Review on Coumarins from the Genus Calophyllum: Molecular Interactions Against HIV Targets and Insights from Computational and Experimental Studies
DOI:
https://doi.org/10.48048/tis.2026.13647Keywords:
Calophyllaceae, Calophyllum, Coumarin, Molecular docking, ADMET, Anti-HIV, CalanolidesAbstract
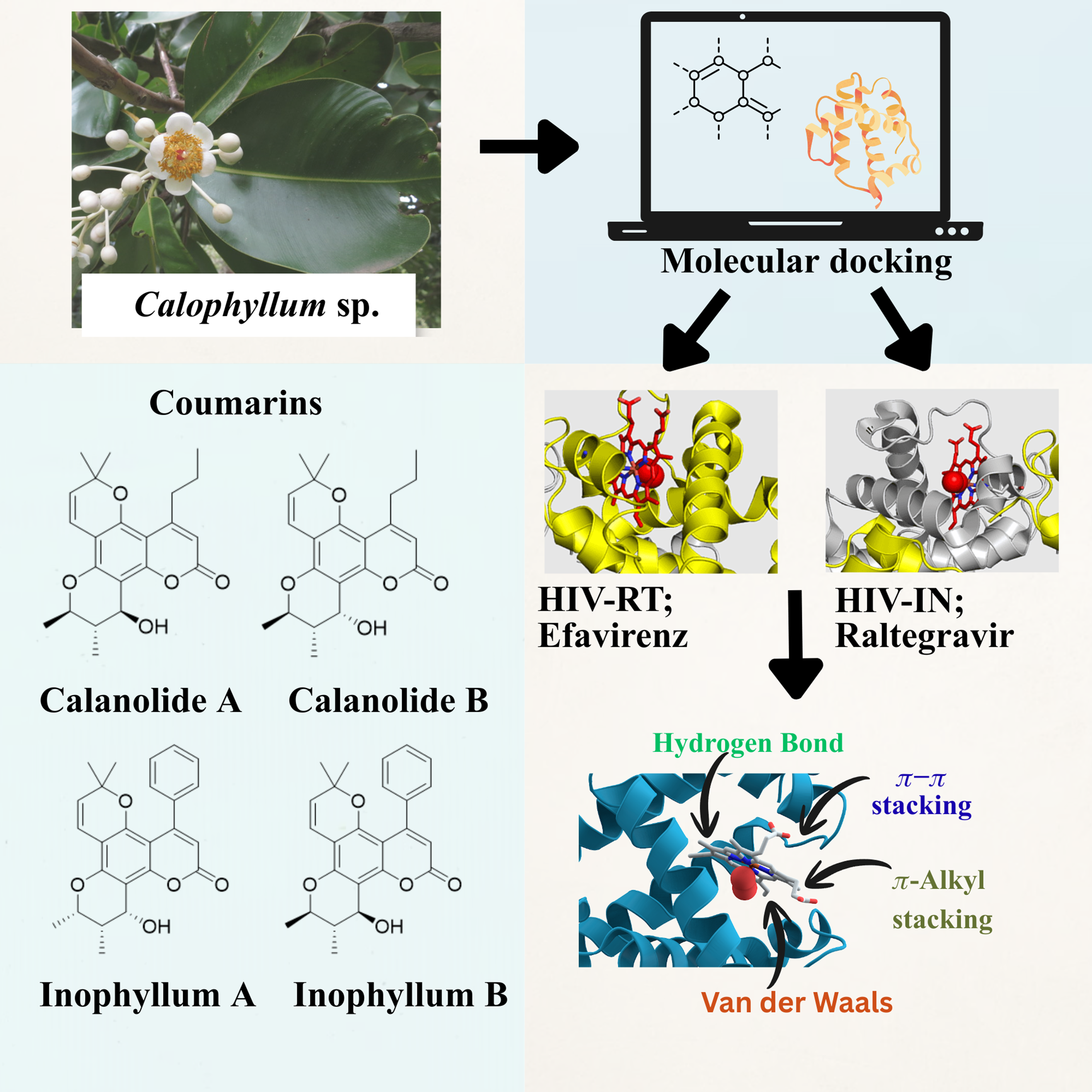
The Calophyllum genus is a diverse group of tropical trees known for producing a wide variety of bioactive compounds, including xanthones, coumarins, triterpenoids, and flavonoids. Coumarins possess several pharmacological activities such as anti-HIV, anti-inflammatory, anti-bacterial, and anti-coagulation effects. Calanolide A, Calanolide B, and several Inophyllum derivatives are among the compounds that exhibit a significant inhibition of HIV-1 reverse transcriptase (RT) that is beyond the action of some HIV-1 RT inhibitors in market drugs. This research conducted the docking of HIV-1 RT and integrase (IN) to assess its antiviral coumarins derived from Calophyllum species. Coumarins are benzopyrones characterized by a conjugated aromatic system that is crucial for anchoring at the RT- NNRTI binding pocket and the IN catalytic core. Hydroxyl, methoxy, and acetoxy groups increase their binding affinity for enzymes and specificity. The molecular docking scores support these findings, where Inophyllum E, Soulattrolone, and other compounds show significantly better binding with RT and IN than the controls Efavirenz and Raltegravir. The impressive effectiveness can be attributed to their structural variety, hydrophobic interactions, and optimally placed functional groups, which allow for considerable and stable complexing within the active sites. Consequently, the Calophyllum coumarins are deemed valuable for the future development of novel antiviral drugs.
HIGHLIGHTS
- Calophyllum species are rich sources of bioactive coumarins with notable pharmacological potential, particularly antiviral properties against HIV targets.
- Molecular docking demonstrated strong interactions of Calophyllum-derived coumarins with HIV-1 reverse transcriptase and integrase active sites.
- Key compounds such as Inophyllum E and Soulattrolide exhibited higher binding affinity than standard drugs Efavirenz and Raltegravir.
- Structural features including hydroxyl, methoxy, and acetoxy substituents enhance enzyme binding and stability within catalytic pockets.
- Calophyllum coumarins represent promising lead molecules for the development of next-generation anti-HIV therapeutics.
GRAPHICAL ABSTRACT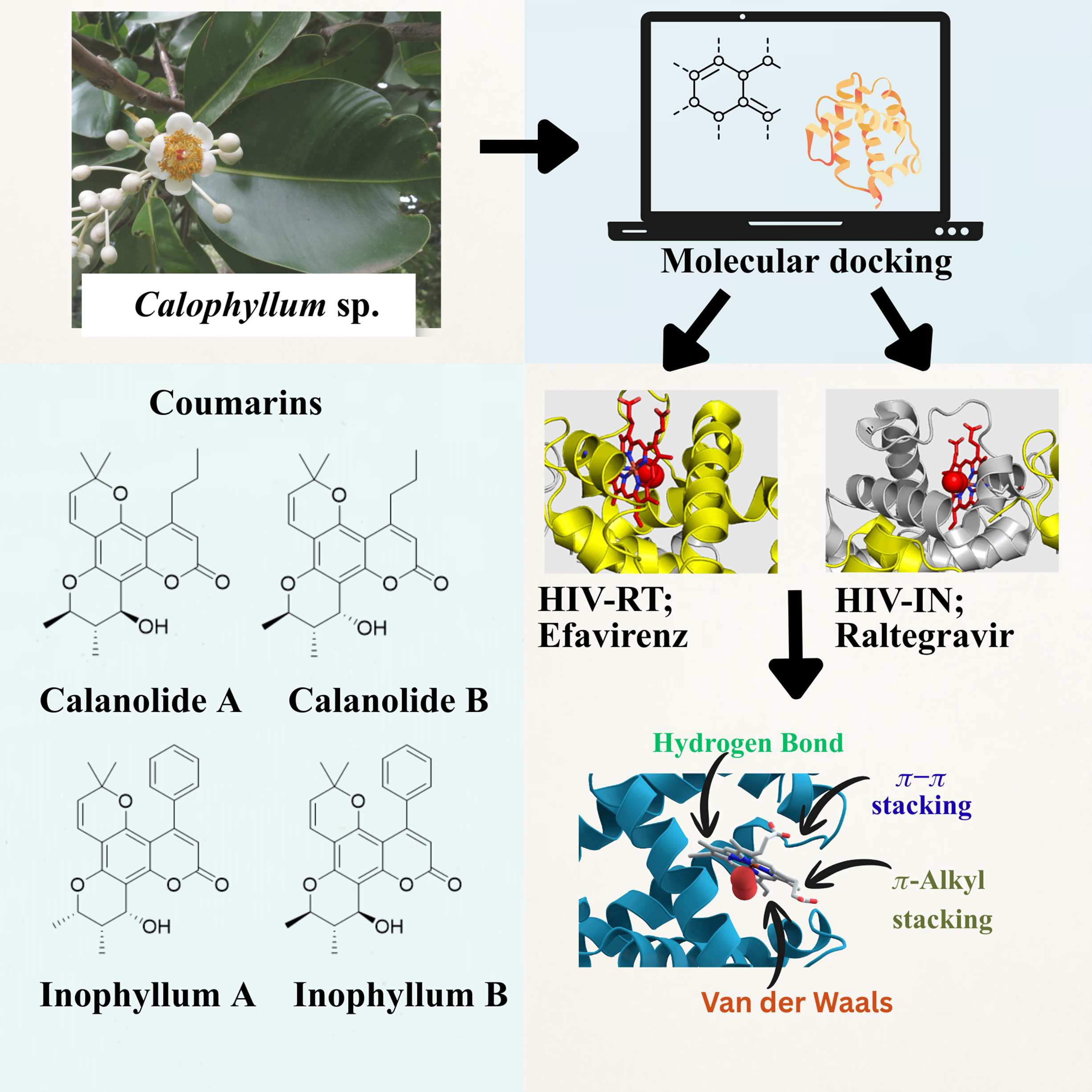
Downloads
References
S Ferdosh. The extraction of bioactive agents from Calophyllum inophyllum L. and their pharmacological properties. Scientia Pharmaceutica 2024; 92(1), 6.
A Atabani and AD César. Calophyllum inophyllum L. A prospective non-edible biodiesel feedstock. Study of biodiesel production, properties, fatty acid composition, blending and engine performance. Renewable and Sustainable Energy Reviews 2014; 37, 644-655.
HM Alshibl, ES Al-Abdullah, ME Haiba, HM Alkahtani, GEA Awad, AH Mahmoud, BMM Ibrahim, A Bari and A Villinger. Synthesis and evaluation of new coumarin derivatives as antioxidant, antimicrobial, and anti-inflammatory agents, Molecules 2020; 25(14), 3251.
SJ Hamid and T Salih. Design, synthesis, and anti-inflammatory activity of some coumarin Schiff base derivatives: In silico and in vitro study. Drug Design, Development and Therapy 2022; 16, 2275-2288.
KV Sashidhara, M Kumar, RK Modukuri, R Sonkar, G Bhatia, A Khanna, S Rai and R Shukla. Synthesis and anti-inflammatory activity of novel biscoumarin-chalcone hybrids. Bioorganic & Medicinal Chemistry Letters 2011; 21(15), 4480-4484.
A Pribowo, J Girish, M Gustiananda, RG Nandhira and P Hartrianti. Potential of tamanu (Calophyllum inophyllum) oil for atopic dermatitis treatment. Evidence-Based Complementary and Alternative Medicine 2021; 2021(1), 6332867.
Y Kashman, KR Gustafson, RW Fuller, JH Cardellina, JB McMahon, MJ Currens, RW Buckheit, SH Hughes, GM Cragg and MR Boyd. The calanolides, a novel HIV-inhibitory class of coumarin derivatives from the tropical rainforest tree Calophyllum lanigerum. Journal of Medicinal Chemistry 1992; 35(15), 2735-2743.
MP de Béthune. Non-nucleoside reverse transcriptase inhibitors (NNRTIs), their discovery, development, and use in the treatment of HIV-1 infection: A review of the last 20 years (1989-2009). Antiviral Research 2010; 85(1), 75-90.
RW Buckheit, EL White, V Fliakas-Boltz, J Russell, TL Stup, TL Kinjerski, MC Osterling, A Weigand and JP Bader. Unique anti-human immunodeficiency virus activities of the nonnucleoside reverse transcriptase inhibitors calanolide A, costatolide, and dihydrocostatolide. Antimicrobial Agents and Chemotherapy 1999; 43(8), 1827-1834.
NLDL Mata, N Kumarasamy, V Khol, OT Ng, KV Nguyen, TP Merati, TT Pham, MP Lee, N Durier and M Law. Improved survival in HIV treatment programmes in Asia. Antiviral Therapy 2016; 21(6), 517-527.
M Li, F. Yu, B Zhu, J Xiao, C Yan, X Yang, X Liang, F Wang, H Zhang and F Zhang. Interactions between human immunodeficiency virus and human endogenous retroviruses. Journal of Virology 2025; 99(3), e02319.
Z Hosseini, A Ebadi, T Aghamolaei and S Nedjat. A model for explaining adherence to antiretroviral therapy in patients with HIV/AIDS: A grounded theory study. Health & Social Care in the Community 2022; 30(6), e5735.
Q Xiao, D Guo and S Chen. Application of CRISPR/Cas9-based gene editing in HIV-1/AIDS therapy. Frontiers in Cellular and Infection Microbiology 2019; 9, 69.
ND Kolanu. CRISPR-Cas9 gene editing: Curing genetic diseases by inherited epigenetic modifications. Global Medical Genetics 2024; 11(01), 113-122.
H Lin, G Li, X Peng, A Deng, L Ye, L Shi, T Wang and J He. The use of CRISPR/Cas9 as a tool to study human infectious viruses. Frontiers in Cellular and Infection Microbiology 2021; 11, 590989.
AAD Zailan, T Karunakaran, MH Abu Bakar and VJY Mian. The Malaysian genus Calophyllum (Calophyllaceae): A review on its phytochemistry and pharmacological activities. Natural Product Research 2021; 36(17), 4569-4579.
V Flores-Morales, AP Villasana-Ruíz, I Garza-Veloz, S González-Delgado and ML Martinez-Fierro. Therapeutic effects of coumarins with different substitution patterns. Molecules 2023; 28(5), 2413.
OM Tsivileva, OV Koftin and NV Evseeva. Coumarins as fungal metabolites with potential medicinal properties. Antibiotics 2022; 11(9), 1156.
M Lončar, M Jakovljević, D Šubarić, M Pavlić, V Buzjak Služek, I Cindrić and M Molnar. Coumarins in food and methods of their determination. Foods 2020; 9(5), 645.
SD Sarker and L Nahar. Progress in the chemistry of naturally occurring coumarins. Progress in the Chemistry of Organic Natural Products 2017; 106, 241-304.
S Gupta and P Gupta. The genus Calophyllum: Review of ethnomedicinal uses, phytochemistry and pharmacology. In: J Singh, V Meshram and M Gupta (Eds.). Bioactive Natural Products in Drug Discovery. Springer, Singapore, 2020.
T Ma, P Zheng, X Li, X Hong and G Liu. Function, pharmaceutical, and pharmacological research and development of natural tetracyclic dipyranocoumarin (+)-calanolide A and its analogs. In: G Liu (Ed.). Medicinal chemistry in drug development. Elsevier, Amsterdam, Netherland, 2025, p. 651-687.
RK Arora, N Kaur, Y Bansal and G Bansal. Novel coumarin-benzimidazole derivatives as antioxidants and safer anti-inflammatory agents. Acta Pharmaceutica Sinica B 2014; 4(5), 368-375.
A Stefanachi, F Leonetti, L Pisani, M Catto and A Carotti. Coumarin: a natural, privileged and versatile scaffold for bioactive compounds. Molecules 2018; 23(2), 250.
NS Firouza, T Karunakaran, N Mokhtar, R Santhanam, VJY Mian and MH Abu Bakar. Chemical constituents from the stem barks of Calophyllum recurvatum P.F. Stevens and Calophyllum andersonii P.F. Stevens and their in vitro hepatotoxic activity. Natural Product Research 2024; 39(9), 2587-2593.
AD Patil, AJ Freyer, DS Eggleston, RC Haltiwanger, MF Bean, PB Taylor, MJ Caranfa, AL Breen, HR Bartus and RK Johnson. The inophyllums, novel inhibitors of HIV-1 reverse transcriptase isolated from the Malaysian tree Calophyllum inophyllum Linn. Journal of Medicinal Chemistry 1993; 36(26), 4131-4138.
S Cao, X Wu, K Sim, BH Tan, JJ Vittal, JT Pereira and S Goh. Minor coumarins from Calophyllum teysmannii var. inophylloide and synthesis of cytotoxic calanone derivatives. Helvetica Chimica Acta 1998; 81(5-8), 1404-1416.
TC McKee, CD Covington, RW Fuller, HR Bokesch, S Young, JH Cardellina, MR Kadushin, DD Soejarto, PF Stevens, GM Cragg and MR Boyd. Pyranocoumarins from tropical species of the genus Calophyllum: a chemotaxonomic study of extracts in the National Cancer Institute collection. Journal of Natural Products 1998; 61(10), 1252-1256.
KR Gustafson, HR Bokesch, RW Fuller, JH Cardellina, MR Kadushin, DD Soejarto and MR Boyd. Calanone, a novel coumarin from Calophyllum teysmannii. Tetrahedron Letters 1994; 35(32), 5821-5824.
MB Zakaria, Vijayasekaran, Z Ilham and NA Muhamad. Anti-inflammatory activity of Calophyllum inophyllum fruit extracts. Procedia Chemistry 2014; 13, 218-220.
NI Aminudin, F Ahmad, M Taher and RM Zulkifli. α-Glucosidase and 15-lipoxygenase inhibitory activities of phytochemicals from Calophyllum symingtonianum. Natural Product Communications 2015; 10(9), 1585-1587.
TC McKee, RW Fuller, CD Covington, JH Cardellina, RJ Gulakowski, BL Krepps and JB McMahon, MR Boyd. New pyranocoumarins isolated from Calophyllum lanigerum and Calophyllum teysmannii. Journal of Natural Products 1996; 59(8), 754-758.
T Ishikawa. Anti-HIV-1 active Calophyllum coumarins: distribution, chemistry, and activity. Heterocycles 2000; 53(2), 453-474.
M Huerta-Reyes, MC Basualdo, F Abe, M Jimenez-Estrada, C Soler and R Reyes-Chilpa. HIV-1 inhibitory compounds from Calophyllum brasiliense leaves. Biological and Pharmaceutical Bulletin 2004; 27(9), 1471-1475.
S Cao, K Sim, J Pereira and S Goh. Coumarins from Calophyllum teysmannii (Guttiferae). Phytochemistry 1998; 47(5), 1051-1055.
N Mokhtar, T Karunakaran, R Santhanam, MH Abu Bakar and VJY Mian. Phenolics and triterpenoids from stem bark of Calophyllum lanigerum var. austrocoriaceum (Whitmore) P.F. Stevens and their cytotoxic activities. Natural Product Research 2024; 38(5), 873-878.
F Laure, P Raharivelomanana, J Butaud, J Bianchini and EM Gaydou. Screening of anti-HIV-1 inophyllums by HPLC-DAD of Calophyllum inophyllum leaf extracts from French Polynesia islands. Analytica Chimica Acta 2008; 624(1), 147-153.
SP Joshi, VB Deodhar and UD Phalgune. ChemInform abstract: a new coumarin from the seeds of Calophyllum inophyllum Linn. Indian Journal of Chemistry 2000; 39, 560-561.
SP Joshi, SR Kulkarni, UD Phalgune and VG Puranik. New dipyranocoumarin from the leaves of Calophyllum apetalum Willd. Natural Product Research 2013; 27, 1896-1901.
D Guilet, JJ Hélesbeux, D Séraphin, T Sévenet, P Richomme and J Bruneton. Novel cytotoxic 4-phenylfuranocoumarins from Calophyllum dispar. Journal of Natural Products 2001; 64(5), 563-568.
D Guilet, D Séraphin, D Rondeau, P Richomme and J Bruneton. Cytotoxic coumarins from Calophyllum dispar. Phytochemistry 2001; 58(4), 571-575.
NH Zamakshshari, GCL Ee, SS Teh, SK Daud and I Safinar. Natural product compounds from Calophyllum depressinervosum. Pertanika Journal of Tropical Agricultural Science 2016; 39(2), 249-255.
SB Daud, GC Ee, EA Malek, SS The and I See. A new coumarin from Calophyllum hosei. Natural Product Research 2014; 28(19), 1534-1538.
CK Lim, S Hemaroopini, SY Gan, SM Loo, JR Low, VY Jong, HC Soo, CO Leong, CW Mai and CF Chee. In vitro cytotoxic activity of isolated compounds from Malaysian Calophyllum species. Medicinal Chemistry Research 2016; 25(8), 1686-1694.
FL Yong. 2015, Phytochemical and antioxidant studies of Calophyllum sclerophyllum. Bachelor’s Thesis. Universiti Tunku Abdul Rahman, Perak, Malaysia.
IA Noh, VJY Mian. Phytochemicals, antimicrobials and antioxidants studies of the stem bark extract from Calophyllum ferrugineum. Scientific Research Journal 2020; 17(2), 1-12.
G Ee, K Ng, Y Taufiq-Yap, M Rahmani, A Ali and R Muse. Mucigerin, a new coumarin from Calophyllum mucigerum (Guttiferae). Natural Product Research 2004; 18(2), 123-128.
NI Aminudin, F Ahmad, M Taher and RM Zulkifli. Incrassamarin A-D: Four new 4-substituted coumarins from Calophyllum incrassatum and their biological activities. Phytochemistry Letters 2016; 16, 287-293.
GC Ee, SH Mah, SS Teh, M Rahmani, R Go and YH Taufiq-Yap. Soulamarin, a new coumarin from stem bark of Calophyllum soulattri. Molecules 2011; 16(11), 9721-9727.
KH Tee, GC Ee, IS Ismail, T Karunakaran, SS Teh, VY Jong and SM Mohd Nor. A new coumarin from stem bark of Calophyllum wallichianum. Natural Product Research 2018; 32(21), 2565-2570.
MSM Sahimi, GCL Ee, SS Teh, AAF Ismail and MA Sukari. Chemical constituents of Calophyllum benjaminum and Calophyllum javanicum and their bioactivities. Open Conference Proceedings Journal 2013; 4, 127.
CK Lim, YP Ham, LQ Lim and VY Jong. 4-Alkylcoumarins and a phloroglucinol from the stem bark of Calophyllum gracilentum. Phytochemistry Letters 2019; 30, 99-102.
AS Salihu, WMNHW Salleh and T Ogunwa. Computational exploration of flavonoids from the genus Knema with anti-inflammatory potential. Journal of the Serbian Chemical Society 2024; 89(7-8), 1039-1051.
PC Agu, CA Afiukwa, OU Orji, EM Ezeh, IH Ofoke, CO Ogbu, EI Ugwuja and PM Aja. Molecular docking as a tool for the discovery of molecular targets of nutraceuticals in diseases management. Scientific Reports 2023; 13(1), 40160.
MS Bilal, SA Ejaz, S Naseem, PA Channar, A Saeed, S Zargar, R Ujan, R Sahito, Q Abbas and TA Wani. Synthesis, in vitro evaluation and computational modelling of benzene sulfonamide derivatives as Dickkopf-1 inhibitors for anticancer drug development. Scientific Reports 2025; 15(1), 68901.
CA Lipinski. Lead- and drug-like compounds: The rule-of-five revolution. Drug Discovery Today: Technologies 2004; 1(4), 337-341.
A Daina, O Michielin and V Zoete. SwissADME: A free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Scientific Reports 2017; 7(1), 42717.
DE Pires, TL Blundell and DB Ascher. pkCSM: Predicting small-molecule pharmacokinetic and toxicity properties using graph-based signatures. Journal of Medicinal Chemistry 2015; 58(9), 4066-4072.
HR Dharmaratne, WM Wanigasekera, E Mata-Greenwood and JM Pezzuto. Inhibition of human immunodeficiency virus type-1 reverse transcriptase activity by cordatolides isolated from Calophyllum cordato-oblongum. Planta Medica 1998; 64(05), 460-461.
MC Yimdjo, AG Azebaze, AE Nkengfack, AM Meyer, B Bodo and ZT Fomum. Antimicrobial and cytotoxic agents from Calophyllum inophyllum. Phytochemistry 2004; 65(20), 2789-2795.
T Pengsuparp, M Serit, SH Hughes, DD Soejarto and JM Pezzuto. Specific inhibition of human immunodeficiency virus type-1 reverse transcriptase mediated by soulattrolide, a coumarin isolated from the latex of Calophyllum teysmannii. Journal of Natural Products 1996; 59(9), 839-842.
L Nahar, AD Talukdar, D Nath, S Nath, A Mehan, FMD Ismail and SD Sarker. Naturally occurring calanolides: Occurrence, biosynthesis, and pharmacological properties including therapeutic potential. Molecules 2020; 25(21), 4983.
C Ito, M Itoigawa, Y Mishina, VC Filho, F Enjo, H Tokuda, H Nishino and H Furukawa. Chemical constituents of Calophyllum brasiliense. 2. Structure of three new coumarins and cancer chemopreventive activity of 4-substituted coumarins. Journal of Natural Products 2003; 66(3), 368-371.
HR Dharmaratne, JR Mayuri Sajeevani, GP Marasinghe and EMS Ekanayake. Distribution of pyranocoumarins in Calophyllum cordato-oblongum. Phytochemistry 1998; 49(4), 995-998.
V Nguyen, C Truong, BC Nguyen, TV Vo, T Dao, V Nguyen, DT Trinh, HK Huynh and C Bui. Anti-inflammatory and wound healing activities of calophyllolide isolated from Calophyllum inophyllum Linn. PloS One 2017; 12(10), e0185674.
C Ito, T Murata, M Itoigawa, K Nakao, N Kaneda and H Furukawa. Apoptosis-inducing activity of 4-substituted coumarins from Calophyllum brasiliense in human leukaemia HL-60 cells. Journal of Pharmacy and Pharmacology 2006; 58(7), 975-980.
RC Saxena, R Nath, G Palit, SK Nigam and KP Bhargava. Effect of calophyllolide, a nonsteroidal anti-inflammatory agent, on capillary permeability. Planta Medica 1982; 44(04), 246-248.
N Jaiswal, N Gunaganti, CK Maurya, T Narender and AK Tamrakar. Free fatty acid-induced impairment of insulin signaling is prevented by the diastereomeric mixture of calophyllic acid and isocalophyllic acid in skeletal muscle cells. European Journal of Pharmacology 2015; 746, 70-77.
J Prasad, A Shrivastava, AK Khanna, G Bhatia, SK Awasthi and T Narender. Antidyslipidemic and antioxidant activity of the constituents isolated from the leaves of Calophyllum inophyllum. Phytomedicine 2012; 19(14), 1245-1249.
F Zafar, A Gupta, K Thangavel, K Khatana, AA Sani, A Ghosal, P Tandon and N Nishat. Physicochemical and pharmacokinetic analysis of anacardic acid derivatives. ACS Omega 2020; 5(11), 6021-6030.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






