Complex Optical Investigation of Sodium Superoxide Loaded Phosphovanadate Glass System in Ultra-Violet and Visible Region
DOI:
https://doi.org/10.48048/tis.2022.2077Keywords:
Optical properties, Sodium superoxide, Phosphovanadate glass, Optical conductivity, Urbach energy, Direct band gap, Dielectric constantAbstract
Sodium superoxide loaded phosphovanadate based glass systems were prepared from a mixture of vanadium pentoxide (V2O5), phosphorus pentoxide (P2O5) boric acid (H3BO3) and sodium superoxide (NaO2) using a melt-quenching method. Amorphous phase of as-prepared glass system confirmed using XRD technique. Surface morphology of glass system studied using scanning electron microscope. Ultraviolet-visible spectroscopy was employed to extract complex optical parameters like direct and indirect optical band gap, Urbach energy, refractive index, complex dielectric constant and optical conductivity. The absorption bands in the region 200 - 400 nm are recognized to the ligand-to-metal charge transfer. The region of 400 - 600 nm is ascribed to the pair excitation processes. The refractive index increases initially and then decreases for 15 and 20 mol % of NaO2 due non bridging oxygen (NBO) atoms. The 25 mol % of NaO2 sample shows maximum value of extinction coefficient and refractive index. The direct and indirect band gap energies vary in between 2.067 to 1.824 eV and 1.869 to 1.495 eV, respectively. With increase in concentration of NaO2, the effective band gap of NaO2 decreases because band edge shifted into forbidden gap due to increase in defect levels below the conduction. This primary report on sodium superoxide loaded phosphovanadate based glass systems opens wide avenue for battery and supercapacitor applications. Tailing in the bandgap was observed and found to obey Urbach rule.
HIGHLIGHTS
- Sodium superoxide-phosphovanadate glass system prepared by melt-quenching method
- Complex optical properties of prepared glass system studied successfully
- Glass system have characteristics of trapping of light
- Also, glass system has ability to slow down light
GRAPHICAL ABSTRACT 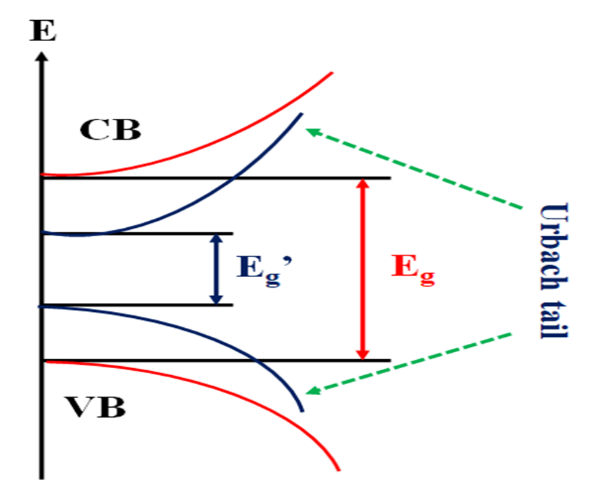
Downloads
References
GI Petrov, VV Yakovlev and J Squier. Raman microscopy analysis of phase transformation mechanisms in vanadium dioxide. Appl. Phys. Lett. 2002; 81, 1023-5.
MM El-Desoky and MS Al-Assiri. Structural and polaronic transport properties of semiconducting CuO–V2O5–TeO glasses. Mater. Sci. Eng. B 2007; 137, 237-46.
GM Clark and AN Pick. DTA study of the reactions of V2O5 with metal (II) oxides. J. Therm. Anal. 1975; 7, 289-300.
H Liu and D Tang. Synthesis of ZnV2O6 powder and its cathodic performance for lithium secondary battery. Mater. Chem. Phys. 2009; 114, 656-9.
E Mansour, YM Moustafa, GM El-Damrawi, SA El-Maksoud and H Doweidar. Memory switching of Fe2O3–BaO–V2O5 glasses. Physica B 2001; 305, 242-9.
K Jeyalakshmi, S Vijayakumar, S Nagamuthu and G Muralidharan. Effect of annealing temperature on the supercapacitor behaviour of β-V2O5 thin films, Mater. Res. Bull. 2013; 48, 760-6.
L Murawski. Electrical conductivity in iron-containing oxide glasses. J. Mater. Sci. 1982; 17, 2155-63.
NF Mott. Conduction in glasses containing transition metal ions. J. Non-Cryst. Solids 1968; 1, 1-17.
MA Hassan, FM Ebrahim, MG Moustafa, ZMA El-Fattah and MM El-Okr. Unraveling the hidden Urbach edge and Cr6+ optical transitions in borate glasses. J. Non Cryst. Solids 2019; 515, 157.
AI Ismail, A Samir, F Ahmad, LI Soliman and A Abdelghany. Spectroscopic studies and the effect of radiation of alkali borate glasses containing chromium ions. J. Non Cryst. Solids 2021; 565, 120743.
S Yusub and D Krishna Rao. The role of chromium ions on dielectric and spectroscopic properties of Li2O–PbO–B2O3–P2O5 glasses. J. Non-Cryst. Solids 2014; 398-399, 1-9.
RK Brow and DR Tallant. Structural design of sealing glasses, J. Non-Cryst. Solids 1997; 222, 396-406.
TO Hardwell. Solid-state lasers: Properties and applications. Nova Science Pub Inc, New York, 2008, p. 227.
B Denker, B Galagan, V Osiko, S Sverchkov and E Dianov. Luminescent properties of Bi-doped boro-alumino-phosphate glasses. Appl. Phys. B 2007; 87, 135-7.
O Mao, RL Turner, IA Courtney, BD Fredericksen, MI Buckett, LJ Krause and JR Dahn. Active/inactive nanocomposites as anodes for Li-Ion batteries. Electrochem. Solid-State Lett. 1999; 2, 3-5.
CR Kesavulu, RPS Chakradhar, RS Muralidhara, JL Rao and RV Anavekar. EPR, optical absorption and photoluminescence properties of Cr3+ ions in lithium borophosphate glasses. J. Alloys Compd. 2010; 496, 75-80.
JT Tsai, CY Huang and ST Lin. The development of conductive pastes for solar cells. Adv. Mater. Res. 2012; 557- 559, 1201-4.
JW Yu and K Oh. New in-line fiber band pass filters using high silica dispersive optical fibers. Opt. Commu. 2002; 204, 111–8.
F Li, Z Wei, A Manthiram, Y Feng, J Maa and L Maid. Sodium-based batteries: From critical materials to battery systems. J. Mater. Chem. A 2019; 7, 9406-31.
X Xu, KS Hui, DA Dinh, KN Hui and H Wang. Recent advances in hybrid sodium-air batteries, Mater. Horiz. 2019; 6, 1306-35.
X Ren and Y Wu. A low-overpotential potassium-oxygen battery based on potassium superoxide. J. Am. Chem. Soc. 2013; 135, 2923-6.
P Hartmann, CL Bender, M Vracar, A Garsuch, AK Durr, J Janek and P Adelhelm. A rechargeable room-temperature sodium superoxide (NaO2) battery. Nat. Mater. 2012; 12, 228-32.
KR Nemade and SA Waghuley. Novel synthesis approach for stable sodium superoxide (NaO2) nanoparticles for LPG sensing application. Int. Nano Lett. 2017; 7, 233-6.
M He, KC Lau, X Ren, N Xiao, WD McCulloch, LA Curtiss and Y Wu. Concentrated electrolyte for the sodium-oxygen battery: Solvation structure and improved cycle life. Angew. Chem. Int. Ed. 2016; 55, 1-6.
E Peled, D Golodnitsky, H Mazor, M Goor and S Avshalomov. Parameter analysis of a practical lithium- and sodium-air electric vehicle battery. J. Powe. Sour. 2011; 196, 6835-40.
H Park, J Kim, MH Lee, SK Park, D Kim, Y Bae, Y Ko, B Lee and K Kang. Highly durable and stable sodium superoxide in concentrated electrolytes for sodium-oxygen batteries. Adv. Energy Mater. 2018; 8, 1801760.
MS Sadeq and MA Abdo. Effect of iron oxide on the structural and optical properties of alumino-borate glasses. Ceram. Int. 2021; 47, 2043-9.
S Thakur, A Kaur and L Singh. Mixed valence effect of Se6+ and Zr4+ on structural, thermal, physical, and optical properties of B2O3–Bi2O3–SeO2–ZrO2 glasses. Opt. Mat. 2019; 96, 109338.
RV Barde. Preparation, characterization and CO2 gas sensitivity of polyaniline doped with sodium superoxide. Mater. Res. Bull. 2016; 73, 70-6.
RV Barde and SA Waghuley. Preparation and electrical conductivity of novel vanadate borate glass system containing graphene oxide. J. Non-Cryst. Solids 2013; 376, 117-25.
RV Barde and SA Waghuley. Transport properties of rare earth CeO2 doped phospho-vanadate glass systems. J. Chin. Adv. Mater. Soc. 2014; 2, 273-83.
M Fox. Optical properties of solids. Oxford University Press, New York, 2001, p. 274.
JB Lian, XC Duan, JM Ma, P Peng, T Kim and WJ Zheng. Hematite (alpha-Fe2O3) with various morphologies: Ionic liquid-assisted synthesis, formation mechanism, and properties. ACS Nano 2009; 3, 3749-61.
T Wang, S Zhou, C Zhang, J Lian, Y Liang and W Yuan. Facile synthesis of hematite nanoparticles and nanocubes and their shape-dependent optical properties. J. Chem. 2014; 38, 46-9.
S Gedia, V Reddy, M Reddya, C Parkb, JC Wookb and KTR Reddy. Comprehensive optical studies on SnS layers synthesized by chemical bath deposition. Opti. Mate. 2015; 42, 468-75.
RV Barde. Influence of CeO2 content on complex optical parameters of phosphovanadate glass system, Spectroch Spectrochim. Acta A Mol. Biomol. Spectrosc. 2016; 153, 160-4.
RV Barde, KR Nemade and SA Waghuley. Complex optical study of V2O5–P2O5–B2O3–GO glass systems by ultraviolet-visible spectroscopy. Opti. Mate. 2015; 40, 118-21.
KL Chopra and I Kaur. Thin film phenomena. McGraw-Hill, New York, 1969, p. 736.
S Song, Y Wang, X Yuan, W Yao, W Jing. Characterization and preparation of Sn-doped CuGaS2 thin films by paste coating. Mater. Lett. 2015; 148, 41-4.
S Shailajha, K Geetha, P Vasantharani and SPSA Kadhar. Spectrochi. Acta Part A: Mole. and Biomol. Spectro. 2015; 138, 846-56.
K Boubaker. A physical explanation to the controversial Urbach tailing universality. Eur. Phys. J. Plus 2011; 126, 10.
B Choudhur, B Borah and A Choudhury. Extending photocatalytic activity of TiO2 nanoparticles to visible region of illumination by doping of cerium. Photochem. Photobiol. 2012; 88, 257-64.
B Choudhury, M Dey and A Choudhury. Defect generation, d-d transition, and band gap reduction in Cu-doped TiO2 nanoparticles. Int. Nano Lett, 2013; 3, 1-8.
CO Ayieko1, RJ Musembi, SM Waita, BO Aduda and PK Jain. Structural and optical characterization of nitrogen-doped TiO2 thin films deposited by spray pyrolysis on fluorine doped tin oxide (FTO) coated glass slides. Int. J. Ene. Eng. 2012; 2, 67-72.
AQ Abdullah. Surface and volume energy loss, optical conductivity of rhodamine 6G dye (R6G), Chem. and Mat. Res. 2013; 3, 56-63.
RV Barde, KR Nemade and SA Waghuley. Complex optical study of V2O5–P2O5–B2O3–Dy2O glass systems. J. Taib. Uni. Sci. 2015; 10, 340-4.
P Sharma and SC Katyal. Determination of optical parameters of a-(As2Se3)90Ge10 thin film. J. Phys. D: Appl. Phys. 2007; 40, 2115-20.
NA Bakr, AM Funde, VS Waman, MM Kamble, RR Hawaldar, DP Amalnerkar and SR Jadkar, Determination of the optical parameters of a-Si: H thin films deposited by hot wire-chemical vapour deposition technique using transmission spectrum only. Pramana J. Phys. 2011; 76, 519-31.
NF Mott and EA Davis. Conduction in non-crystalline systems V conductivity, optical absorption and photoconductivity in amorphous semiconductors. Philos. Mag. 1970; 22, 0903-22.
F Yakuphanoglu, A Cukurovali and I Yilmaz. Refractive index and optical absorption properties of the complexes of a cyclobutane containing thiazolylhydrazone ligand. Opt. Mater. 2005; 27, 1363-8.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2022 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.