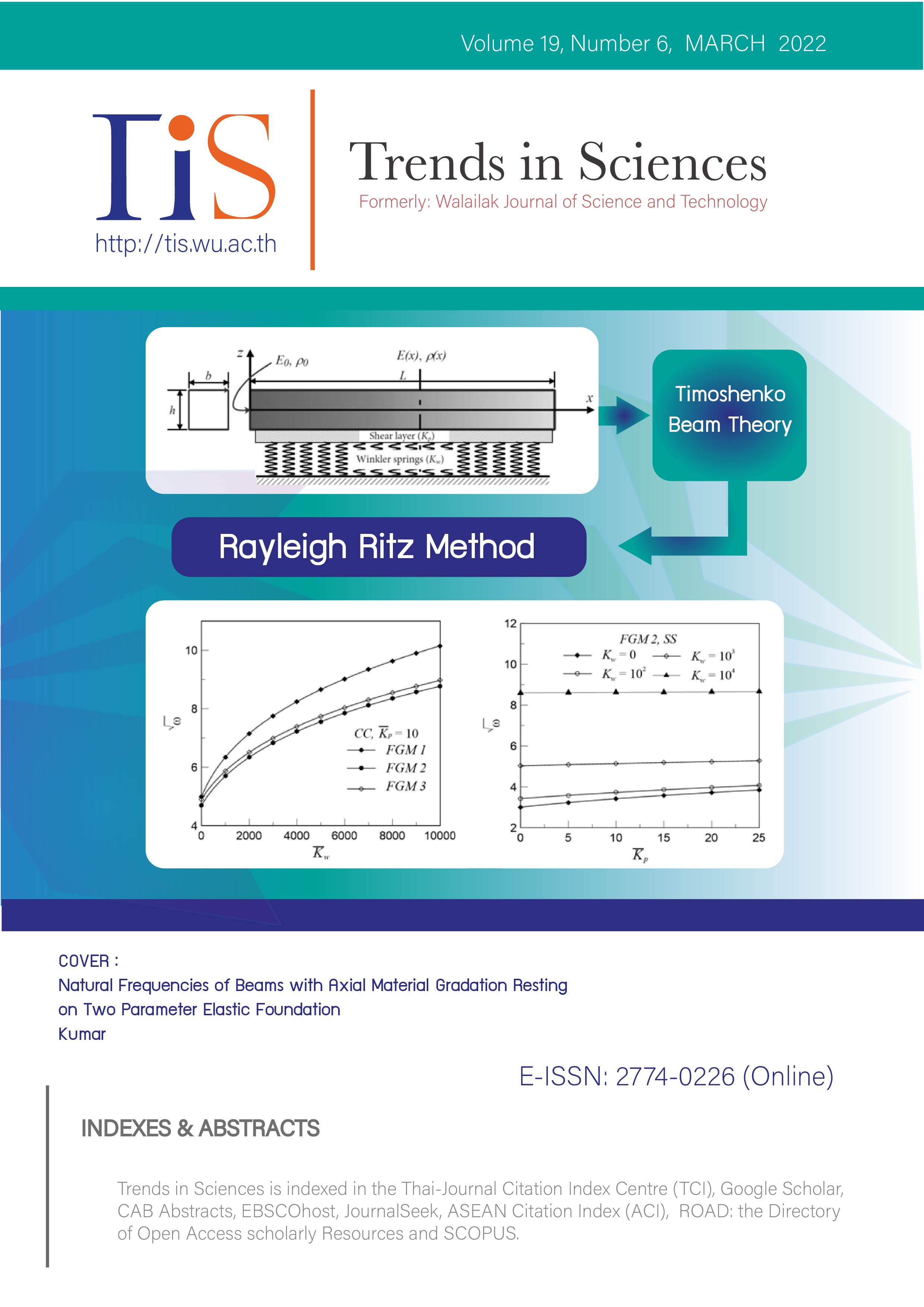
Optimization of Removal of Cadmium (II) using Response Surface Methodology
DOI:
https://doi.org/10.48048/tis.2022.2997Keywords:
Cd (II), Calotropis procera, Adsorption, RSMAbstract
A huge quantity of wastewater is generated and dumped directly into the natural environment. Industrial wastewater recycling is common in India and is mainly used for agriculture in the surrounding areas. This has led to high levels of cadmium and other heavy metals in the air, water, and soil. Natural adsorbents provide an effective solution for wastewater treatment. Wooden objects can be used to remove heavy metals from contaminated water. The current investigation attempts to remove cadmium from the cadmium-containing aqueous solution using the root of Calotropis Procera. The adsorption process was developed with batch studies to quantify the removal of cadmium (Y) using 3 flexible process variables namely, pH, residence time (CT), and adsorbent dose (AD). Interaction between variables, a 20 (23) central composite design (CCD) was developed using the response surface method (RSM). The results of the adsorption verification tests were performed and show excellent compliance with the model prediction. The value of F (550.78) and the return coefficient (R2) of 0.9796 proved the validity of the improved quadratic model.
HIGHLIGHTS
- Cadmium (Cd-II) is a neuro-toxic substance, which gets easily introduced, assimilated into the food chain/natural habitat, and is responsible for severe health problems
- The agricultural waste and biomass i.e. roots of Calotropis Procera was used to treat heavy metals and Cadmium (II) from the wastewater
- The advanced RSM and Central Composite Design (CCD) have been employed for the design of experiments
- DOE is employed with three process parameters viz., pH, residence time and adsorbent dosage to optimise the process
- The results obtained from this investigation indicate that Calotropis Procera is a promising natural adsorbent for the treatment of effluents with Cd (II) ions
GRAPHICAL ABSTRACT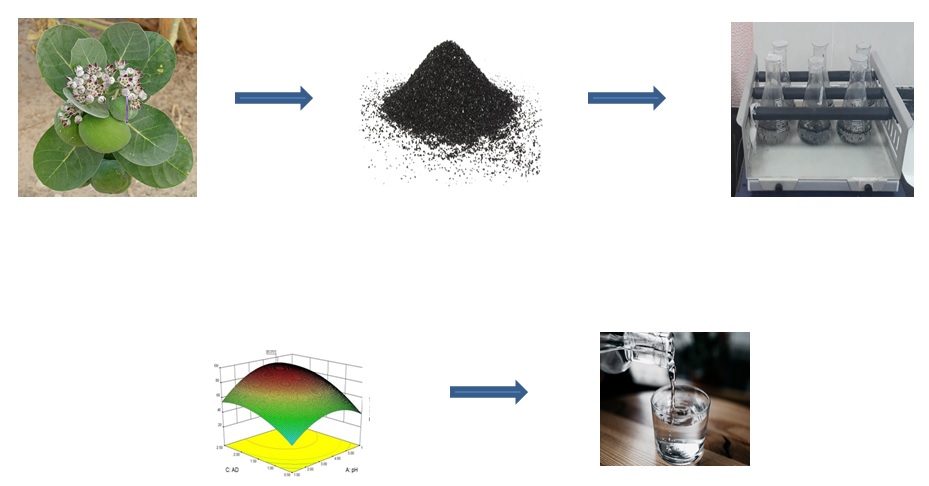
Downloads
References
FA Olabemiwo, BS Tawabini, F Patel, TA Oyehan, M Khaled and T Laoui. Cadmium removal from contaminated water using polyelectrolyte-coated industrial waste fly ash. Bioinorg. Chem. Appl. 2017; 2017, 1-13.
R Singh, R Chadetrik, R Kumar, K Bishnoi, D Bhatia, A Kumar, NR Bishnoi and N Singh. Biosorption optimization of lead (II), cadmium (II) and copper (II) using response surface methodology and applicability in isotherms and thermodynamics modeling. J. Hazard. Mater. 2010; 174, 623-34.
M Iqbal, N Iqbal, IA Bhatti, N Ahmad and M Zahid. Response surface methodology application in optimization of cadmium adsorption by shoe waste: A good option of waste mitigation by waste. Ecol. Eng. 2016; 88, 265-75.
BIS. Indian standard drinking water specification. Bureau of Indian Standards, New Delhi. 2012, p. 2-3.
JT Li, JW Qiu, XW Wang, Y Zhong, CY Lan and WS Shu. Cadmium contamination in orchard soils and fruit trees and its potential health risk in Guangzhou, China. Environ. Pollut. 2016; 143, 159-65.
TG Ammari, I Al-Labadi, A Tahboub and A Ghrair. Assessment of unmodified wetland bio-waste: Shoots of Cyperus laevigatus, for cadmium adsorption from aqueous solutions. Process Saf. Environ. Prot. 2015; 95, 77-85.
M Jain, VK Garg, R Paliwal, K Kadirvelu and S Chaudhry. Optimization of cadmium (II) removal from water using sunflower waste carbon-a statistical approach. Toxin Rev. 2010; 40, 1-10.
UK red list substances, Available at: https://www.legislation.gov.uk/uksi/1991/472/contents/made, accessed June 2020.
SN Hosamane. Removal of arsenic by phytoremediation-a study of two plant spices. Int. J. Eng. Sci. Technol. 2012; 1, 218-24.
L Friberg. Cadmium and the kidney. Environ. Health Perspect. 1984; 54, 1-11.
KD Eum, MS Lee and D Paek. Cadmium in blood and hypertension. Sci. Total Environ. 2008; 407, 147-53.
T Matsukura, C Inaba, EA Weygant, D Kitamura, R Janknecht, H Matsumoto, DP Hyink, S Kashiwada and T Obara. Extracellular vesicles from human bone marrow mesenchymal stem cells repair organ damage caused by cadmium poisoning in a medaka model. Physiol. Rep. 2019; 7, e14172.
RC Patra, AK Rautray and D Swarup. Oxidative stress in lead and cadmium toxicity and its amelioration. Vet. Med. Int. 2011; 2011, 457327.
Drinking Water Contaminants- Cadmium, Available at: https://www.freedrinkingwater.com/water-contamination/cadmium-contaminants-removal-water.htm, accessed June 2020.
HW Chen, MM Frey, D Clifford, LS McNeill and M Edwards. Arsenic treatment considerations. J. Am. Water Work Assoc. 1999; 91, 74-85.
C Duran, D Ozdes, HT Akcay, H Serencam and M Tufekci. Co (II), and Ni (II) ions in environmental samples by carrier element free co-precipitation method prior to their flame atomic absorption spectrometric determination. Desalination Water Treat. 2015; 53, 390-7.
S Ahmed, S Chughtai and MA Keane. The removal of cadmium and lead from aqueous solution by ion exchange with NaY zeolite. Sep. Purif. Technol. 1998; 13, 57-64.
KS Rao, M Mohapatra, S Anand and P Venkateswarlu. Review on cadmium removal from aqueous solutions. Int. J. Eng. Sci. Technol. 2011; 2, 81-103.
SA Nosier. Removal of cadmium ions from industrial wastewater by cementation. Chem. Biochem. Eng. Q. 2003; 17, 219-24.
KS Rao, M Mohapatra, S Anand and P Venkateswarlu. Review on cadmium removal from aqueous solutions. Int. J. Eng. Sci. Technol. 2010; 2, 81-103.
M Raad, M Srour, A Hijazi, R Hmedeh, Z Damaj, M Hamieh, AA Lami, M Kassir and WE Khatib. Removal of cadmium (II) Ions from waste water by adsorption onto the powder of lebanese anacyclus nigllifolius boiss: A comparative study. Am. J. Phytomed. Clin. Ther. 2017; 5, 11.
VK Gupta, CK Jain, I Ali, M Sharma and VK Saini. Removal of cadmium and nickel from wastewater using bagasse fly ash-a sugar industry waste. Water Res. 2003; 37, 4038-44.
Y Lin, X He, G Han, Q Tian and W Hu. Removal of crystal violet from aqueous solution using powdered mycelial biomass of Ceriporia lacerata P2. Res. J. Environ. Sci. 2011; 23, 2055-62.
D Garai and V Kumar. A Box-Behnken design approach for the production of xylanase by Aspergillus candidus under solid state fermentation and its application in saccharification of agro residues and Parthenium hysterophorus L. Ind. Crop. Prod. 2013; 44, 352-63.
M Khajeh. Application of Box-Behnken design in the optimization of a magnetic nanoparticle procedure for zinc determination in analytical samples by inductively coupled plasma optical emission spectrometry. J. Hazard. Mater. 2009; 172,385-9.
SN Hosamane, K Ghurlhosur and S Bagali. Optimization of lactic acid production from cane sugar molasses. Research Res. J. Pharm. Biol. Chem. Sci. 2008; 4, 872-7.
M Iqbal, N Iqbal, IA Bhatti, N Ahmad and M Zahid. Response surface methodology application in optimization of cadmium adsorption by shoe waste: A good option of waste mitigation by waste. Ecol. Eng. 2016, 8, 265-75.
RH Myers, D Montgomery and CM Anderson-Cook. Response surface methodology: Process and product optimization using designed experiments. John Wiley & Sons, Inc., New York, 2016.
S Chatterjee, A Kumar, S Basu and S Dutta. Application of response surface methodology for methylene blue dye removal from aqueous solution using low-cost adsorbent. Chem. Eng. J. 2012; 181, 289-99.
S Sen, S Nandi and S Dutta. Application of RSM and ANN for optimization and modeling of biosorption of chromium (VI) using cyanobacterial biomass. Appl. Water Sci. 2018; 8, 148.
M Amini, H Younesi, N Bahramifar, AAZ Lorestani, F Ghorbani, A Daneshi and M Sharifzadeh. Application of response surface methodology for optimization of lead biosorption in an aqueous solution by Aspergillus niger. J. Hazard. Mater. 2008; 154, 694-702.
SN Hosamane and GP Desai. Air pollution modelling from meteorological parameters using artificial neural network. In: DJ Hemanth and S Smys (Eds.). computational vision and bio inspired computing. Springer, Cham, 2018, p. 466-75.
W Yong-Fei and W Cheng-Guo. The application of response surface methodology. J. Cent. Univ. For National. (Nat. Sci. Ed.). 2005; 2005, 3.
PK Pandey, Y Verma, S Choubey, M Pandey and K Chandrasekhar. Biosorptive removal of cadmium from contaminated groundwater and industrial effluents. Bioresour. Technol. 2008; 99, 4420-47.
M Jiang, X Jin, X Lu and Z Chen. Adsorption of Pb (II), Cd (II), Ni (II) and Cu (II) onto natural kaolinite clay. Desalination 2010; 252, 33-9.
Downloads
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.