UHPLC Analysis of Phenolic Compounds from Phanera strychnifolia Leaf Extracts, and Their Alpha-glucosidase and Alpha-Amylase Inhibitory Activities Supported by Molecular Docking
DOI:
https://doi.org/10.48048/tis.2026.13308Keywords:
Bauhinia strychnifolia, Diabetes mellitus, Extraction, Lysiphyllum strychnifolium, α-Glucosidase inhibitory activity, Molecular docking, Quantitative analysis, Bauhinia strychnifolia, Diabetes mellitus, Extraction, Lysiphyllum strychnifolium, α-Glucosidase inhibitory activity, Molecular docking, Quantitative analysisAbstract
Phanera strychnifolia is a traditional medicinal plant showing remarkable antihyperglycemic activity. To develop as a phytopharmaceutical product for Diabetes, an in-depth investigation of the α-glucosidase and α-amylase inhibitory activities of the main constituents was conducted. The developed UHPLC method was validated for qualitative and quantitative analyses of the major constituents gallic acid (1), trilobatin (2), yanangdaengin (3), and phloretin (4). The performed stress tests provide information about storage conditions and about the stabilities of these compounds under various conditions. The optimized extraction with ethanol/water (1:1, v/v) gave the highest yield of gallic acid (1), and the dihydrochalcones trilobatin (2), yanangdaengin (3), and phloretin (4). Yanangdaengin (3) exhibited potent α-glucosidase inhibitory activity (IC50 of 10.42 µM), which was about 30 times higher active than the commercial drug acarbose (IC50 of 329.09 µM). This result was also confirmed by computerized molecular docking. Our work confirms the suitability of hydrophilic leaf extracts from P. strychnifolia as a potential herbal medicine for the treatment of postprandial hyperglycemia and provides valuable information for the phytopharmaceutical development of this plant.
HIGHLIGHTS
- Alpha-glucosidase and alpha-amylase inhibitory activities from Phanera strychnifolia leaf extracts were studied. The bioactive compounds were confirmed with molecular docking.
- Yanangdaengin (3) exhibited potent α-glucosidase inhibitory activity (IC50 of 10.42 µM), which was about 30 times more active than the commercial drug acarbose (IC50 of 329.09 µM).
- The UHPLC method for qualitative and quantitative analyses of gallic acid (1), trilobatin (2), yanangdaengin (3), and phloretin (4), the major constituents in Phanera strychnifolia leaf extract was developed and validated.
- A suitable extraction method of this plant was developed and optimized. Extraction with ethanol/water (1:1, v/v) gave the highest yield of gallic acid (1), and the dihydrochalcones trilobatin (2), yanangdaengin (3), and phloretin (4).
GRAPHICAL ABSTRACT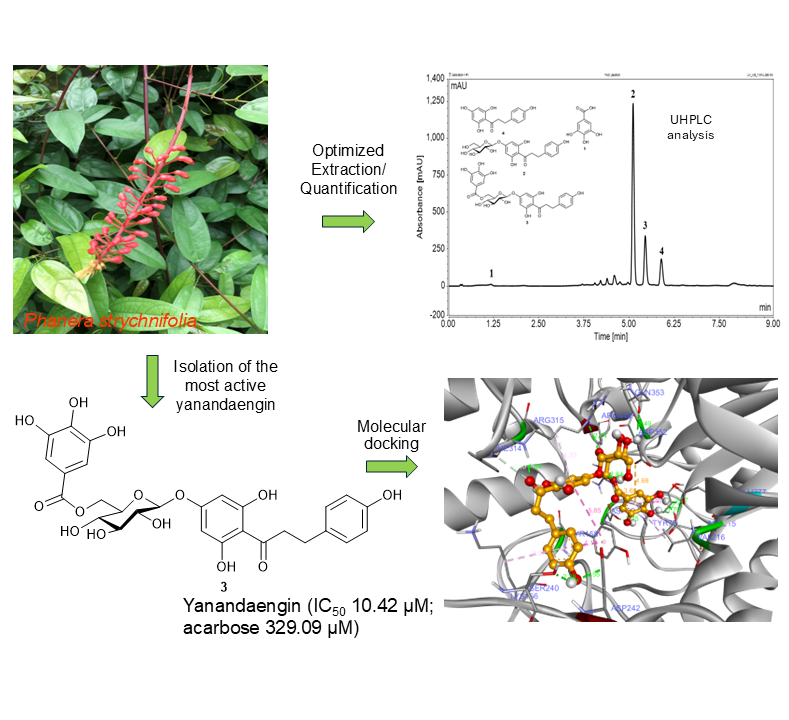
Downloads
References
SA Antar, NA Ashour, M Sharaky, M Khattab, NA Ashour, RT Zaid, EJ Roh, A Elkamhawy and AA Al-Karmalawy. Diabetes mellitus: Classification, mediators, and complications; A gate to identify potential targets for the development of new effective treatments. Biomedicine & Pharmacotherapy 2023; 168, 115734.
U Galicai-Garcia, A Benito-Vicente, S Jebari, A Larrea-Sebal, H Siddiqi, KB Uribe, H Ostolaza and C Martin. Pathophysiology of Type 2 Diabetes Mellitus. International Journal of Molecular Sciences 2020; 21, 6275.
GBD 2021 Diabetes Collaborators. Global, regional, and national burden of diabetes from 1990 to 2021, with projections of prevalence to 2050: A systematic analysis for the Global Burden of Disease Study 2021. Lancet 2023; 402, 203-234.
DP Farias, FF de Araujo, IA Neri-Numa and GM Pastore. Antidiabetic potential of dietary polyphenols: A mechanistic review. Food Research International 2021; 145, 110383.
K Shapiro and WC Gong. Natural Products Used for Diabetes. Journal of the American Pharmaceutical Association 2002; 42, 217-222.
KW Jiang, SR Gu, SJ Li, TY Tu and DX Zhang. Three new combinations in the genus Phanera (Fabaceae: Cercidoideae) including the lectotypification of Bauhinia strychnifolia. Phytotaxa 2023; 584, 52-54.
R Pooma and S Suddee. Thai plant names. Tem Smitinand revised edition 2014. Office of the Forest Herbarium, Department of National Park, Wildlife and Plant Conservation, Bangkok, Thailand, 2014.
W Wuttidhamavej. Thai Traditional Medicine. Odean Store Press, Bangkok, Thailand, 1997.
AS Goli, VH Sato, H Sato, S Chewchinda, J Leanpolchareanchai, J Nontakham, J Yahuafai, T Thilavech, P Meesawatsom and M Maitree. Antihyperglycemic effects of Lysiphyllum strychnifolium leaf extract in vitro and in vivo. Pharmaceutical Biology 2023; 61(1), 189-200.
VH Sato, S Chewchinda, N Nuamnaichati, S Mangmool, B Sungthong, P Lertsatitthanakorn, S Ohta and H Sato. Pharmacological mechanisms of the water leaves extract of Lysiphyllum strychnifolium for its Anti-Inflammatory and Anti-Hyperuricemic actions for gout treatment. Pharmacognosy Magazine 2019; 14(60), 98.
S Kongkiatpaiboon, N Duangdee, N Tayana, J Schinnerl, M Bacher and S Chewchinda. Yanangdaengin, a dihydrochalcone glucoside galloyl ester as active antioxidative agent from leaves of Lysiphyllum strychnifolium (syn. Bauhinia strychnifolia). Chinese Herbal Medicine 2020; 12(2), 452-455.
M Kidruangphokin, N Suphrom and S Boonphong. -Glucosidase Inhibitory and Antioxidant Activities of Ethanolic Extracts of Different Parts of Lysiphyllum strychnifolium and Their Constituents. Journal of Herbs, Spices & Medicinal Plants 2023; 29(3), 274-287.
Validation of Analytical Procedures Q2(R2). Complete Revision of Guideline. 38. Available at: https://database.ich.org/sites/default/files/ICH_Q2-R2_Document_Step2_Guideline_2022_0324.pdf, accessed January 2026.
S Kongkiatpaiboon, N Duangdee, S Chewchinda, O Poachanukoon and K Amnuaypattanapon. Development and validation of stability indicating HPLC method for determination of adrenaline tartrate. Journal of King Saud University-Science 2019; 31(1), 48-51.
S Chewchinda, S Kongkiatpaiboon and P Sithisarn. Evaluation of antioxidant activities, total phenolics and total flavonoid contents of aqueous extracts of leaf, stem, and root of Aerva lanata. Chiang Mai University Journal of Natural Sciences 2019; 18(3), 345-357.
S Chewchinda, N Leakaya, H Sato and VH Sato. Antidiabetic effects of Maclura cochinchinensis (Lour.) Corner heartwood extract. Journal of Traditional and Complementary Medicine 2021; 11(1), 68-74.
T Limcharoen, P Chaniad, P Chonsut, C Punsawad, T Juckmeta, A Konyanee, IR Rais and S Sangkaew. Alpha-Glucosidase inhibition, antioxidant activities, and molecular docking study of krom luang chumphon khet udomsak, a Thai Traditional Remedy. Advances in Pharmacological and Pharmaceutical Science 2024; 2024, 1322310.
K Yamamoto, H Miyake, M Kusunoki and S Osaki. Crystal structures of isomaltases from Saccharomyces cerevisiae and in complex with its competitive inhibitor maltose. The FEBS Journal 2010; 277(20), 4205-4214.
J Eberhardt, D Santos-Martins, AF Tillack and S Forli. AutoDock Vina 1.2.0: New docking methods, expanded force field, and python bindings. Journal of Chemical Information and Modeling 2021; 61(8), 3891-3898.
O Trott and AJ Olson. AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. Journal of Computational Chemistry 2010; 31(2), 455-461.
K Thongra-Ar, P Rojsanga, S Chewchinda, S Mangmool and P Sithisarn. Antioxidant, α-glucosidase and α-amylase inhibitory activities of Persicaria odorata. Chiang Mai University Journal of Natural Sciences 2021; 20(3), e2021051.
L Wang, M Liu, F Yin, Y Wang, X Li, Y Wu, C Ye and J Liu. Trilobatin, a novel SGLT1/2 inhibitor, selectively induces the proliferation of human hepatoblastoma cells. Molecules 2019; 24(18), 3390.
M Lunder, M Janic, M Sabovic and A Janez. SGLT-2 inhibitors: A novelty in the treatment of type 2 diabetes. Zdravniski Vestnik 2018; 87, 493-505.
H Wang, MI Fowler, DJ Messenger, JJ Ordaz-Ortiz, X Gu, S Shi, LA Terry, MJ Berry, G Lian and S Wang. Inhibition of the intestinal postprandial glucose transport by gallic acid and gallic acid derivatives. Food & Function 2021; 12(12), 5399-5406.
S Kongkiatpaiboon, N Tayana, W Inthakusol, N Duangdee, S Chewchinda and V Keeratinijakal. HPLC quantification of chemical markers from Lysiphyllum strychnifolium. Revista Brasileira de Farmacognosia 2022; 32, 266-272.
W Songoen, W Phanchai, J Schinnerl, L Brecker, M Thabpho, S Tharamak, W Pluempanupat, S Sukkhaeng and S Chansuthep. Identification of specialized metabolites from Artocarpus lacucha as potent alpha-glucosidase and acetylcholinesterase inhibitors: Enzyme kinetic, in vitro and in silico study. Journal of Natural Medicines 2025; 79(4), 896-912.
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






