Sequestration of Brilliant Green Dye by Coriander Leaf: Isotherm and Kinetic Studies
DOI:
https://doi.org/10.48048/tis.2022.3070Keywords:
Adsorption, Coriander, Pseudo-2nd order, Isotherm, Taguchi optimizationAbstract
Adsorption process has been performed to investigate the potentiality of coriander leaf to sequester brilliant green (BG) dye from the aqueous phase. Coriander leaf of 75-micron size has been characterized by SEM, BET, FTIR etc. In the present study, emphasis has been given on the removal of BG dye under different conditions viz. dose of biomaterial (10.0 - 100.0 mg), pH (2.0 - 12.0) and dye concentration (10.0 - 100.0 mg/L) by Taguchi optimization. The batch experiments have been conducted at 25 ℃ with stirring speed of 120.0 rpm. The kinetic study is performed at two different temperatures, 30 and 40 ℃. The batch adsorption process for sequestration of the dye followed pseudo-2nd order kinetics (R2 = 0.99). The study also reveals that the Freundlich isotherm is the best fitted (R2 = 0.96) model. The adsorption capacity is 68.49 mg/g and the overall removal efficiency is above 80.0 %.
HIGHLIGHTS
- The coriander leaf (75-micron) has been investigated as an effective adsorbent for Brilliant green dye in aqueous environment
- Effects of physico-chemical properties of adsorbent (BET, pZC, zeta potential, bulk density, moisture, ash etc.) and the role of several operating parameters (contact time, pH, dose and concentration) have been investigated
- Langmuir, Freundlich isotherm models and pseudo-second-order kinetic model favour the adsorption phenomena
- L9 orthogonal array (3X3) of Taguchi optimization study with ANOVA analysis predicts the role of control parameters
GRAPHICAL ABSTRACT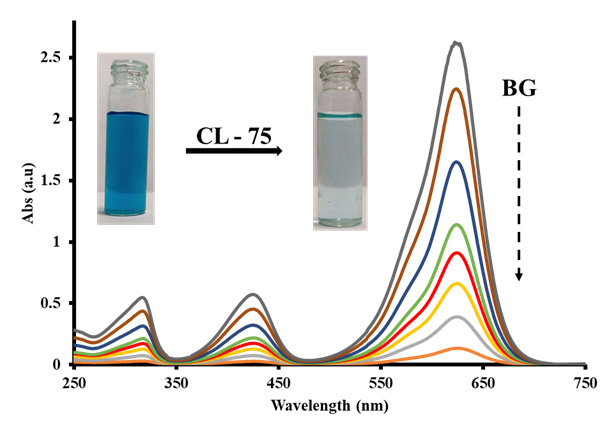
Downloads
References
G Mckay, JF Porter and GR Prasad. The removal of dye colours from aqueous solutions by adsorption on low-cost materials. Water Air Soil Pollut. 1999; 114, 423-38.
A Bhatnagar and AK Minocha. Assessment of the biosorption characteristics of lychee (Litchi chiesas) peel waste for the removal of Acid Blue 25 dye from water. Environ. Technol. 2010; 31, 97-105.
R Sivashankar, AB Sathya, K Vasantharaj and V Sivasubramanian. Magnetic composite an environmental super adsorbent for dye sequestration - A review. Environ. Nanotechnol. Monit. Manag. 2014; 1-2, 36-49.
VK Gupta and Suhas. Application of low-cost adsorbents for dye removal - a review. J. Environ. Manag. 2009; 90, 2313-42.
KAR Rao and MK Uddin. Adsorption properties of coriander seed powder (Coriandrum sativum): Extraction and pre-concentration of Pb (II), Cu (II) and Zn (II) ions from aqueous solution. Adsorp. Sci. Technol. 2010; 30, 127-46.
S Agarwal, VK Gupta, M Ghasemi and J Azimi-Amin. Peganum harmala-L seeds adsorbent for the rapid removal of noxious brilliant green dyes from aqueous phase. J. Mol. Liq. 2017; 231, 296-305.
VK Gupta, PJM Carrott, MMLR Carrott and Suhas. Low-cost adsorbents: Growing approach to wastewater treatment - a review. Crit. Rev. Environ. Sci. Technol. 2009; 39, 783-842.
A Srinivasan and T Viraraghavan. Decolorization of dye wastewaters by biosorbents: A review. J. Environ. Manag. 2010; 91, 1915-29.
LP Nimish, BK Sanjay, MB Nayna and DR Jaimik. Phytopharmacological properties of Coriander Sativum as a potential medicinal tree: An overview. J. Appl. Pharm. Sci. 2011; 1, 20-5.
M Nadeem, FM Anjum, MI Khan, S Tehseen, A El-Ghorab and JI Sultan. Nutritional and medicinal aspects of coriander (Coriandrum sativum L.): A review. Br. Food J. 2013; 115, 743-55.
D Karunasagar, MVB Krishna, SV Rao and J Arunachalam. Removal and preconcentration of inorganic and methyl mercury from aqueous media using a sorbent prepared from the plant Coriandrum sativum. J. Hazard. Mater. 2005; 118, 133-9.
RAK Rao and M Kashifuddin. Adsorption properties of coriander seed powder (Coriandrum sativum): Extraction and pre-concentration of Pb (II), Cu (II) and Zn (II) ions from aqueous solution. Adsorp. Sci. Technol. 2012; 30, 127-46.
R Yahya, SN Taqui, A Hassan, F Khanum and AA Syed. Valorization of nutraceutical industrial coriander seed spent by the process of sustainable adsorption system of acid black 52 from aqueous solution. Int. J. Environ. Res. 2019; 13, 639-59.
BK Nandi, A Goswami and MK Purkait. Adsorption characteristics of brilliant green dye on Kaoli. J. Hazard. Mater. 2009; 161, 387-95.
A Ghazali, M Shirani, A Semnani, V Zare-Shahabadi and M Nekoeinia. Optimization of crystal violet adsorption onto Date palm leaves as a potent biosorbent from aqueous solutions using response surface methodology and ant colony. J. Environ. Chem. Eng. 2018; 6, 3942-50.
SN Taqui, R Yahya, A Hassan, F Khanum and AA Syed. Valorization of nutraceutical industrial coriander seed spent by the process of sustainable adsorption system of Acid Black 52 from aqueous solution. Int. J. Environ. Res. 2019; 13, 639-59.
NK Mondal, A Samanta, S Chakraborty and WA Shaikh. Enhanced chromium (VI) removal using banana peel dust: Isotherms, kinetics and thermodynamics study. Sust. Water Resour. Manage. 2018; 4, 489-97.
Z Rawajfiha and N Nsour. Thermodynamic analysis of sorption isotherms of chromium (VI) anionic species on reed biomass. J. Chem. Thermodyn. 2008; 40, 846-51.
V Katheresan, J Kansedo and SY Lau. Efficiency of various recent wastewater dye removal methods: A review. J. Environ. Chem. Eng. 2018; 6, 4676-97.
A Kausar, M Iqbal, A Javeda, K Aftaba, ZH Nazli, HN Bhatti and S Nouren. Dyes adsorption using clay and modified clay: A review. J. Mol. Liq. 2018; 256, 395-407.
MA Salem, RG Elsharkawy and MF Hablas. Adsorption of brilliant green dye by polyaniline/silver nanocomposite: Kinetic, equilibrium, and thermodynamic studies. Eur. Polym. J. 2016; 75, 577-90.
KV Kumar and S Sivanesan. Prediction of optimum sorption isotherm: Comparison of linear and non-linear method. J. Hazard. Mater. 2005; 126, 198-201.
F Haghseresht and GQ Lu. Adsorption characteristics of phenolic compounds onto coal-reject-derived adsorbents. Energy Fuels 1998; 12, 1100-7.
N Ayawei, AN Ebelegi and D Wankasi. Modelling and interpretation of adsorption isotherms. J. Chem. 2017; 2017, 3039817.
HY Yen and CP Lin. Adsorption of Cd (II) from wastewater using spent coffee grounds by Taguchi optimization. Desalin. Water Treat. 2015; 57, 11154-61.
Downloads
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.







