In vitro Inhibition of Protein Glycation, Methylglyoxal, α-Glucosidase, and Metal Ions by a Thai Polyherbal Formulation
DOI:
https://doi.org/10.48048/tis.2026.12635Keywords:
Benchalokawichian Remedy, Flavonoids, α-glucosidase, Metal chelation, Antiglycation, Benchalokawichian remedy, Flavonoids, α-glucosidase, Metal chelation, Antiglycation, Methylglyoxal-trappingAbstract
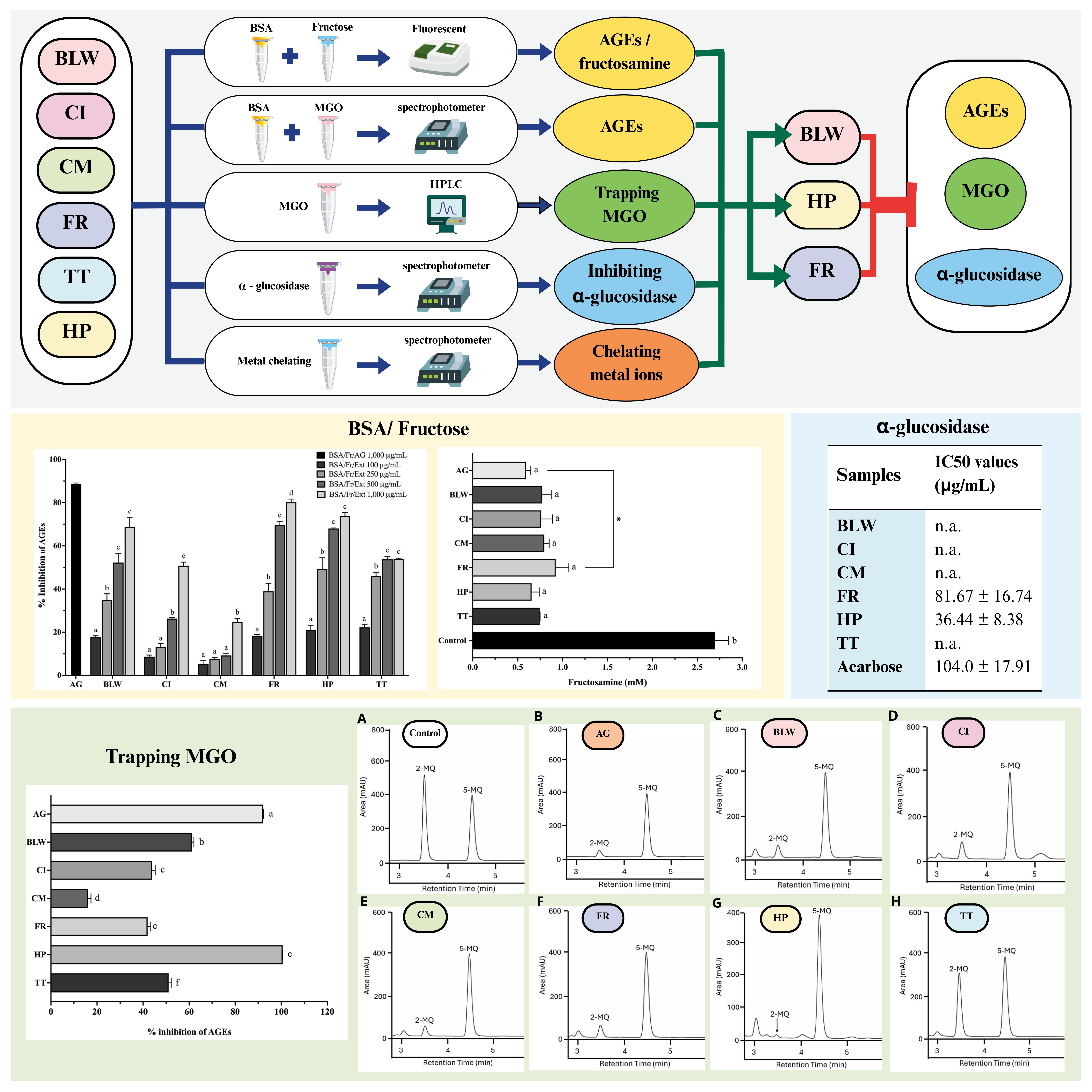
Glycation processes generate advanced glycation end products (AGEs) and intermediates, such as methylglyoxal (MGO), which contribute to non-communicable diseases like diabetes and cardiovascular and cerebrovascular disorders. This study explores the antiglycation, carbohydrate-hydrolyzing enzyme inhibition, MGO-trapping, and metal-chelating properties of hydroethanolic extracts of Benchalokawichian (BLW) and its individual herbal components. The total flavonoid content was assessed using the aluminum chloride method, revealing Tiliacora triandra and Ficus racemosa as rich flavonoid sources. Antiglycation activity was evaluated using bovine serum albumin (BSA)/fructose and BSA/MGO models, with aminoguanidine (AG) as a positive control. All extracts demonstrated concentration-dependent antiglycation effects. Notably, Harrisonia perforata at 1 mg/mL significantly inhibited AGE formation by 73.79 ± 1.55% in BSA/fructose and 99.58 ± 0.11% in BSA/MGO models, and exhibited a substantial MGO-trapping efficiency of 98.19 ± 0.02%, surpassing the standard agent AG at 92.24 ± 0.12%. The extracts’ capacity to inhibit carbohydrate-hydrolyzing enzymes was assessed through α-glucosidase activity. H. perforata and F. racemosa showed significant inhibitory effects, with IC50 values of 36.44 ± 8.38 and 81.67 ± 16.74 μg/mL, respectively, outperforming the standard drug acarbose (IC50 104 ± 17.91 μg/mL). Metal chelation capacity, measured through an iron-ferrozine assay, was consistently lower than that of the standard chelating agent EDTA across all tested extracts. In conclusion, H. perforata demonstrates promising potential in reducing glycation and inhibiting α-glucosidase enzymes involved in glucose metabolism, indicating its possible application in functional foods or as a therapeutic agent for diabetes management. Further research is warranted to explore its clinical benefits.
HIGHLIGHTS
- Benchalokawichian (BLW) and its herbal components demonstrate concentration-dependent antiglycation activity.
- Ethanol root extracts of Harrisonia perforata (HP), Ficus racemosa (FR), and BLW effectively trap methylglyoxal and inhibit AGE formation.
- HP and FR exhibit superior α-glucosidase inhibitory IC50 values compared to the standard drug acarbose.
- Tilacora triandra root extract has the highest flavonoid content but lacks significant antiglycation or antidiabetic effects.
- HP, FR, and BLW hold promise as antiglycation agents for functional foods or therapeutic use.
GRAPHICAL ABSTRACT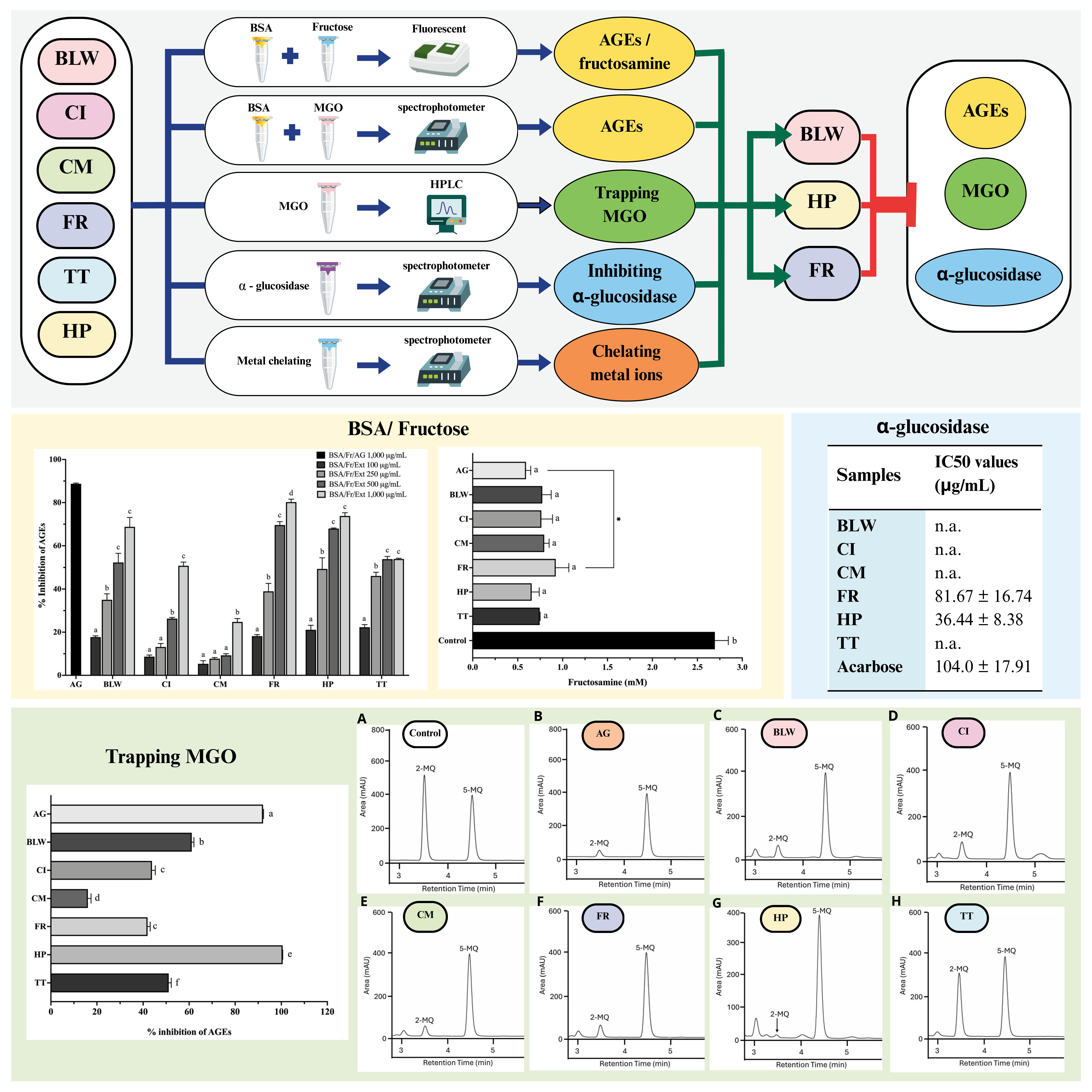
Downloads
References
E Qaed, W Aldahmash and MA Mahyoub. Advanced glycation end products (AGEs) and their role in diabetes mellitus and related complications: Mechanisms and therapeutic insights. Glycoconjugate Journal 2025; 42, 209-223.
MI Khan, F Ashfaq, AA Alsayegh, A Hamouda, F Khatoon, TN Altamimi, FS Alhodieb and MMA Beg. Advanced glycation end product signaling and metabolic complications: Dietary approach. World Journal of Diabetes 2023; 14(7), 995-1012.
AL Oliveira, MG de Oliveira, FZ Mónica and E Antunes. Methylglyoxal and advanced glycation end products (AGEs): Targets for the prevention and treatment of diabetes-associated bladder dysfunction? Biomedicines 2024; 12(5), 939.
CG Schalkwijk and CDA Stehouwer. Methylglyoxal, a highly reactive dicarbonyl compound, in diabetes, its vascular complications, and other age-related diseases. Physiological Reviews 2020; 100(1), 407-461.
P Vangrieken, JLJM Scheijen, PMH Schiffers, MPH Van De Waarenburg, S Foulquier and CCG Schalkwijk. Modelling the effects of elevated methylglyoxal levels on vascular and metabolic complications. Scientific Reports 2025; 15(1), 6025.
Q Song, J Liu, L Dong, X Wang and X Zhang. Novel advances in inhibiting advanced glycation end product formation using natural compounds. Biomedicine & Pharmacotherapy 2021; 140, 111750.
NT Moldogazieva, IM Mokhosoev, TI Mel’nikova, YB Porozov and AA Terentiev. Oxidative stress and advanced lipoxidation and glycation end products (ALEs and AGEs) in aging and age-related diseases. Oxidative Medicine and Cellular Longevity 2019; 2019, 3085756.
I Sakpakdeejaroen, T Juckmeta and A Itharat. Development and validation of RP-HPLC method to determine anti-allergic compound in Thai traditional remedy called Benjalokawichian. Journal of the Medical Association of Thailand 2014; 97(S8), S76-S80.
C Singharachai, C Palanuvej, H Kiyohara, H Yamada and N Ruangrungsi. Pharmacognostic specification of five root species in Thai traditional medicine remedy: Ben-Cha-Lo-Ka-Wi-Chian. Pharmacognosy Journal 2011; 3(21), 1-11.
P Nadee, A Jaruchotikamol and B Cushnie. Antioxidant capacity and phytochemical analysis of the traditional Thai remedy Benchalokawichian. Tropical Journal of Natural Product Research 2024; 8(8), 8054-8060.
T Juckmeta and A Itharat. Anti-inflammatory and antioxidant activities of Thai traditional remedy called “Ya-ha-rak”. Journal of Health Research 2012; 26(4), 205-210.
W Chandranipapongse. Study the effect of an antipyretic drug, Thai herbal Ha-Rak formula on platelet aggregation in healthy Thai volunteers: A randomized, placebo - controlled trial. Siriraj Medical Journal 2017; 69(5), 283-289.
N Srichaipor, P Pongcharoen, P Kanokkangsadal and A Itharat. Skin irritation and allergic testing of Thai herbal extracts (Ha-Rak with Turmeric) in healthy volunteers. Thammasat Medical Journal 2020; 20(2), 165-174.
K Jirasripongpun, N Jirakanjanakit, S Pola and K Obsuwan. Antioxidation and anti-melanogenesis of three colored flowers of Dendrobium hybrids. Science, Engineering and Health Studies 2022; 16, 22030006.
GL Zuo, HY Kim, YN Guillen Quispe, ZQ Wang, SH Hwang, KO Shin and SS Lim. Efficient separation of phytochemicals from Muehlenbeckia volcanica (Benth.) Endl. by polarity-stepwise elution counter-current chromatography and their antioxidant, antiglycation, and aldose reductase inhibition potentials. Molecules 2021; 26(1), 224.
I Fecka, K Bednarska and A Kowalczyk. In vitro antiglycation and methylglyoxal trapping effect of Peppermint Leaf (Mentha × piperita L.) and its polyphenols. Molecules 2023; 28(6), 2865.
A Ali, R Shahu, P Balyan, S Kumari, R Ghodmare, R Jobby and P Jha. Antioxidation and antiglycation properties of a natural sweetener: Stevia rebaudiana. Sugar Tech 2022; 24(2), 563-575.
P Chayaratanasin, S Adisakwattana and T Thilavech. Protective role of Clitoria ternatea L. flower extract on methylglyoxal-induced protein glycation and oxidative damage to DNA. BMC Complementary Medicine and Therapies 2021; 21(1), 80.
S Amin, B Ullah, M Ali, A Rauf, H Khan, E Uriarte and E Sobarzo-Sánchez. Potent in vitro α-glucosidase inhibition of secondary metabolites derived from Dryopteris cycadina. Molecules 2019; 24(3), 427.
İ Gulcin and SH Alwasel. Metal ions, metal chelators and metal chelating assay as antioxidant method. Processes 2022; 10(1), 132.
LF Li, MD Wang, CY Zhang, MY Jin, HL Chen, H Luo, TY Hou, ZJ Zhang and H Li. Influence of hydroxyl substitution on the inhibition of flavonoids in advanced glycation end-products formation in glucose-lysine-arginine maillard reaction models. Food Research International 2025; 207, 116068.
Z Khiya, Y Oualcadi, A Gamar, F Berrekhis, T Zair and FE Hilali. Correlation of total polyphenolic content with antioxidant activity of hydromethanolic extract and their fractions of the Salvia officinalis leaves from different regions of Morocco. Journal of Chemistry 2021; 2021(1), 8585313.
W Semmarath, P Arjsri, K Srisawad, S Umsumarng and P Dejkriengkraikul. Luteolin-rich extract from Harrisonia perforata (Blanco) Merr. root alleviates SARS-CoV-2 spike protein-stimulated lung inflammation via inhibition of MAPK/NLRP3 inflammasome signaling pathways. Life 2025; 15(7), 1077.
FM Djeujo, V Stablum, E Pangrazzi, E Ragazzi and G Froldi. Luteolin and Vernodalol as bioactive compounds of leaf and root Vernonia amygdalina extracts: Effects on α-glucosidase, glycation, ROS, cell viability, and in silico ADMET parameters. Pharmaceutics 2023; 15(5), 1541.
N Yadav, JD Palkhede and SY Kim. Anti-glucotoxicity effect of phytoconstituents via inhibiting MGO-AGEs formation and breaking MGO-AGEs. International Journal of Molecular Sciences 2023; 24(8), 7672.
AB Uceda, L Mariño, R Casasnovas and M Adrover. An overview on glycation: Molecular mechanisms, impact on proteins, pathogenesis, and inhibition. Biophysical Reveiws 2024; 16(2), 189-218.
W Jia, R Ma, R Zhang, Z Fan and L Shi. Synthetic-free compounds as the potential glycation inhibitors performed in in vitro chemical models: Molecular mechanisms and structure requirements. Trends in Food Science & Technology 2022; 128, 147-159.
VP Reddy, P Aryal and EK Darkwah. Advanced glycation end products in health and disease. Microorganisms 2022; 10(9), 1848.
L Mou, P Hu, X Cao, Y Chen, Y Xu, T He, Y Wei and R He. Comparison of bovine serum albumin glycation by ribose and fructose in vitro and in vivo. Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease 2022; 1868(1), 166283.
B Stratmann. Dicarbonyl stress in diabetic vascular disease. International Journal of Molecular Sciences 2022; 23(11), 6186.
H Liu, X Huo, S Wang and Z Yin. The inhibitory effects of natural antioxidants on protein glycation as well as aggregation induced by methylglyoxal and underlying mechanisms. Colloids and Surfaces B: Biointerfaces 2022; 212, 112360.
AM Dirir, M Daou, AF Yousef and LF Yousef. A review of α-glucosidase inhibitors from plants as potential candidates for the treatment of type-2 diabetes. Phytochemistry Reviews 2022; 21(4), 1049-1079.
D Hadiarti, W Haryadi, S Matsjeh, RT Swasono and N Awaliyah. Profiling of α-glucosidase inhibitors from ethyl acetate fraction of Buas-buas (Premna serratifolia) leaves using UHPLC-Q-Orbitrap HRMS and protein-ligand interaction with molecular docking. Journal of Applied Pharmaceutical Science 2022; 13(2), 89-98.
S Lv, J Li, Y Hu, J Cai, G Nan, Y Kong, X Shen, L Zhu, S Yang and C Dong. Multi-target therapeutic potential of Arctii Fructus Lignans in diabetes mellitus and its complications: A mechanistic review. Pharmaceuticals 2025; 18(10), 1569.
SA Halim, S Jabeen, A Khan and A Al-Harrasi. Rational design of novel inhibitors of α-glucosidase: An application of quantitative structure activity relationship and structure-based virtual screening. Pharmaceuticals 2021; 14(5), 482.
N Pradubyat, F Madaka, T Songsak and S Jongrungruangchok. In vitro biological activity of Tiliacora triandra (Colebr.) Diels root extract. Journal of Current Science and Technology 2024; 15(1), 77.
R Evstatiev, A Cervenka, T Austerlitz, G Deim, M Baumgartner, A Beer, A Krnjic, C Gmainer, M Lang, A Frick, H Schachner, V Khare and C Gasche. The food additive EDTA aggravates colitis and colon carcinogenesis in mouse models. Scientific Reports 2021; 11(1), 5188.
P Mucha, A Skoczyńska, M Małecka, P Hikisz and E Budzisz. Overview of the antioxidant and anti-inflammatory activities of selected plant compounds and their metal ions complexes. Molecules 2021; 26(16), 4886.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






