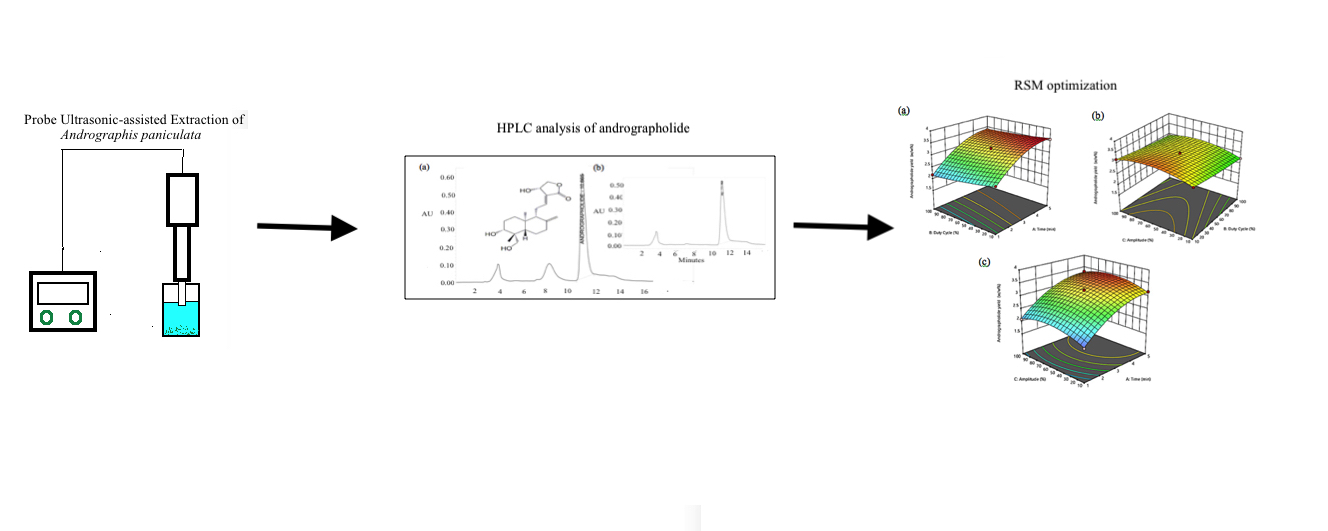
Downloads
References
L Li, GGL Yue, JKM Lee, ECW Wong, KP Fung, J Yu, CBS Lau and PWY Chiu. Gene expression profiling reveals the plausible mechanisms underlying the antitumor and antimetastasis effects of Andrographis paniculata in esophageal cancer. Phytother. Res. 2018; 32, 1388-96.
HC Liao, YJ Chou, CC Lin, SH Liu, A Oswita, YL Huang, YL Wang, JL Syu, CM Sun, CM Leu and SL Fu. Andrographolide and its potent derivative exhibit anticancer effects against imatinib-resistant chronic myeloid leukemia cells by downregulating the Bcr-Abl oncoprotein. Biochem. Pharmacol. 2019; 163, 308-20.
AK Mishra, A Mishra and P Chattopadhyay. Herbal cosmeceuticals for photoprotection from Ultraviolet B radiation: A review. Trop. J. Pharm. Res. 2011; 10, 351-60.
Bastiana, A Widyawaruyanti, H Ilmi, L Tumewu, B Prasetyo, AF Hafid and Aryati. A tablet derived from Andrographis paniculata complements dihydroartemisinin-piperaquine treatment of malaria in pregnant mice. J. Basic Clin. Physiol. Pharmacol. 2021; 33, 175-83.
RA Burgos, P Alarcón, J Quiroga, C Manosalva and J Hancke. Andrographolide, an anti-inflammatory multitarget drug: All roads lead to cellular metabolism. Molecules 2021; 26, 5.
E Mussard, A Cesaro, E Lespessailles, B Legrain, S Berteina-Raboin and H Toumi. Andrographolide, a natural antioxidant: An update. Antioxidants 2019; 8, 571.
HS Pati, DD Jadhav, A Paul, FA Mulani, SJ Karegaonkar and HV Thulasiram. Regioselective and efficient enzymatic synthesis of antimicrobial andrographolide derivatives. Bioorg. Med. Chem. Lett. 2018; 28, 1132-7.
NA Murugan, CJ Pandian and J Jeyakanthan. Computational investigation on Andrographis paniculata phytochemicals to evaluate their potency against SARS-CoV-2 in comparison to known antiviral compounds in drug trials. J. Biomol. Struct. Dyn. 2020; 39, 4415-26.
S Kaushik, L Dar, S Kaushik and JP Yadav. Identification and characterization of new potent inhibitors of dengue virus NS5 proteinase from Andrographis paniculata supercritical extracts on in animal cell culture and in silico approaches. J. Ethnopharmacol. 2021; 267, 113541.
H Shi, W Guo, H Zhu, M Li, COL Ung, H Hu and S Han. Cost-effectiveness analysis of Xiyanping injection (andrographolide sulfonate) for treatment of adult community acquired pneumonia: A retrospective, propensity score-matched cohort study. Evid. Based Complement. Alternat. Med. 2019; 2019, 4510591.
J Lu, Y Ma, J Wu, H Huang, X Wang, Z Chen, J Chen, H He and C Huang. A review for the neuroprotective effects of andrographolide in the central nervous system. Biomed. Pharmacother. 2019; 117, 109078.
A Tandoh, CA Danquah, PPS Ossei, CK Benneh, WG Ayibor and E Woode. Protective effects of andrographolide against diclofenac-induced gastric damage. Sci. Afr. 2021; 13, e00944.
AJ Salunkhe and RN Patil. Hepatoprotective effect of ethanolic extract of Andrographis paniculata against thioacetamide induced toxicity in the liver of albino rats. Res. J. Life Sci. Bioinform. Pharm. Chem. Sci. 2018; 4, 549-59.
World Health Organization. WHO monographs on selected medicinal plants. Vol II. AITPBS Publications and Distributors, Geneva, 2002, p. 12-24.
Y Dai, SR Chen, L Chai, J Zhao, Y Wang and Y Wang. Overview of pharmacological activities of Andrographis paniculata and its major compound andrographolide. Crit. Rev. Food Sci. Nutr. 2019; 59, S17-S29.
S Sharma and YP Sharma. Comparison of different extraction methods and HPLC method development for the quantification of andrographolide from Andrographis paniculata (Burm. f.) Wall. ex Nees. Ann. Phytomed. 2018; 7, 119-30.
PR Rao and VK Rathod. Microwave assisted three phase extraction of andrographolide from Andrographis paniculata. J. Biol. Act. Prod. Nat. 2019; 9, 215-26.
AC Kumoro, M Hasan and H Singh. Extraction of andrographolide from Andrographis paniculata dried leaves using supercritical CO2 and ethanol mixture. Ind. Eng. Chem. Res. 2018; 58, 742-51.
UI Abubakar, LS Chua, NF Musa, S Ngadiran and R Aziz. Influence of process variables for green solid-liquid extraction of andrographolide from Andrographis paniculata. J. Mater. Environ. Sci. 2021; 12, 137-52.
VV Chia, SF Pang and J Gimbun. Mass spectrometry analysis of auxiliary energy-induced terpenes extraction from Andrographis paniculata. Ind. Crops Prod. 2020; 155, 112828.
K Kumar, S Srivastav and VS Sharanagat. Ultrasound assisted extraction (UAE) of bioactive compounds from fruit and vegetable processing by-products: A review. Ultrason. Sonochem. 2021; 70, 105325.
C Wen, J Zhang, H Zhang, CS Dzah, M Zandile, Y Duan, H Ma and X Luo. Advances in ultrasound assisted extraction of bioactive compounds from cash crops: A review. Ultrason. Sonochem. 2018; 48, 538-49.
D Panda and S Manickam. Cavitation technology - the future of greener extraction method: A review on the extraction of natural products and process intensification mechanism and perspectives. Appl. Sci. 2019; 9, 766.
LS Chua, NA Latiff and M Mohamad. Reflux extraction and cleanup process by column chromatography for high yield of andrographolide enriched extract. J. Appl. Res. Med. Aromatic Plants 2016; 3, 64-70.
R Foujdar, MB Bera and HK Chopra. Optimization of process variables of probe ultrasonic‐assisted extraction of phenolic compounds from the peel of Punica granatum Var. Bhagwa and it’s chemical and bioactivity characterization. J. Food Process. Preserv. 2020; 44, e14317.
AC Kumoro, M Hasan and H Singh. Effects of solvent properties on the Soxhlet extraction of diterpenoid lactones from Andrographis paniculata leaves. ScienceAsia 2009; 35, 306-9.
S Ziembowicz, M Kida and P Koszelnik. Sonochemical formation of hydrogen peroxide. In: Proceedings of the 2nd International Electronic Conference on Water Sciences, Basel, Switzerland. 2017, p. 188.
P Phattanawasin, U Sotanaphun, J Burana-Osot and N Piyapolrungroj. Isolation and characterization of the acid and base degradation products of andrographolide. Phamarzie 2018; 73, 559-62.
J Burke. Solubility parameters: Theory and application. In: C Jensen (Ed.). AIC book and paper group annual. Vol. III. Washington, DC, 1984, p. 13-58.
VV Chia, SF Pang, S Abdullah, MM Yusoff and J Gimbun. Microwave-assisted extraction of andrographolide, 14-deoxy-11,12-didehydroandrographolide and neoandrographolide from Andrographis paniculata: An optimisation study. Curr. Nutr. Food Sci. 2020; 16, 1318-25.
International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use. ICH guideline Q3C (R6) on impurities: Guideline for Residual Solvents. ICH Harmonised Guideline, Available at: https://database.ich.org/sites/default/files/Q3C-R6_Guideline_Error Correction_2019_0410_0.pdf, accessed January 2020.
R Wongkittipong, L Prat, S Damronglerd and C Gourdon. Solid-liquid extraction of andrographolide from plants-experimental study, kinetic reaction and model. Sep. Purif. Technol. 2004; 40, 147-54.
KL Tan and SH Yeo. Bubble dynamics and cavitation intensity in milli-scale channels under an ultrasonic horn. Ultrason. Sonochem. 2019; 58, 104666.
L Lavilla and C Bendicho. Fundamentals of ultrasound-assisted extraction. In: Water extraction of bioactive compounds. Elsevier, Netherlands, 2017, p. 291-316.
K Peng, FGF Qin, R Jiang and S Kang. Interpreting the influence of liquid temperature on cavitation collapse intensity through bubble dynamic analysis. Ultrason. Sonochem. 2020; 69, 105253.
M Bhan, S Satija, C Garg, H Dureja and M Garg. Optimization of ionic liquid-based microwave assisted extraction of a diterpenoid lactone-andrographolide from Andrographis paniculata by response surface methodology. J. Mol. Liq. 2017; 229, 161-6.
S Chakraborty, R Uppaluri and C Das. Optimization of ultrasound-assisted extraction (UAE) process for the recovery of bioactive compounds from bitter gourd using response surface methodology (RSM). Food. Bioprod. Process. 2020; 120, 114-22.
RK Rout, A Kumar and PS Rao. A multivariate optimization of bioactive compounds extracted from oregano (Origanum vulgare) leaves using pulsed mode sonication. J. Food. Meas. Charact. 2021; 15, 3111-22.
Y Wang, X Zhang, X Ma, K Zhang, S Li, X Wang, X Liu, J Liu, W Fan, Y Li, Q Li and X Zhu. Study on the kinetic model, thermodynamic and physicochemical properties of Glycyrrhiza polysaccharide by ultrasonic assisted extraction. Ultrason. Sonochem. 2019; 51, 249-57.
T Brás, AFC Paulino, LA Neves, JG Crespo and MF Duarte. Ultrasound assisted extraction of cynaropicrin from Cynara cardunculus leaves: Optimization using the response surface methodology and the effect of pulse mode. Ind. Crops Prod. 2020; 150, 112395.
NA Patience, D Schieppati and DC Boffito. Continuous and pulsed ultrasound pectin extraction from navel orange peels. Ultrason. Sonochem. 2021; 73, 105480.
R Sharma, B Bhunia, A Mondal, TK Bandyopadhyay, I Devi, G Oinam, R Prasanna, G Abraham and ON Tiwari. Statistical optimization of process parameters for improvement of phycobiliproteins (PBPs) yield using ultrasound-assisted extraction and its kinetic study. Ultrason. Sonochem. 2020; 60, 104762.
W Li, H Yang, TE Coldea and H Zhao. Modification of structural and functional characteristics of brewer's spent grain protein by ultrasound assisted extraction. LWT 2021; 139, 110582.
Z Mojerlou and A Elhamirad. Optimization of ultrasound-assisted extraction (UAE) of phenolic compounds from olive cake. J. Food Sci. Technol. 2018; 55, 977-84.
PR More and SS Arya. Intensification of bio-actives extraction from pomegranate peel using pulsed ultrasound: Effect of factors, correlation, optimization and antioxidant bioactivities. Ultrason. Sonochem. 2021; 72, 105423.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2022 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.