Studies on Preparation and Evaluation of Soluble 1:1 Stoichiometric Curcumin Complex for Colorectal Cancer Treatment
DOI:
https://doi.org/10.48048/tis.2021.1403Keywords:
Curcumin, Colorectal cancer, MTT assay, Hoechst staining, Functionalized dyeing testAbstract
This study investigates the complex of curcumin (CMN), which has enhanced solubility and hence, higher cytotoxicity compared to free CMN. Insilco molecular modelling and phase solubility (PS) studies were performed with the drug and carriers for interaction. The complex was characterized by in vitro drug release, FT-IR, PXRD, TGA, DSC, SEM, DLS, and functionalized dyeing test. The result showed that the CMN-PEG6000 complex produced significant properties of solubility (≈ 190 folds) and dissolution (80.68 % at 30 min), with stability constants equivalent to 309 and 377 M-1 at 25 and 37 °C, respectively. It exhibited AL type of isotherm indicating 1:1 stoichiometry. The result from the in vitro cytotoxicity showed that 50 % inhibition (IC50) was achieved on the SW480 and Caco-2 cells at an amount of complex that was considerably lesser than free CMN. Apoptosis study showed that the cells underwent cell death mainly by apoptosis with a small number by necrosis.
HIGHLIGHTS
- Enhanced curcumin solubility up to 190 fold higher than pure curcumin was investigated
- The phase solubility results of curcumin range from 5.7×10-4 M-1 and 7.8×10-4 M-1 at 25 and 37 °C, respectively
- First time of novel dyeing test was performed with complex of curcumin, signified its solubility
- This study provides useful approach for obtaining curcumin products with maximum aqueous solubility
GRAPHICAL ABSTRACT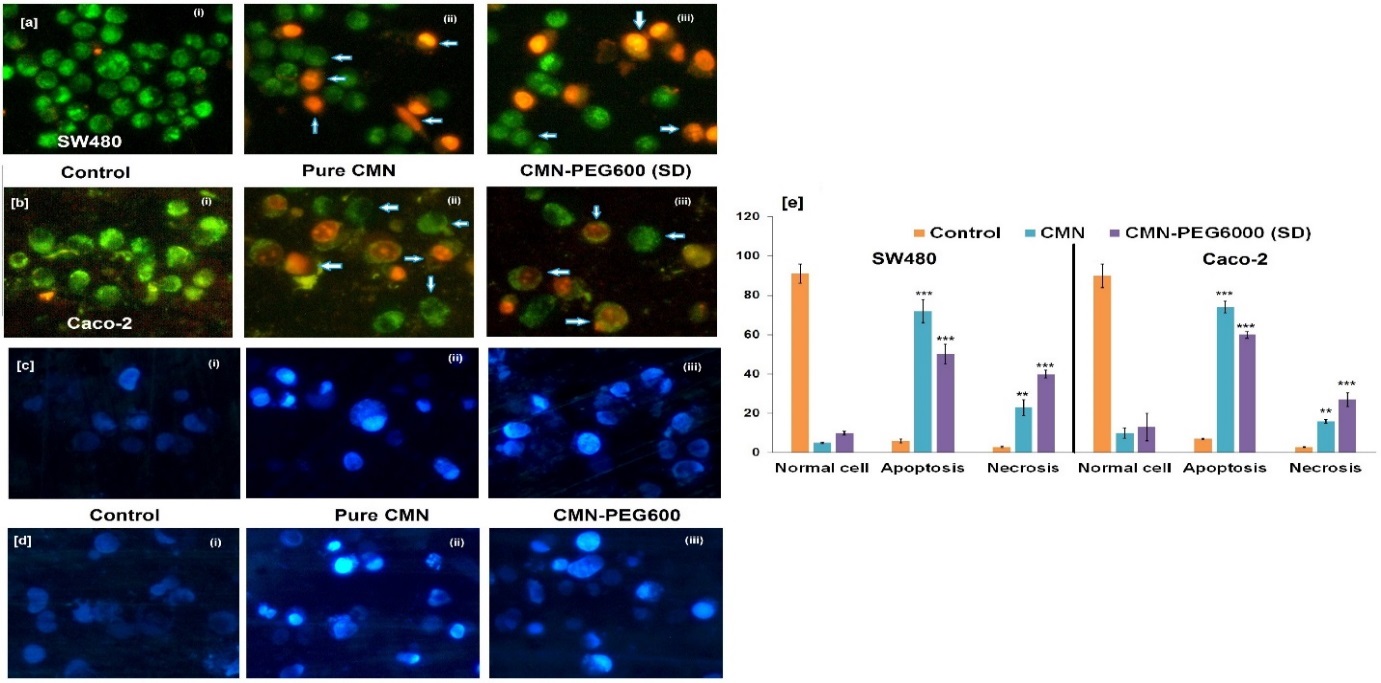
Downloads
References
NC Hall and AT Ruutiainen. Colorectal cancer: Imaging conundrums. Surg. Oncol. Clin. N Am. 2018; 27, 289-302.
A Farinetti, V Zurlo, A Manenti, F Coppi and AV Mattioli. Mediterranean diet and colorectal cancer: A systematic review. Nutrition 2017; 43-44, 83-8.
MK John, H Xie, EC Bell and D Liang. Development and pharmacokinetic evaluation of curcumin co-solvent formulation. Anticancer Res. 2013; 33, 4285-91.
SS Bansal, M Goel, F Aqil, MV Vadhanam and RC Gupta. Advanced drug delivery systems of curcumin for cancer chemoprevention. Cancer Prev. Res (Phila) 2011; 4, 1158-71.
T Higuchi and KA Connors. Phase solubility techniques. Adv. Anal. Chem. Instrum. 1965; 4, 117-212.
C Nicolescu, CC Arama, A Nedelcu and CM Monciu. Phase solubility studies of the inclusion complexes of repaglinide with ß-cyclodextrin and ß-cyclodextrin derivatives. Farmacia 2010; 58, 620-8.
MMJ Moideen, A Alqahtani, K Venkatesan, F Ahmad, K Kalpana, M Gayasuddin, RA Shaik, KMM Ibraheem, MEDM Salama and SY Abed. Application of the Box-Behnken design for the production of soluble curcumin: Skimmed milk powder inclusion complex for improving the treatment of colorectal cancer. Food Sci. Nutr. 2020; 8, 6643-59.
A Almeida, S Possemiers, MN Boone, TD Beer, T Quinten, LV Hoorebeke, JP Remon and C Vervaet. Ethylene vinyl acetate as a matrix for oral sustained release dosage forms produced via hot-melt extrusion. Eur. J. Pharm. Biopharm. 2011; 77, 297-305.
MJ Muthu, K Kavitha, KS Chitra and S Nanthineeswari. Soluble curcumin prepared using four different carriers by solid dispersions: Phase solubility, molecular modelling and physicochemical characterization. Trop. J. Pharm. Res. 2019; 18, 1581-8.
MMJM Moideen, K Karuppaiyan, R Kandhasamy and S Seetharaman. Skimmed milk powder and pectin decorated solid lipid nanoparticle containing soluble curcumin used for the treatment of colorectal cancer. J. Food Process Eng. 2019; 43, e13246.
NS Kumar, M Ganapathy, S Sharmila, M Shankar, M Vimalan and IV Potheher. ZnO/Ni(OH)2 core-shell nanoparticles: Synthesis, optical, electrical and photoacoustic property analysis. J. Alloys Compd. 2017; 703, 624-32.
L Liu, J Xu, H Zheng, K Li, W Zhang, K Li and H Zhang. Inclusion complexes of laccaic acid A with ß-cyclodextrin or its derivatives: Phase solubility, solubilization, inclusion mode, and characterization. Dyes Pigm. 2017; 139, 737-46.
JM Mohamed, A Alqahtani, F Ahmad, V Krishnaraju and K Kalpana. Pectin co-functionalized dual layered solid lipid nanoparticle made by soluble curcumin for the targeted potential treatment of colorectal cancer. Carbohydr. Polym. 2021; 252, 117180.
S Karthik, R Sankar, K Varunkumar and V Ravikumar. Romidepsin induces cell cycle arrest, apoptosis, histone hyperacetylation and reduces matrix metalloproteinases 2 and 9 expression in bortezomib sensitized non-small cell lung cancer cells. Biomed. Pharmacother. 2014; 68, 327-34.
R Iacovino, JV Caso, F Rapuano, A Russo, M Isidori, M Lavorgna, G Malgieri and C Isernia. Physicochemical characterization and cytotoxic activity evaluation of hydroxymethyl ferrocene: β-cyclodextrin inclusion complex. Molecules 2012; 17, 6056-70.
M El-Badry. Physicochemical characterization and dissolution properties of meloxicam-gelucire 50/13 binary systems. Sci. Pharm. 2011; 79, 375-86.
W Ali, AC Williams and CF Rawlinson. Stochiometrically governed molecular interactions in drug: Poloxamer solid dispersions. Int. J. Pharm. 2010; 391, 162-8.
AB Gangurde, HS Kundaikar, SD Javeer, DR Jaiswar, MS Degani and PD Amin. Enhanced solubility and dissolution of curcumin by a hydrophilic polymer solid dispersion and its In-silico molecular modeling studies. J. Drug Deliv. Sci. Technol. 2015; 29, 226-37.
CS Mangolim, C Moriwaki, AC Nogueira, F Sato, ML Baesso, AM Neto and G Matioli. Curcumin-β-cyclodextrin inclusion complex: Stability, solubility, characterisation by FT-IR, FT-Raman, X-ray diffraction and photoacoustic spectroscopy, and food application. Food Chem. 2014; 153, 361-70.
M Palanisamy and J Khanam. Solid dispersion of prednisolone: Solid state characterization and improvement of dissolution profile. Drug Dev. Ind. Pharm. 2011; 37, 373-86.
F Sadeghi, M Ashofteh, A Homayouni, M Abbaspour, A Nokhodchi and HA Garekani. Antisolvent precipitation technique: A very promising approach to crystallize curcumin in presence of polyvinyl pyrrolidon for solubility and dissolution enhancement. Colloids Surf. B Biointerfaces 2016; 147, 258-64.
J Li, I Lee, GH Shin, X Chen and HJ Park. Curcumin-Eudragit® E PO solid dispersion: A simple and potent method to solve the problems of curcumin. Eur. J. Pharm. Biopharm. 2015; 94, 322-32.
UMD Lekshmi, G Poovi, N Kishore and PN Reddy. In vitro characterization and in vivo toxicity study of repaglinide loaded poly (methyl methacrylate) nanoparticles. Int. J. Pharm. 2010; 396, 194-203.
F Kurniawansyah, L Quachie, R Mammucari and NR Foster. Improving the dissolution properties of curcumin using dense gas antisolvent technology. Int. J. Pharm. 2017; 521, 239-48.
SJ George and DT Vasudevan. Characterization, and solubility of 2-HP-ß-Cyclodextrin-Meclizine HCl inclusion complexes. J. Young Pharm. 2012; 4, 220-7.
AA Thorat and SV Dalvi. Solid-state phase transformations and storage stability of curcumin polymorphs. Cryst. Growth Des. 2015; 15, 1757-70.
M Khairuddin, E Pramono, SB Utomo, V Wulandari, WA Zahrotul and F Clegg. The effect of polyethylene glycol Mw 400 and 600 on stability of Shellac Waxfree. J. Phys. Conf. Ser. 2016; 776, 1757-8981.
A Popat, S Karmakar, S Jambhrunkar, C Xu and C Yu. Curcumin-cyclodextrin encapsulated chitosan nanoconjugates with enhanced solubility and cell cytotoxicity. Colloids Surf. B Biointerfaces 2014; 117, 520-7.
CS Kumar, A Mahesh, MG Antoniraj, S Vaidevi and K Ruckmani. Ultrafast synthesis of stabilized gold nanoparticles using aqueous fruit extract of Limonia acidissima L and conjugated epirubicin: Targeted drug delivery for treatment of breast cancer. RSC Adv. 2016; 32, 26874-82.
S Xu, J Chen, B Wang and Y Yang. Sustainable and hydrolysis-free dyeing process for polylactic acid using nonaqueous medium. ACS Sustain. Chem. Eng. 2015; 3, 1039-46.
S Manju and K Sreenivasan. Gold nanoparticles generated and stabilized by water soluble curcumin-polymer conjugate: Blood compatibility evaluation and targeted drug delivery onto cancer cells. J. Coll. Inter. Sci. 2012; 368, 144-51.
SDA Abel and SK Baird. Honey is cytotoxic towards prostate cancer cells but interacts with the MTT reagent: Considerations for the choice of cell viability assay. Food Chem. 2018; 241, 70-8.
R Dhivya, P Jaividhya, A Riyasdeen, M Palaniandavar, G Mathan and MA Akbarsha. In vitro antiproliferative and apoptosis-inducing properties of a mononuclear copper(II) complex with dppz ligand, in two genotypically different breast cancer cell lines. Biometals 2015; 28, 929-43.
G Vignesh, R Senthilkumar, P Paul, VS Periasamy, MA Akbarsha and S Arunachalam. Protein binding and biological evaluation of a polymer-anchored cobalt(III) complex containing a 2,2⁄-bipyridine ligand. RSC Adv. 2014; 4, 57483-92.
Downloads
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.







