Synthesis, Characterization, In vitro Antiproliferative and Cytotoxic Effect of Ruthenium(II)-Bipyridine-Benzoylpicolinic Acid Complex on SK-MEL-28 and Normal L6 Cell Lines
DOI:
https://doi.org/10.48048/tis.2022.3040Keywords:
[Ru(bpy)2(bzpic)2]2 complex, SK-MEL-28 and L6 cell lines, MTT assay, Antiproliferative effect, Fluorescence microscopy, Anti-skin cancer activityAbstract
The most seriously spread malignant tumor of melanocytes throughout the body is a melanoma type of skin cancer. SK-MEL-28 cell line is one of a series of melanoma cell lines. The objective of the present investigation is to study the anticancer activity and cytotoxicity of novel [Ru(bpy)2(bzpic)2]2+ (bpy = 2,2ʹ-bipyridine and bzpic = 3-Benzoylpicolinic acid) complex on SK-MEL-28 and normal L6 cell lines using MTT assay method. The morphology of the cancerous SK-MEL-28 cell line at various concentrations of the [Ru(bpy)2(bzpic)2]2+ complex is assessed by 2-fold dilution method, and the in vitro antiproliferative effect of the complex on the cancerous SK-MEL-28 cell line is analyzed by direct microscopic observation method. The percentage viability of the complex on cancerous SK-MEL-28 cell line at various concentrations is determined. The IC50 value of the complex against the SK-MEL-28 cell is found to be 39.109 µg/mL which shows good antiproliferative effect. The IC50 value of the complex against normal L6 cell line is found to be 55.315 µg/mL. The results revealed that the percentage of growth inhibition of the cell is based on dose-dependent manner and the formation of formazan crystal indicates this. The fluorescent microscopy observation clearly determines that the synthesized [Ru(bpy)2(bzpic)2]2+ complex shows late apoptosis on SK-MEL-28 cell line and early apoptosis on L6 cell line. Hence, it is evident that the synthesized complex exhibits good in vitro antiproliferative effect with lower cytotoxicity.
HIGHLIGHTS
- Synthesis of [Ru(bpy)2(bzpic)2]2+ complex
- Direct and fluorescent microscopic observation of [Ru(bpy)2(bzpic)2]2+ complex on SK-MEL-28 and normal L6 cell lines
- In vitro antiproliferative and cytotoxic effects of [Ru(bpy)2(bzpic)2]2+ complex on SK-MEL-28 and normal L6 cell lines
GRAPHICAL ABSTRACT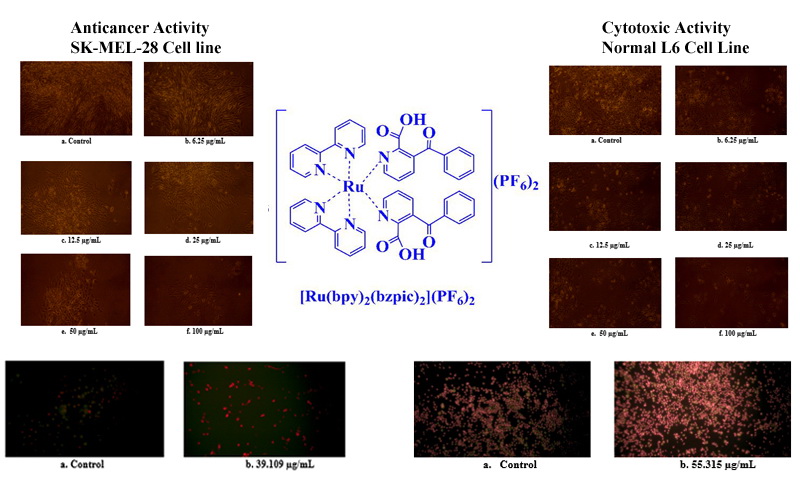
Downloads
References
P Das, N Deshmukh, N Badore, C Ghulaxe and P Patel. A review article on melanoma. J. Pharm. Sci. Res. 2016; 8, 112-7.
E Perera, N Gnaneswaran, N Jennens and RR Sinclair. Malignant Melanoma. Health Care 2014; 2, 1-19.
N Popgeorgiev, B Bonneau, J Prudent and G Gillet. Control of programmed cell death during zebrafish embryonic development. In: Y Bozkurt (Ed.). Recent advances in zebrafish researches. IntechOpen, 2018.
G Fridman, A Shereshevsky, MM Jost, AD Brooks, A Fridman, AGV Vasilets and G Friedman. Floating Electrode dielectric barrier discharge plasma in air promoting apoptotic behavior in melanoma skin cancer cell lines. Plasma Chem. Plasma. Process. 2007; 27, 163-76.
RG Kerry, KE Ukhurebor, S Kumari, GK Maurya, S Patra, B Panigrahi, S Majhi, JR Rout, MDP Rodriguez-Torres, G Das, HS Shin and JK Patra. A comprehensive review on the applications of nano-biosensor-based approaches for non-communicable and communicable disease detection. Biomater. Sci. 2021; 9, 3576-602.
RB Onyancha, UO Aigbe, KE Ukhurebor and PW Muchiri. Facile synthesis and applications of carbon nanotubes in heavy-metal remediation and biomedicals fields: A comprehensive review. J. Mol. Struct. 2021; 1238, 130462.
U Ndagi, N Mhlongo and ME Soliman. Metal complexes in cancer therapy-an update from drug design perspective. Drug Des. Dev. Ther. 2017; 11, 599-616.
JCS Lopes, JL Damasceno, PF Oliveira, APM Guedes, DC Tavares, VM Deflon, NP Lopes, M Pivatto, AA Batista, PIS Maia and GV Poelhsitz. Ruthenium(II) complexes containing anti-inflammatory drugs as ligands: Synthesis, characterization and in vitro cytotoxicity activities on cancer cell lines. J. Braz. Chem. Soc. 2015; 26, 1838-47.
FE Poynton, SA Bright, S Blasco, DC Williams, JM Kelly and T Gunnlaugsson. The development of ruthenium(II) polypyridyl complexes and conjugates for in vitro cellular and in vivo applications. Chem. Soc. Rev. 2017; 46, 7706-56.
A Srishailam, YP Kumar, PV Reddy, N Nambigari, U Vuruputuri, SS Singh and S Satyanarayana. Cellular uptake, cytotoxicity, apoptosis, DNA-binding, photocleavage and molecular docking studies of ruthenium(II) polypyridyl complexes. J. Photochem. Photobiol. B Biol. 2014; 132, 111-23.
C Ravi, RK Vuradi, S Avudoddi, PK Yata, VR Putta, G Srinivas, R Merugu and S Satyanarayana. Synthesis, spectral studies, DNA binding, photocleavage, antimicrobial and anticancer activities of isoindol Ru(II) polypyridyl complexes. Nucleos. Nucleot. Nucleic Acids 2019; 38, 788-806.
I Kostova. Ruthenium complexes as anticancer agents. Curr. Med. Chem. 2006; 13, 1085-107.
Z Incesu, K Bljnkl, G Akalin and NG Karaburun.The effects of some phenanthroline ruthenium(II) complexes on A549 cell proliferation. Turk. J. Pharm. Sci. 2013; 10, 193-204.
L Xu, NJ Zhong, YY Xie, HL Huang, GB Jiang and YJ Liu. Synthesis, characterization, in vitro cytotoxicity, and apoptosis-inducing properties of ruthenium(II) complexes. PloS One 2014; 9, e96082.
RS Grant, SE Coggan and GA Smythe. The physiological action of picolinic acid in the human brain. Int. J. Tryptophan. Res. 2009; 2, 71-9.
P Subramaniam and NT Selvi. Picolinic acid promoted oxidative decarboxylation of phenyl sulfinylacetic acid by Cr(VI). Bull. Chem. Soc. Ethiop. 2016; 30, 137.
PJ Aggett, PK Fenwick and H Kirk. An in vitro study of the effect of picolinic acid on metal translocation across lipid bilayers. J. Nutr. 1989; 119, 1432-7.
P Sengupta, R Dinda, S Gosch and WS Sheldrick. Synthesis and characterization of some ruthenium(II) complexes of N-heterocyclic carboxylic acids x-ray structures of cis-[Ru(PPh3)2(L1)2].2CH3OH and cis-[Ru(PPh3)2(L3H)2] (L1=pyridine-2-carboxylic acid and L3H2=imidazole 4,5-dicarboxylic acid). Polyhedron 2010; 20, 3349-54.
EM Shoukry, NG Hosny, MF Amin and EF Mohamed. Complex formation equilibria of ternary complexes of Cu(II) involving pyridine-2-carboxylic acid and various biologically relevant ligands. Int. J. Curr. Res. Chem. Pharm. Sci. 2016; 3, 32-43.
A Sieroslawska and A Rymuszka. Assessment of the cytotoxic impact of cyanotoxin beta-N-methylamino-L-alanine on a fish immune cell line. Aquat. Toxicol. 2019; 212, 214-21.
JH Zhang, J Yu, WX Li and CP Cheng. Evaluation of Mn2+ stimulated and Zn2+ inhibited apoptosis in rat corpus luteal cells by flow cytometry and fluorochromes staining. Chin. J. Physiol. 1998; 41, 121-6.
S Castellano, C Sun and R Kostelnik. Analysis of the NMR spectrum of pyridine. J. Chem. Phys. 1967; 46, 327.
E Corral, ACG Hotze, HD Dulk, A Leczkowska, A Rodger, MJ Hannon and J Reedijk, Ruthenium polypyridyl complexes and their modes of interaction with DNA: Is there a correlation between these interactions and the antitumor activity of the compounds? J. Biol. Inorg. Chem. 2009; 14, 439-48.
C Irace, G Misso, A Capuozzo, M Piccolo, C Riccardi, A Luchini, M Caraglia, L Paduano, D Montesarchio and R Santamaria. Antiproliferative effects of ruthenium-based nucleolipidic nanoaggregates in human models of breast cancer in vitro: Insights into their mode of action. Sci. Rep. 2017; 7, 45236.
M Galletta, F Puntoriero, S Campagna, C Chiorboli, M Quesada, S Goeb and R Ziessel. Absorption spectra, photophysical properties, and redox behavior of ruthenium(II) polypyridine complexes containing accessory dipyrromethene-BF2 chromophores. J. Phys. Chem. A 2006; 110, 4348-58
TS Kamatchi, N Chitrapriya, SLA Kumar, JY Jung, H Puschmann, FR Fronczeke and K Natarajan. The effect of incorporating carboxylic acid functionalities into 2,2՛-bipyridine on the biological activity of the complexes formed: Synthesis, structure, DNA/protein interaction, antioxidant activity and cytotoxicity. RSC Adv. 2017; 7, 16428-43
J Wang and J Yi. Cancer cell killing via ROS: To increase or decrease, that is the question. Canc. Bio Ther. 2008; 7, 1875-84.
MQ Warraicha, A Ghionb, L Perdisatta, LO Neilla, A Caseya and CO Connor. In vitro cytotoxicity, cellular uptake, reactive oxygen species and cell cycle arrest studies of novel ruthenium(II) polypyridyl complexes towards A549 lung cancer cell line. Drug Chem. Toxicol. 2019; 44, 319-29.
V Pierroz, T Joshi, A Leonidova, C Mari, J Schur, I Ott, L Spiccia, S Ferrari and G Gasser. Molecular and cellular characterization of the biological effects of ruthenium(II) complexes incorporating 2 pyridyl-2-pyrimidine-4- carboxylic acid. J. Am. Chem. Soc. 2012; 134, 20376-87.
Z Deng, P Gao, L Yu, B Ma, Y You, L Chan, C Mei and T Chen. Ruthenium complexes with phenylterpyridine derivatives target cell membrane and trigger death receptors-mediated apoptosis in cancer cells. Biomaterials 2017; 129, 111-26.
JP Barolli, RS Corrêa, FS Miranda, JU Ribeiro, J Ellena, V Moreno, MR Cominettig and AA Batista. Polypyridyl ruthenium complexes: Novel DNA-Intercalating agents against human breast tumor. J. Braz. Chem. Soc. 2017; 28, 1879-89.
RK Vuradi, K Dandu, PK Yata, RR Mallepally, N Chintakuntla, R Ch, SS Thakur, CM Rao, S Satyanarayana. Studies on the DNA binding and anticancer activity of Ru(ii) polypyridyl complexes by using a (2-(4-(diethoxymethyl)-1H-imidazo[4,5-f] [1,10] phenanthroline)) intercalative ligand. New J. Chem. 2018; 42, 846-59.
C Zhang, BJ Han, CC Zeng, SH Lai, W Li, B Tang, D Wan, GB Jiang and YJ Liu. Synthesis, characterization, in vitro cytotoxicity and anticancer effects of ruthenium(II) complexes on BEL-7402 cells. J. Inorg. Biochem. 2016; 157, 62-72.
P Xiao, B Zheng, J Sun and J Yang. Biochanin A induces anticancer effects in SK-Mel-28 human malignant melanoma cells via induction of apoptosis, inhibition of cell invasion and modulation of NF-κB and MAPK signaling pathways. Oncol. Lett. 2017; 14, 5989-93.
A Mori, T Suzukia, K Nakajima. Crystal structure of bis (2,2՛-bipyridine) [N՛-(quinolin-2-ylmethylidene) pyridine-2-carbohydrazide]-ruthenium(II)bis (tetra fluoride borate) dichloromethane trisolvate. Acta Crystallogr. E Crystallogr. Comm. 2015; 71, 142-5.
RZ Zarić, MS Pirković and N Hamzagić. Ruthenium(II) complexes as potential apoptosis inducers in cancer therapy. Serbian J. Exp. Clin. Res. 2019. https://doi.org/10.2478/sjecr-2019-0016.
Downloads
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.