Terpene-Based Phytochemicals as a Novel Strategy for Biofilm Disruption and Enhanced Wound Healing
DOI:
https://doi.org/10.48048/tis.2026.12629Keywords:
Wound healing, Chronic wounds, Biofilm disruption, Terpene-based phytochemicals, Antibiofilm agents, NanoemulsionsAbstract
Wound healing is a complex, multi-phase process involving hemostasis, inflammation, proliferation, and remodeling. Chronic wounds, including diabetic ulcers and pressure sores, often stall in the inflammatory phase due to persistent infections, oxidative stress, and impaired tissue regeneration. Microbial biofilms, particularly from Staphylococcus aureus and Pseudomonas aeruginosa, shield bacteria from host defenses and antibiotics, contributing to the majority of chronic infections and hospital-acquired infections. This review evaluates the antibiofilm and antimicrobial potential of terpene-based phytochemicals and examines their formulation into nanoemulsion-based delivery systems. Published studies were analyzed for mechanisms of biofilm disruption, bacterial inhibition, and enhancement of antimicrobial activity via nanoemulsions. Terpene phytochemicals disrupt biofilm architecture, inhibit bacterial proliferation, compromise cell membrane integrity, and interfere with quorum sensing and adhesion processes. When delivered in nanoemulsions, their solubility, stability, and bioavailability are improved, enhancing antimicrobial efficacy. These compounds also promote tissue repair and regeneration, addressing both infection control and wound healing. By simultaneously targeting biofilm-protected pathogens and supporting tissue regeneration, terpene-based phytochemicals offer a biocompatible, nature-inspired therapeutic strategy for chronic wound management. Their combined antibiofilm, antimicrobial, and tissue-regenerative actions underscore their potential as next-generation agents for advanced wound care.
HIGHLIGHTS
- Dual-action potential: Terpene phytochemicals disrupt biofilms and support tissue regeneration, addressing chronic wound challenges.
- Enhanced efficacy via nanoformulations: Nanoemulsions and nanocarriers improve stability, solubility, and bioavailability, enabling targeted delivery.
- Synergy with conventional therapies: Terpenes potentiate antibiotics, antimicrobial peptides, and photodynamic therapy, reducing resistance and accelerating healing.
- Advanced delivery strategies: Microneedles, hydrogels, and scaffold-based systems provide controlled, localized, and sustained release.
- Translational and regenerative promise: Integration with microbiome modulation, growth factors, and stem cell therapies positions terpenes as next-generation, patient-centered therapeutics.
GRAPHICAL ABSTRACT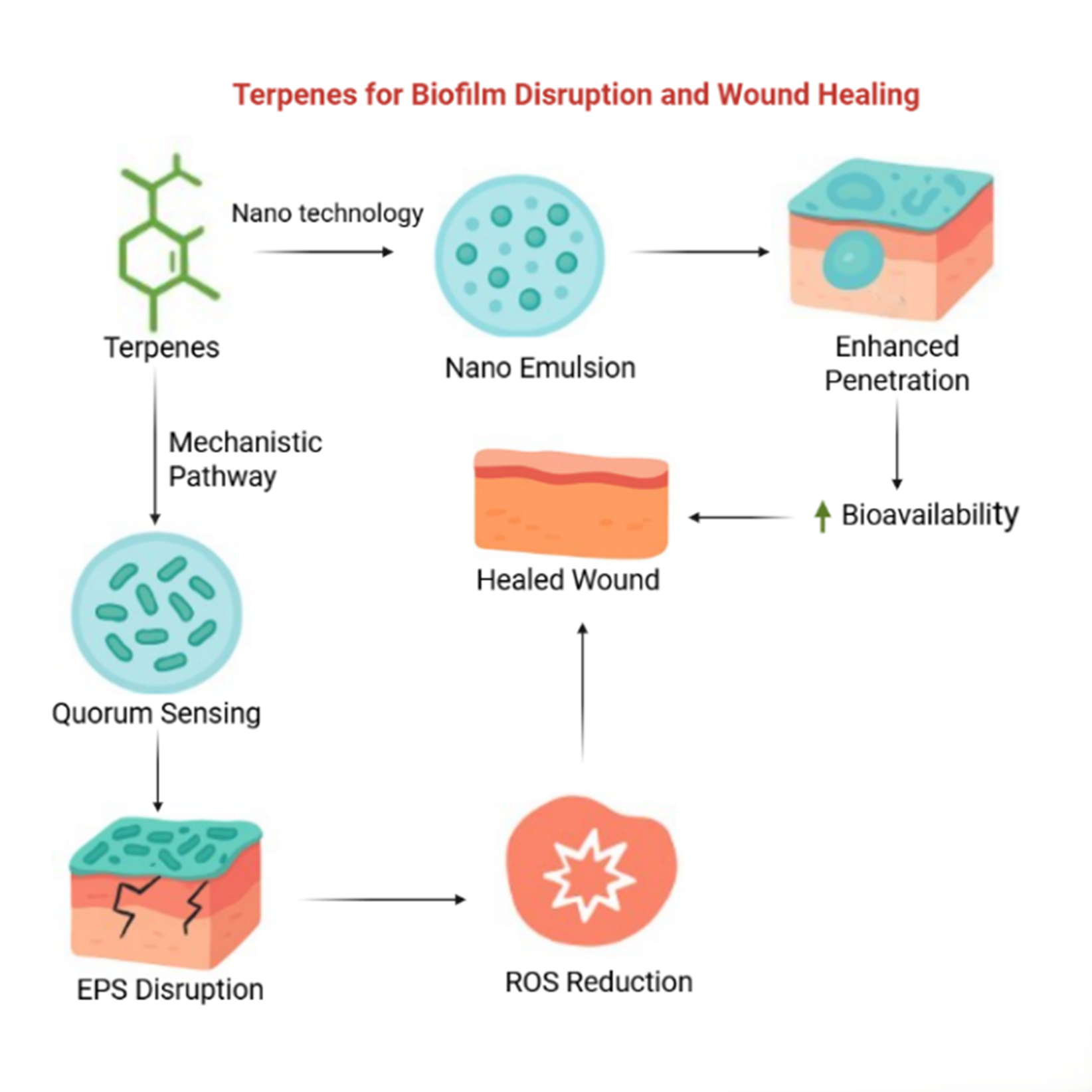
Downloads
References
M Bartold and S Ivanovski. Biological processes and factors involved in soft and hard tissue healing. Periodontology 2000 2025; 97(1), 16-42.
A Ahmad and SA Zainal. Photobiomodulation therapy for wound healing: A narrative review. IIUM Medical Journal Malaysia 2025; 24(1), 31-39.
S Enoch and DJ Leaper. Basic science of wound healing. Surgery 2005; 23(2), 37-42.
L Cañedo-Dorantes and M Cañedo-Ayala. Skin acute wound healing: a comprehensive review. International Journal of Inflammation 2019; 2019(1), 3706315.
L Häkkinen, H Larjava and L Koivisto. Granulation tissue formation and remodeling. Endodontic Topics 2011; 24(1), 94-129.
J M Reinke and H Sorg. Wound repair and regeneration. European Surgical Research 2012; 49(1), 35-43.
L Zhang, C Weng, Z Zhao and X Fu. Extracorporeal shock wave therapy for chronic wounds: A systematic review and meta-analysis of randomized controlled trials. Wound Repair and Regeneration 2017; 25(4), 697-706.
G Han and R Ceilley. Chronic wound healing: A review of current management and treatments. Advances in Therapy 2017; 34, 599-610.
H Strang, A Kaul, U Parikh, L Masri, S Saravanan, H Li, Q Miao and S Balaji. Role of cytokines and chemokines in wound healing. In: D Bagchi, A Das and S Roy (Eds.). Wound healing, tissue repair, and regeneration in diabetes. Academic Press, Massachusetts, United States, 2020, p. 197-235.
SM McCarty and SL Percival. Proteases and delayed wound healing. Advances in Wound Care 2013; 2(8), 438-447.
M Cano Sanchez, S Lancel, E Boulanger and R Neviere. Targeting oxidative stress and mitochondrial dysfunction in the treatment of impaired wound healing: A systematic review. Antioxidants 2018; 7(8), 98.
A Almatroudi. Biofilm resilience: Molecular mechanisms driving antibiotic resistance in clinical contexts. Biology 2025; 14(2), 165.
H Ragupathi, MM Pushparaj, SM Gopi, DK Govindarajan and K Kandaswamy. Biofilm matrix: A multifaceted layer of biomolecules and a defensive barrier against antimicrobials. Archives of Microbiology 2024; 206(11), 432.
T Bjarnsholt, K Buhlin, YF Dufrêne, M Gomelsky, A Moroni, M Ramstedt, KP Rumbaugh, T Schulte, L Sun, B Åkerlund and U Römling. Biofilm formation - what we can learn from recent developments. Journal of Internal Medicine 2018; 284(4), 332-345.
N Khardori and M Yassien. Biofilms in device-related infections. Journal of Industrial Microbiology and Biotechnology 1995; 15(3), 141-147.
MM Zafer, GA Mohamed, SR Ibrahim, S Ghosh, C Bornman and MA Elfaky. Biofilm-mediated infections by multidrug-resistant microbes: a comprehensive exploration and forward perspectives. Archives of Microbiology 2024; 206(3), 101.
NK Ragupathi, B Veeraraghavan, E Karunakaran and PN Monk. Biofilm-mediated nosocomial infections and its association with antimicrobial resistance: Detection, prevention, and management. Frontiers in Medicine 2022; 9, 987011.
RD Scott. The direct medical costs of healthcare-associated infections in US hospitals and the benefits of prevention, Available at: https://stacks.cdc.gov/view/cdc/11550/cdc_11550_DS1.pdf, accessed October 2025.
V Jean-Pierre, A Boudet, P Sorlin, Q Menetrey, R Chiron, JP Lavigne and H Marchandin. Biofilm formation by Staphylococcus aureus in the specific context of cystic fibrosis. International Journal of Molecular Sciences 2022; 24(1), 597.
C Moser, PØ Jensen, K Thomsen, M Kolpen, M Rybtke, AS Lauland, H Trøstrup and T Tolker-Nielsen. Immune responses to Pseudomonas aeruginosa biofilm infections. Frontiers in Immunology 2021; 12, 625597.
AS Pugazhendhi, F Wei, M Hughes and M Coathup. Bacterial adhesion, virulence, and biofilm formation. In: M Coathup (Ed.). Musculoskeletal infection. Springer International Publishing, Cham, Switzerland, 2022, p. 19-64.
I Ahmad, A Nadeem, F Mushtaq, N Zlatkov, M Shahzad, AV Zavialov, SN Wai and BE Uhlin. Csu pili dependent biofilm formation and virulence of Acinetobacter baumannii. npj Biofilms and Microbiomes 2023; 9(1), 101.
T Das, S Sehar and M Manefield. The roles of extracellular DNA in the structural integrity of extracellular polymeric substance and bacterial biofilm development. Environmental Microbiology Reports 2013; 5(6), 778-786.
CB Whitchurch, T Tolker-Nielsen, PC Ragas and JS Mattick. Extracellular DNA required for bacterial biofilm formation. Science 2002; 295, 1487.
PS Stewart, B White, L Boegli, T Hamerly, KS Williamson, MJ Franklin, B Bothner, GA James, S Fisher, FG Vital-Lopez and A Wallqvist. Conceptual model of biofilm antibiotic tolerance that integrates phenomena of diffusion, metabolism, gene expression, and physiology. Journal of Bacteriology 2019; 201(22), e00128-19.
K Azeem, S Fatima, A Ali, A Ubaid, FM Husain and M Abid. Biochemistry of bacterial biofilm: Insights into antibiotic resistance mechanisms and therapeutic intervention. Life 2025; 15(1), 49.
A Prinzi and R Rohde. The role of bacterial biofilms in antimicrobial resistance. American Society for Microbiology 2023; Available from: https://asm.org/articles/2023/march/the-role-of-bacterial-biofilms-in-antimicrobial-re
A Jain and DK Parihar. Antibacterial, biofilm dispersal and antibiofilm potential of alkaloids and flavonoids of Curcuma. Biocatalysis and Agricultural Biotechnology 2018; 16, 677-682.
X Song, YX Xia, ZD He and HJ Zhang. A review of natural products with anti-biofilm activity. Current Organic Chemistry 2018; 22(8), 789-817.
NA Mahizan, SK Yang, CL Moo, AAM Song, CM Chong, CW Chong, A Abushelaibi, SH Lim and KS Lai. Terpene derivatives as potential agents against antimicrobial resistance (AMR) pathogens. Molecules 2019; 24(14), 2631.
C Shi, M Che, X Zhang, Z Liu, R Meng, X Bu, H Ye and N Guo. Antibacterial activity and mode of action of totarol against Staphylococcus aureus in carrot juice. Journal of Food Science and Technology 2018; 55, 924-934.
R Jaiswal, TK Beuria, R Mohan, SK Mahajan and D Panda. Totarol inhibits bacterial cytokinesis by perturbing the assembly dynamics of FtsZ. Biochemistry 2007; 46(14), 4211-4220.
M Majid, H Naveed, SA Mohsin, MT Khan, W Hussain, Y Ruan, H Lu, H Fatima and X Hu. Na¬ture’s answer to superbugs: Berberis lycium and its potent isoquinoline alkaloid, Berberine-A, can target bacterial enzymome to combat antibiotic re¬sistance. Journal of Antibiotic Research 2024. http://dx.doi.org/10.2139/ssrn.4984042
Z Javanmard, M Pourhajibagher and A Bahador. Advancing anti-biofilm strategies: innovations to combat biofilm-related challenges and enhance efficacy. Journal of Basic Microbiology 2024; 64(12), e2400271.
A García, B González, C Harvey, I Izquierdo-Barba and M Vallet-Regí. Effective reduction of biofilm through photothermal therapy by gold core@shell based mesoporous silica nanoparticles. Microporous and Mesoporous Materials 2021; 328, 111489.
E Álvarez, B González, D Lozano, AL Doadrio, M Colilla and I Izquierdo-Barba. Nanoantibiotics based in mesoporous silica nanoparticles: new formulations for bacterial infection treatment. Pharmaceutics 2021; 13(12), 2033.
K Summer, Q Guo, L Liu, B Barkla, S Giles and K Benkendorff. Antimicrobial proteins from oyster hemolymph improve the efficacy of conventional antibiotics. PLoS One 2025; 20(1), e0312305.
O Alliod, JP Valour, S Urbaniak, H Fessi, D Dupin and C Charcosset. Preparation of oil-in-water nanoemulsions at large scale using premix membrane emulsification and Shirasu Porous Glass (SPG) membranes. Colloids and Surfaces A 2018; 557, 76-84.
S Noori, F Zeynali and H Almasi. Antimicrobial and antioxidant efficiency of nanoemulsion-based edible coating containing ginger (Zingiber officinale) essential oil and its effect on safety and quality attributes of chicken breast fillets. Food Control 2018; 84, 312-320.
H Cheng, Q Zhu and J Wan. Impact of cinnamaldehyde nanoemulsion on Listeria monocytogenes biofilms on broth-treated food contact surfaces. LWT 2025; 117500.
S Yao, L Hao, R Zhou, Y Jin, J Huang and C Wu. Multispecies biofilms in fermentation: biofilm formation, microbial interactions, and communication. Comprehensive Reviews in Food Science and Food Safety 2022; 21(4), 3346-3375.
CR Armbruster and MR Parsek. New insight into the early stages of biofilm formation. Proceedings of the National Academy of Sciences 2018; 115(17), 4317-4319.
T Tolker-Nielsen. Biofilm development. In: M Ghannoum, M Parsek, M Whiteley and PK Mukherjee (Eds.). Microbial biofilms. Wiley, New Jersey, United States, 2015, p. 51-66.
MS Khan, MM Altaf and I Ahmad. Chemical nature of biofilm matrix and its significance. In: I Ahmad and FM Husain. Biofilms in plant and soil health. Wiley, New Jersey, United States, 2017, p. 151-177.
L Karygianni, Z Ren, H Koo and T Thurnheer. Biofilm matrixome: Extracellular components in structured microbial communities. Trends in Microbiology 2020; 28(8), 668-681.
E Balducci, F Papi, DE Capialbi and LD Bino. Polysaccharides’ structures and functions in biofilm architecture of antimicrobial-resistant (AMR) pathogens. International Journal of Molecular Sciences 2023; 24(4), 4030.
J S Ramírez-Larrota and U Eckhard. An introduction to bacterial biofilms and their proteases, and their roles in host infection and immune evasion. Biomolecules 2022; 12(2), 306.
D Campoccia, L Montanaro and CR Arciola. Extracellular DNA (eDNA): A major ubiquitous element of the bacterial biofilm architecture. International Journal of Molecular Sciences 2021; 22(16), 9100.
DR Dixon and RP Darveau. Lipopolysaccharide heterogeneity: Innate host responses to bacterial modification of lipid A structure. Journal of Dental Research 2005; 84(7), 584-595.
J Jo, A Price-Whelan and LE Dietrich. Gradients and consequences of heterogeneity in biofilms. Nature Reviews Microbiology 2022; 20(10), 593-607.
SN Abraham, N Sharon and I Ofek. Adhesion and colonisation. Molecular Medical Microbiology 2002; 1, 629-644.
KY Le and M Otto. Quorum-sensing regulation in staphylococci-an overview. Frontiers in Microbiology 2015; 6, 1174.
TJ Polaske, KH West, K Zhao, DL Widner, JT York and HE Blackwell. Chemical and biomolecular insights into the Staphylococcus aureus agr quorum sensing system: current progress and ongoing challenges. Israel Journal of Chemistry 2023; 63(5-6), e202200096.
T Abee, Á T Kovács, OP Kuipers and S Van der Veen. Biofilm formation and dispersal in Gram-positive bacteria. Current Opinion in Biotechnology 2011; 22(2), 172-179.
L Zhou, Y Zhang, Y Ge, X Zhu and J Pan. Regulatory mechanisms and promising applications of quorum sensing-inhibiting agents in control of bacterial biofilm formation. Frontiers in Microbiology 2020; 11, 589640.
MJ Franklin, DE Nivens, JT Weadge and PL Howell. Biosynthesis of the Pseudomonas aeruginosa extracellular polysaccharides, alginate, Pel, and Psl. Frontiers in Microbiology 2011; 2, 167.
A Hartmann. Quorum sensing N-acyl-homoserine lactone signal molecules of plant beneficial Gram-negative rhizobacteria support plant growth and resistance to pathogens. Rhizosphere 2020; 16, 100258.
Z Wang, X Hou, G Shang, G Deng, K Luo and M Peng. Exploring fatty acid β-oxidation pathways in bacteria: from general mechanisms to DSF signaling and pathogenicity in Xanthomonas. Current Microbiology 2024; 81(10), 336.
YW He, Y Deng, Y Miao, S Chatterjee, TM Tran, J Tian and S Lindow. DSF-family quorum sensing signal-mediated intraspecies, interspecies, and inter-kingdom communication. Trends in Microbiology 2023; 31(1), 36-50.
M Gallique, M Bouteiller and A Merieau. The type VI secretion system: A dynamic system for bacterial communication? Frontiers in Microbiology 2017; 8, 1454.
A Vetrivel, M Ramasamy, P Vetrivel, S Natchimuthu, S Arunachalam, GS Kim and R Murugesan. Pseudomonas aeruginosa biofilm formation and its control. Biologics 2021; 1(3), 312-336.
Z Pang, R Raudonis, BR Glick, TJ Lin and Z Cheng. Antibiotic resistance in Pseudomonas aeruginosa: mechanisms and alternative therapeutic strategies. Biotechnology Advances 2019; 37(1), 177-192.
HC Neu. The role of Pseudomonas aeruginosa in infections. Journal of Antimicrobial Chemotherapy 1983; 11(SB), 1-3.
A Nasser, MM Dallal, S Jahanbakhshi, T Azimi and L Nikouei. Staphylococcus aureus: Biofilm formation and strategies against it. Current Pharmaceutical Biotechnology 2022; 23(5), 664-678.
A Pantosti, A Sanchini and M Monaco. Mechanisms of antibiotic resistance in Staphylococcus aureus. Future Microbiology 2007; 2(3), 323-334.
MZ David and RS Daum. Treatment of Staphylococcus aureus infections. In: MZ David and RS Daum (Eds.). Staphylococcus aureus: Microbiology, pathology, immunology, therapy and prophylaxis. Springer, Cham, Swizerland, 2017, p. 325-383.
G Laverty, SP Gorman and BF Gilmore. Biomolecular mechanisms of Pseudomonas aeruginosa and Escherichia coli biofilm formation. Pathogens 2014; 3(3), 596-632.
A Ito, A Taniuchi, T May, K Kawata and S Okabe. Increased antibiotic resistance of Escherichia coli in mature biofilms. Applied and Environmental Microbiology 2009; 75(12), 4093-4100.
TA Russo and JR Johnson. Medical and economic impact of extraintestinal infections due to Escherichia coli: focus on an increasingly important endemic problem. Microbes and Infection 2003; 5(5), 449-456.
D Senadheera and DG Cvitkovitch. Quorum sensing and biofilm formation by Streptococcus mutans. In: Bacterial signal transduction: Networks and drug targets. Springer, New York, 2008, p. 178-188.
J Nan, E Brostromer, XY Liu, O Kristensen and XD Su. Bioinformatics and structural characterization of a hypothetical protein from Streptococcus mutans: implication of antibiotic resistance. PLoS One 2009; 4(10), e7245.
SM Daboor, FS Masood, MS Al-Azab and EE Nori. A review on Streptococcus mutans with its diseases dental caries, dental plaque and endocarditis. Indian Journal of Microbiology Research 2015; 2(2), 76-82.
T Atriwal, K Azeem, FM Husain, A Hussain, MN Khan, MF Alajmi and M Abid. Mechanistic understanding of Candida albicans biofilm formation and approaches for its inhibition. Frontiers in Microbiology 2021; 12, 638609.
LE Cowen, JB Anderson and LM Kohn. Evolution of drug resistance in Candida albicans. Annual Review of Microbiology 2002; 56(1), 139-165.
CJ Nobile and AD Johnson. Candida albicans biofilms and human disease. Annual Review of Microbiology 2015; 69(1), 71-92.
JA Gaddy and LA Actis. Regulation of Acinetobacter baumannii biofilm formation. Future Microbiology 2009; 4(3), 273-278.
I Kyriakidis, E Vasileiou, ZD Pana and A Tragiannidis. Acinetobacter baumannii antibiotic resistance mechanisms. Pathogens 2021; 10(3), 373.
A Gedefie, W Demsis, M Ashagrie, Y Kassa, M Tesfaye, M Tilahun, H Bisetegn and Z Sahle. Acinetobacter baumannii biofilm formation and its role in disease pathogenesis: A review. Infection and Drug Resistance 2021; 10, 3711-3719.
C Vuotto, F Longo, MP Balice, G Donelli and PE Varaldo. Antibiotic resistance related to biofilm formation in Klebsiella pneumoniae. Pathogens 2014; 3(3), 743-758.
ME Guerra, G Destro, B Vieira, AS Lima, LF Ferraz, AP Hakansson, M Darrieux and TR Converso. Klebsiella pneumoniae biofilms and their role in disease pathogenesis. Frontiers in Cellular and Infection Microbiology 2022; 12, 877995.
A Toledo-Arana, J Valle, C Solano, MJ Arrizubieta, C Cucarella, M Lamata, B Amorena, J Leiva, JR Penadés and I Lasa. The enterococcal surface protein, Esp, is involved in Enterococcus faecalis biofilm formation. Applied and Environmental Microbiology 2001; 67(10), 4538-4545.
IU Rathnayake, M Hargreaves and F Huygens. Antibiotic resistance and virulence traits in clinical and environmental Enterococcus faecalis and Enterococcus faecium isolates. Systematic and Applied Microbiology 2012; 35(5), 326-333.
SB Bhardwaj. Enterococci: An important nosocomial pathogen. Pathogenic Bacteria 2019; 12, 1-15.
P Chakraborty, S Bajeli, D Kaushal, BD Radotra and A Kumar. Biofilm formation in the lung contributes to virulence and drug tolerance of Mycobacterium tuberculosis. Nature Communications 2021; 12(1), 1606.
CA Peloquin and SE Berning. Infection caused by Mycobacterium tuberculosis. Annals of Pharmacotherapy 1994; 28(1), 72-84.
H Yonezawa, T Osaki and S Kamiya. Biofilm formation by Helicobacter pylori and its involvement in antibiotic resistance. BioMed Research International 2015; 2015(1), 914791.
P Sipponen and H Hyvärinen. Role of Helicobacter pylori in the pathogenesis of gastritis, peptic ulcer and gastric cancer. Scandinavian Journal of Gastroenterology 1993; 28(S196), 3-6.
SM Jacobsen and ME Shirtliff. Proteus mirabilis biofilms and catheter-associated urinary tract infections. Virulence 2011; 2(5), 460-465.
E Alqurashi, K Elbanna, I Ahmad and HH Abulreesh. Antibiotic resistance in Proteus mirabilis: mechanism, status, and public health significance. Journal of Pure and Applied Microbiology 2022; 16(3), 1191-1203.
JM Kopaczyk, J Warguła and T Jelonek. The variability of terpenes in conifers under developmental and environmental stimuli. Environmental and Experimental Botany 2020; 180, 104197.
FD Mabou and IB Yossa. Terpenes: Structural classification and biological activities. IOSR Journal of Pharmacy and Biological Sciences 2021; 16, 25-40.
K Wijesekera and AS Dissanayake. Terpenes. In: M Napagoda and L Jayasinghe (Eds.). Chemistry of natural products: Phytochemistry and pharmacognosy of medicinal plants. De Gruyter, Berlin, Germany, 2022, p. 65-90.
H Loza-Tavera. Monoterpenes in essential oils: biosynthesis and properties. In: F Shahidi, P Kolodziejczyk, JR Whitaker, AL Munguia and G Fuller (Eds.). Chemicals via higher plant bioengineering. Advances in experimental medicine and biology. Springer, Boston, United States, 1999, p. 49-62.
F de Faveri Favero, R Grando, FR Nonato, IM Sousa, NC Queiroz, GB Longato, RR Zafred, JE Carvalho, HM Spindola and MA Foglio. Artemisia annua L.: evidence of sesquiterpene lactones’ fraction antinociceptive activity. BMC Complementary and Alternative Medicine 2014; 14(1), 1-10.
ANM Alamgir. Secondary metabolites: Secondary metabolic products consisting of C and H; C, H, and O; N, S, and P elements; and O/N heterocycles. In: ANM Alamgir (Ed.). Therapeutic use of medicinal plants and their extracts, volume 2: Phytochemistry and bioactive compounds. Springer, Cham, Switzerland, 2018, p. 165-309.
JM DeLong and RK Prange. Taxus spp.: Botany, horticulture, and source of anti-cancer compounds. Horticultural Reviews 2006; 32, 299-327.
S Banerjee, S Banerjee, A Bishayee, MN Da Silva, OA Sukocheva, E Tse, N Casarcia and A Bishayee. Cellular and molecular mechanisms underlying the potential of betulinic acid in cancer prevention and treatment. Phytomedicine 2024; 132, 155858.
E Gambino, A Maione, M Guida, L Albarano, F Carraturo, E Galdiero and V Di Onofrio. Evaluation of the pathogenic mixed biofilm formation of Pseudomonas aeruginosa/Staphylococcus aureus and treatment with limonene on 3 different materials by a dynamic model. International Journal of Environmental Research and Public Health 2022; 19(6), 3741.
V Poscente, L Di Gregorio, M Costanzo, C Nobili, R Bernini, L Garavaglia and A Bevivino. Lactiplantibacillus plantarum monolayer enhanced bactericidal action of carvacrol: Biofilm inhibition of viable foodborne pathogens and spoilage microorganisms. Frontiers in Microbiology 2023; 14, 1296608.
S Asadi, B Nayeri-Fasaei, T Zahraei-Salehi, R Yahya-Rayat, N Shams and A Sharifi. Antibacterial and anti-biofilm properties of carvacrol alone and in combination with cefixime against Escherichia coli. BMC Microbiology 2023; 23(1), 55.
SY Wei, YL Li, L Wang, ZY Chu, YC Qin and H Zeng. α-Pinene: inhibitor of Acinetobacter baumannii biofilms and potential therapeutic agent for pneumonia. International Immunopharmacology 2025; 151, 114287.
P Jurado, C Uruén, S Martínez, E Lain, S Sánchez, A Rezusta and J Arenas. Essential oils of Pinus sylvestris, Citrus limon and Origanum vulgare exhibit high bactericidal and anti-biofilm activities against Neisseria gonorrhoeae and Streptococcus suis. Biomedicine & Pharmacotherapy 2023; 168, 115703.
HJ Yoo and SK Jwa. Inhibitory effects of β-caryophyllene on Streptococcus mutans biofilm. Archives of Oral Biology 2018; 88, 42-46.
K Gu, P Ouyang, Y Hong, Y Dai, T Tang, C He, G Shu, X Liang, H Tang, L Zhu and Z Xu. Geraniol inhibits biofilm formation of methicillin-resistant Staphylococcus aureus and increases the therapeutic effect of vancomycin in vivo. Frontiers in Microbiology 2022; 13, 960728.
A Kannappan, M Sivaranjani, R Srinivasan, J Rathna, SK Pandian and AV Ravi. Inhibitory efficacy of geraniol on biofilm formation and development of adaptive resistance in Staphylococcus epidermidis RP62A. Journal of Medical Microbiology 2017; 66(10), 1506-1515.
P K Sagar, P Sharma and R Singh. Inhibition of quorum sensing-regulated virulence factors and biofilm formation by Eucalyptus globulus against multidrug-resistant Pseudomonas aeruginosa. Journal of Pharmacopuncture 2022; 25(1), 37-45.
K Vijayakumar, V Manigandan, D Jeyapragash, V Bharathidasan, B Anandharaj and M Sathya. Eucalyptol inhibits biofilm formation of Streptococcus pyogenes and its mediated virulence factors. Journal of Medical Microbiology 2020; 69(11), 1308-1318.
T Liu, J Kang and L Liu. Thymol as a critical component of Thymus vulgaris L. essential oil combats Pseudomonas aeruginosa by intercalating DNA and inactivating biofilm. LWT 2021; 136, 110354.
ST Khan, M Khan, J Ahmad, R Wahab, OH Abd-Elkader, J Musarrat, HZ Alkhathlan and AA Al-Kedhairy. Thymol and carvacrol induce autolysis, stress, growth inhibition and reduce the biofilm formation by Streptococcus mutans. AMB Express 2017; 7(1), 1-10.
P Gupta, M Sharma, N Arora, V Pruthi and KM Poluri. Chemistry and biology of farnesol and its derivatives: quorum sensing molecules with immense therapeutic potential. Current Topics in Medicinal Chemistry 2018; 18(22), 1937-1954.
B Gaálová-Radochová, S Kendra, L Jordao, L Kursawe, J Kikhney, A Moter and H Bujdáková. Effect of quorum sensing molecule farnesol on mixed biofilms of Candida albicans and Staphylococcus aureus. Antibiotics 2023; 12(3), 441.
L Wang, J Wang, K Zhang, J Zhang, D Cui, J Wang, P Ji, Y Wei and J Li. Linalool as a potential agent for inhibiting Escherichia coli biofilm formation and exopolysaccharide production. BMC Veterinary Research 2025; 21(1), 235.
G Shen, L Yang, X Lv, Y Zhang, X Hou, M Li, M Zhou, L Pan, A Chen and Z Zhang. Antibiofilm activity and mechanism of linalool against food spoilage Bacillus amyloliquefaciens. International Journal of Molecular Sciences 2023; 24(13), 10980.
PM Maquera-Huacho, CC Tonon, MF Correia, RS Francisconi, EA Bordini, É Marcantonio and DM Spolidorio. In vitro antibacterial and cytotoxic activities of carvacrol and terpinen-4-ol against biofilm formation on titanium implant surfaces. Biofouling 2018; 34(6), 699-709.
L Cordeiro, P Figueiredo, H Souza, A Sousa, F Andrade-Júnior, D Medeiros, J Nóbrega, D Silva, E Martins, J Barbosa-Filho and E Lima. Terpinen-4-ol as an antibacterial and antibiofilm agent against Staphylococcus aureus. International Journal of Molecular Sciences 2020; 21(12), 4531.
D Cox-Georgian, N Ramadoss, C Dona and C Basu. Therapeutic and medicinal uses of terpenes. In: N Joshee, S Dhekney and P Parajuli (Eds.). Medicinal plants: From farm to pharmacy. Springer, Cham, Switzerland, 2019, p. 333-359.
D Dufour, V Leung and CM Lévesque. Bacterial biofilm: Structure, function, and antimicrobial resistance. Endodontic Topics 2010; 22(1), 2-16.
TB Farnan, J McCallum, A Awa, AD Khan and SJ Hall. Tea tree oil: In vitro efficacy in otitis externa. The Journal of Laryngology & Otology 2005; 119(3), 198-201.
R Iseppi, M Mariani, S Benvenuti, E Truzzi and P Messi. Effects of Melaleuca alternifolia Chell (Tea Tree) and Eucalyptus globulus Labill. essential oils on antibiotic-resistant bacterial biofilms. Molecules 2023; 28(4), 1671.
C Brinsi, A Abidi, K Hosni, H Tounsi and H Sebai. Protective effect of Eucalyptus globulus extracts against bleomycin-induced pulmonary fibrosis in rats. Journal of Medicinal Food 2022; 25(7), 741–750.
B Saoudi, K Bariz, S Saci, Y Belounis, H Ait Issad, M Abbaci, M A Mustapha, E H Nabti, R Alenazy, M S Alhussaini and A A Alyahya. Enhancing antibiotic efficacy and combating biofilm formation: evaluating the synergistic potential of Origanum vulgare essential oil against multidrug-resistant Gram-negative bacteria. Microorganisms 2024; 12(8), 1651.
C Rossi, C Chaves-López, A Serio, F Anniballi, L Valbonetti and A Paparella. Effect of Origanum vulgare essential oil on biofilm formation and motility capacity of Pseudomonas fluorescens strains isolated from discoloured mozzarella cheese. Journal of Applied Microbiology 2018; 124(5), 1220-1231.
S Kajjari, BK Vanishree, S Janardhanan, VH Patil, C Uppin and SM Hugar. Antimicrobial efficacy of Mangifera indica, Mentha arvensis, and chlorhexidine mouthwashes on Streptococcus mutans and Candida albicans in children: A comparative in vivo study. International Journal of Clinical Pediatric Dentistry 2024; 17(S1), S78.
R Karicheri and B Antony. Antibacterial and antibiofilm activities of peppermint (Mentha piperita Linn) and menthol mint (Mentha arvensis Linn) essential oils on Aggregatibacter actinomycetemcomitans isolated from orodental infections. European Journal of Pharmaceutical and Medical Research 2016; 3, 577-581.
SO Egwuatu, TM Obuotor, OS Taiwo, WE Ike, AE Ojo, PO Isibor, OF Adeniji, FM Oyeyipo, OA Awotoye, P Akinduti and P Egwuatu. Antibacterial efficacy of Thymus vulgaris essential oil against extended-spectrum beta-lactamase-producing Escherichia coli in urinary tract infections. In: Biotechnological Approaches to Sustainable Development Goals. Cham: Springer Nature Switzerland; 2023. p. 159–172.
TW Kang, G Chandrasekaran, EC Hwang, HS Kim and VK Lakshmanan. Characterization and antibacterial activity of PVA-PVP-CS carvacrol-loaded polymer composite films for urinary catheter. International Journal of Polymeric Materials and Polymeric Biomaterials 2018; 67(17), 1016-1027.
R De, P Kundu, S Swarnakar, T Ramamurthy, A Chowdhury, GB Nair and AK Mukhopadhyay. Antimicrobial activity of curcumin against Helicobacter pylori isolates from India and during infections in mice. Antimicrobial Agents and Chemotherapy 2009; 53(4), 1592-1597.
UJ Tsopmene, CRT Kuaté, PN Kayoka-Kabongo, BN Bisso, A Metopa, CT Mofor and JP Dzoyem. Antibiofilm activity of curcumin and piperine and their synergistic effects with antifungals against Candida albicans clinical isolates. Scientifica 2024; 2024(1), 2025557.
A Jerkovic-Mujkic, K Masic and TJ Dzelilovic. Antimicrobial potential of herbal products against Clostridioides difficile. Health 2024; 4, 39-49.
S Justin and B Antony. Antibacterial activity of the essential oils of Syzygium aromaticum (L.) Merr. Perry (Clove), Myristica fragrans Houtt. (Nutmeg) and Zingiber officinale Roscoe (Ginger) against clinical isolates of Clostridium difficile: An in vitro study. International Journal of Contemporary Medical Research 2016; 3, 1085-1089.
R Najafi-Taher, AJ Kohneloo, VE Farsani, NM Rayeni, HR Moghimi, A Ehsani and A Amani. A topical gel of tea tree oil nanoemulsion containing adapalene versus adapalene marketed gel in patients with acne vulgaris: A randomized clinical trial. Archives of Dermatological Research 2022; 314(9), 1-7.
D Lauritano, D Pazzi, A Iapichino, RM Gaudio, MD Muzio, LL Russo and F Pezzetti. Evaluation of the efficacy of a new oral gel containing carvacrol and thymol for home oral care in the management of chronic periodontitis using pcr analysis: A microbiological pilot study. Journal of Biological Regulators & Homeostatic Agents 2016; 30(2S1), 129-134.
VC Patole, SP Chaudhari, AP Pandit and PP Lokhande. Thymol and eugenol loaded chitosan dental film for treatment of periodontitis. Indian Drugs 2019; 56(6), 51-58.
H Hamzah, S U Pratiwi and T Hertiani. Efficacy of thymol and eugenol against polymicrobial biofilm. Indonesian Journal of Pharmacy 2018; 29(4), 214-221.
W Mączka, M Twardawska, M Grabarczyk and K Wińska. Carvacrol - a natural phenolic compound with antimicrobial properties. Antibiotics 2023; 12(5), 824.
F Murina, F Vicariotto and S Di Francesco. Thymol, eugenol and lactobacilli in a medical device for the treatment of bacterial vaginosis and vulvovaginal candidiasis. New Microbiologica 2018; 41(3), 220-224.
C Liu, Y Zhang, S Kong, I Tsui, Y Yu and F Han. Applications and therapeutic actions of complementary and alternative medicine for women with genital infection. Evidence-Based Complementary and Alternative Medicine 2014; 2014(1), 658624.
AC Guimarães, LM Meireles, MF Lemos, MC Guimarães, DC Endringer, M Fronza and R Scherer. Antibacterial activity of terpenes and terpenoids present in essential oils. Molecules 2019; 24(13), 2471.
KJ Dias, GM Miranda, JR Bessa, AC Araújo, PR Freitas, RS Almeida, CL Paulo, J B Neto, HD Coutinho and J Ribeiro-Filho. Terpenes as bacterial efflux pump inhibitors: a systematic review. Frontiers in Pharmacology 2022; 13, 953982.
S Burt. Essential oils: Their antibacterial properties and potential applications in foods - a review. International Journal of Food Microbiology 2004; 94(3), 223-253.
A Masyita, RM Sari, AD Astuti, B Yasir, NR Rumata, TB Emran, F Nainu and J Simal-Gandara. Terpenes and terpenoids as main bioactive compounds of essential oils, their roles in human health and potential application as natural food preservatives. Food Chemistry: X 2022; 13, 100217.
X Jin, M Zhang, J Lu, X Duan, J Chen, Y Liu, W Chang and H Lou. Hinokitiol chelates intracellular iron to retard fungal growth by disturbing mitochondrial respiration. Journal of Advanced Research 2021; 34, 65-77.
M Mikulášová, R Chovanová and Š Vaverková. Synergism between antibiotics and plant extracts or essential oils with efflux pump inhibitory activity in coping with multidrug-resistant staphylococci. Phytochemistry Reviews 2016; 15, 651-662.
W Wang, X Bao, M Bové, P Rigole, X Meng, J Su and T Coenye. Antibiofilm activities of borneol-citral-loaded Pickering emulsions against Pseudomonas aeruginosa and Staphylococcus aureus in physiologically relevant chronic infection models. Microbiology Spectrum 2022; 10(5), e01696-22.
L Gutiérrez, A Escudero, R Batlle and C Nerín. Effect of mixed antimicrobial agents and flavors in active packaging films. Journal of Agricultural and Food Chemistry 2009; 57(18), 8564-8571.
C Gan, E Langa, A Valenzuela, D Ballestero and MR Pino-Otín. Synergistic activity of thymol with commercial antibiotics against critical and high WHO priority pathogenic bacteria. Plants 2023; 12(9), 1868.
B Huerta Lorenzo, Á Galán-Relaño, E Barba-Sánchez, A Romero-Salmoral, AL Solarte Portilla, L Gómez-Gascón and RJ Astorga Márquez. Potentiation of the antimicrobial effect of oxytetracycline combined with cinnamon, clove, oregano, and red thyme essential oils against MDR Salmonella enterica strains. Animals 2024; 14(9), 1347.
H Si, J Hu, Z Liu and ZL Zeng. Antibacterial effect of oregano essential oil alone and in combination with antibiotics against extended-spectrum β-lactamase-producing Escherichia coli. FEMS Immunology & Medical Microbiology 2008; 53(2), 190-194.
F Fratini, C Pecorini, I Resci, E Copelotti, FP Nocera, B Najar and S Mancini. Evaluation of the synergistic antimicrobial activity of essential oils and cecropin A natural peptide on Gram-negative bacteria. Animals 2025; 15(2), 282.
PS Costa, SS Oliveira, EB Souza, EH Brito, CS Cavalcante, SM Morais, AL Leal, HM Barreto, AM Teixeira, CE Nogueira and RO Fontenelle. Antifungal activity and synergistic effect of essential oil from Lippia alba against Trichophyton rubrum and Candida spp. Revista Virtual de Química 2020; 12(6), 0000-0000.
D C Costa, A B Vermelho, C A Almeida, E P Dias, S M Cedrola, M D Arrigoni-Blank, A F Blank, C S Alviano and D S Alviano. Inhibitory effect of linalool-rich essential oil from Lippia alba on the peptidase and keratinase activities of dermatophytes. Journal of Enzyme Inhibition and Medicinal Chemistry 2014; 29(1), 12-17.
J Reichling. Anti-biofilm and virulence factor-reducing activities of essential oils and oil components as a possible option for bacterial infection control. Planta Medica 2020; 86(8), 520-537.
Y Ge, J Tang, H Fu and Y Fu. Terpinen-4-ol liposomes-incorporated chitosan/polyethylene oxide electrospun nanofibrous film ameliorates the external microenvironment of healing cutaneous wounds. Journal of Applied Polymer Science 2021; 138(2), 49670.
JI Deen, AS Zawad, M Uddin, MA Chowdhury, SQ Al Araby and MA Rahman. Terpinen-4-ol, a volatile terpene molecule, extensively electrifies biological systems against oxidative stress-linked pathogenesis. Advances in Redox Research 2023; 9, 100082.
T Kennewell, S Mashtoub, GS Howarth, AJ Cowin and Z Kopecki. Antimicrobial and healing-promoting properties of animal and plant oils for the treatment of infected wounds. Wound Practice & Research: Journal of the Australian Wound Management Association 2019; 27(4), 175-183.
RG Frykberg and J Banks. Challenges in the treatment of chronic wounds. Advances in Wound Care 2015; 4(9), 560-582.
A Scalise, A Bianchi, C Tartaglione, E Bolletta, M Pierangeli, M Torresetti, M Marazzi and GI Di Benedetto. Microenvironment and microbiology of skin wounds: the role of bacterial biofilms and related factors. Seminars in Vascular Surgery 2015; 28(3-4), 151-159.
JS Boateng, KH Matthews, HN Stevens and GM Eccleston. Wound healing dressings and drug delivery systems: A review. Journal of Pharmaceutical Sciences 2008; 97(8), 2892-2923.
G Gainza, S Villullas, JL Pedraz, RM Hernandez and M Igartua. Advances in drug delivery systems to release growth factors for wound healing and skin regeneration. Nanomedicine: Nanotechnology, Biology and Medicine 2015; 11(6), 1551-1573.
A Sultana, M Zare, V Thomas, TS Kumar and S Ramakrishna. Nano-based drug delivery systems: Conventional routes, recent developments, and future prospects. Medicine in Drug Discovery 2022; 15, 100134.
H Bhardwaj, S Khute, R Sahu and RK Jangde. Advanced drug delivery systems for management of chronic diabetic wound healing. Current Drug Targets 2023; 24(16), 1239-1259.
L Pachuau. Recent developments in novel drug delivery systems for wound healing. Expert Opinion on Drug Delivery 2015; 12(12), 1895-1909.
J Jose, KB Khot, SP Thomas, H Chopra, G Gopan, A Bandiwadekar, A Babu, SR Ugare, GM Ashraf, A Dhyani and V Vora. Advances in microneedles-based drug delivery systems on promoting wound healing. Journal of Drug Delivery Science and Technology 2023; 90, 105163.
Y Li, J Wang, Y Wang and W Cui. Advanced electrospun hydrogel fibers for wound healing. Composites Part B: Engineering 2021; 223, 109101.
M Sufiyan, P Kushwaha, M Ahmad, P Mandal and KK Vishwakarma. Scaffold-mediated drug delivery for enhanced wound healing: A review. AAPS PharmSciTech 2024; 25(5), 137.
J K Tee, CN Ong, BH Bay, HK Ho and DT Leong. Oxidative stress by inorganic nanoparticles. Wiley Interdisciplinary Reviews: Nanomedicine and Nanobiotechnology 2016; 8(3), 414-438.
SM Häffner and M Malmsten. Interplay between amphiphilic peptides and nanoparticles for selective membrane destabilization and antimicrobial effects. Current Opinion in Colloid & Interface Science 2019; 44, 59-71.
AK Tiwari, PC Pandey, MK Gupta and RJ Narayan. Nano–bio interaction and antibacterial mechanism of engineered metal nanoparticles: fundamentals and current understanding. Journal of Cluster Science 2025; 36(1), 1-33.
A R Bilia, V Piazzini, L Risaliti, G Vanti, M Casamonti, M Wang and M C Bergonzi. Nanocarriers: a successful tool to increase solubility, stability and optimize bioefficacy of natural constituents. Current Medicinal Chemistry 2019; 26(24), 4631-4656.
D Yadav, K Sandeep, D Pandey and RK Dutta. Liposomes for drug delivery. Journal of Biotechnology & Biomaterials 2017; 7(4), 276.
M Saffari, FH Shirazi and HR Moghimi. Terpene-loaded liposomes and isopropyl myristate as chemical permeation enhancers toward liposomal gene delivery in lung cancer cells: a comparative study. Iranian Journal of Pharmaceutical Research 2016; 15(3), 261.
B Kumar, M Pandey, R Aggarwal and PK Sahoo. A comprehensive review on invasomal carriers incorporating natural terpenes for augmented transdermal delivery. Future Journal of Pharmaceutical Sciences 2022; 8(1), 50.
AC Paiva-Santos, AL Silva, C Guerra, D Peixoto, M Pereira-Silva, M Zeinali, F Mascarenhas-Melo, R Castro and F Veiga. Ethosomes as nanocarriers for the development of skin delivery formulations. Pharmaceutical Research 2021; 38(6), 947-970.
EM Elmowafy, M Tiboni and ME Soliman. Biocompatibility, biodegradation and biomedical applications of poly(lactic acid)/poly(lactic-co-glycolic acid) micro and nanoparticles. Journal of Pharmaceutical Investigation 2019; 49, 347-380.
MM El-Hammadi, AL Small-Howard, M Fernández-Arévalo and L Martín-Banderas. Development of enhanced drug delivery vehicles for 3 cannabis-based terpenes using poly(lactic-co-glycolic acid)-based nanoparticles. Industrial Crops and Products 2021; 164, 113345.
P Patel, N Vyas and M Raval. Safety and toxicity issues of polymeric nanoparticles: A serious concern. In: M Rai, M Patel and R Patel (Eds.). Nanotechnology in medicine: Toxicity and safety. Wiley, New Jersey, United States, 2021, p. 156-173.
E Lasoń. Topical administration of terpenes encapsulated in nanostructured lipid-based systems. Molecules 2020; 25(23), 5758.
R H Müller, K Mäder and S Gohla. Solid lipid nanoparticles (SLN) for controlled drug delivery - a review of the state of the art. European Journal of Pharmaceutics and Biopharmaceutics 2000; 50(1), 161-177.
SV Khairnar, P Pagare, A Thakre, AR Nambiar, V Junnuthula, MC Abraham, P Kolimi, D Nyavanandi and S Dyawanapelly. Review on the scale-up methods for the preparation of solid lipid nanoparticles. Pharmaceutics 2022; 14(9), 1886.
M Kumari and D K Nanda. Potential of curcumin nanoemulsion as antimicrobial and wound healing agent in burn wound infection. Burns 2023; 49(5), 1003-1016.
CR Garcia, MH Malik, S Biswas, VH Tam, KP Rumbaugh, W Li and X Liu. Nanoemulsion delivery systems for enhanced efficacy of antimicrobials and essential oils. Biomaterials Science 2022; 10(3), 633-653.
TN Barradas and KG de Holanda e Silva. Nanoemulsions of essential oils to improve solubility, stability and permeability: A review. Environmental Chemistry Letters 2021; 19(2), 1153-1171.
A Nabawy, JM Makabenta, S Schmidt-Malan, J Park, CH Li, R Huang, S Fedeli, AN Chattopadhyay, R Patel and VM Rotello. Dual antimicrobial-loaded biodegradable nanoemulsions for synergistic treatment of wound biofilms. Journal of Controlled Release 2022; 347, 379-388.
L Li, Y Xie, J Wang, Q Sun, M Gao and C Li. Biofilm microenvironment-activated multimodal therapy nanoplatform for effective antibacterial treatment and wound healing. Acta Biomaterialia 2024; 183, 221-234.
S Fahimirad, F Fattahi, M Hatami and S Shabani. Nanotechnology-based biotherapeutics for physiological wound healing phases. Industrial Crops and Products 2025; 226, 120608.
SS Garg, R Dubey, S Sharma, A Vyas and J Gupta. Biological macromolecule-based nanoformulations for improving wound healing and bacterial biofilm-associated infection: A review. International Journal of Biological Macromolecules 2023; 247, 125636.
M Li, X He, R Zhao, Q Shi, Y Nian and B Hu. Hydrogels as promising carriers for the delivery of food bioactive ingredients. Frontiers in Nutrition 2022; 9, 1006520.
R Ajdary, BL Tardy, BD Mattos, L Bai and O J Rojas. Plant nanomaterials and inspiration from nature: Water interactions and hierarchically structured hydrogels. Advanced Materials 2021; 33(28), 2001085.
M Zagórska-Dziok, A Nowak, A Zgadzaj, E Oledzka, K Kędra, A E Wiącek and M Sobczak. New polymeric hydrogels with cannabidiol and α-terpineol as potential materials for skin regeneration synthesis and physicochemical and biological characterization. International Journal of Molecular Sciences 2024; 25(11), 5934.
I Negut, G Dorcioman and V Grumezescu. Scaffolds for wound healing applications. Polymers 2020; 12(9), 2010.
S Stratton, NB Shelke, K Hoshino, S Rudraiah and SG Kumbar. Bioactive polymeric scaffolds for tissue engineering. Bioactive Materials 2016; 1(2), 93-108.
WJ Duncan. Establishment and evolution of the Otago sheep animal models: 40 years of periodontal, implant and bone regeneration research. New Zealand Dental Journal 2024; 120(4), 149-161.
H Haidari, R Bright, Y Yu, K Vasilev and Z Kopecki. Development of microneedles for antimicrobial drug delivery: A comprehensive review on applications in wound infection management. Small Science 2024; 4(10), 2400158.
TS Alster and PM Graham. Microneedling: a review and practical guide. Dermatologic Surgery 2018; 44(3), 397-404.
L Sun, Z Zhang, Y Cao, Z Lu, Y Zhang, X He, Z Wang and R Pei. pH-responsive and on-demand hydrogen sulfide-releasing injectable hydrogel for wound healing promotion. Chemical Engineering Journal 2025; 509, 160904.
A Ullah, M Jang, H Khan, HJ Choi, S An, D Kim, YR Kim, UK Kim and GM Kim. Microneedle array with a pH-responsive polymer coating and its application in smart drug delivery for wound healing. Sensors and Actuators B: Chemical 2021; 345, 130441.
J Mei, D Xu, L Wang, L Kong, Q Liu and Q Li. Biofilm microenvironment-responsive self-assembly nanoreactors for all-stage biofilm-associated infection through bacterial cuproptosis-like death and macrophage re-rousing. Advanced Materials 2023; 35(36), e2303432.
D Kumar, S Pandey, J Shiekmydeen, M Kumar, S Chopra and A Bhatia. Therapeutic potential of microneedle-assisted drug delivery for wound healing: Current state of the art, challenges, and future perspective. AAPS PharmSciTech 2025; 26(1), 25.
S Dalleau, E Cateau, T Bergès, JM Berjeaud and C Imbert. In vitro activity of terpenes against Candida biofilms. International Journal of Antimicrobial Agents 2008; 31(6), 572-576.
P Kumari, N Arora, A Chatrath, R Gangwar, V Pruthi, KM Poluri and R Prasad. Delineating the biofilm inhibition mechanisms of phenolic and aldehydic terpenes against Cryptococcus neoformans. ACS Omega 2019; 4(18), 17634-17648.
MA Chowdhury, M Ashrafudoulla, SI Mevo, AS Mahamud, CS Reem, AJ Ha and SD Ha. Efficacy of orange terpene against Escherichia coli biofilm on beef and food contact surfaces. Food Research International 2024; 197, 115204.
C Salinas, G Florentín, F Rodríguez, N Alvarenga and R Guillén. Terpenes combinations inhibit biofilm formation in Staphylococcus aureus by interfering with initial adhesion. Microorganisms 2022; 10(8), 1527.
PSS Lima, AM Lucchese, HG Araujo-Filho, PP Menezes, AAS Araujo, LJ Quintans-Junior and JSS Quintans. Inclusion of terpenes in cyclodextrins: preparation, characterization and pharmacological approaches. Carbohydrate Polymers 2016; 151, 965-987.
MAR Martins, LP Silva, O Ferreira, B Schröder, JAP Coutinho and SP Pinho. Terpenes solubility in water and their environmental distribution. Journal of Molecular Liquids 2017; 241, 996-1002.
B Sapra, S Jain and AK Tiwary. Percutaneous permeation enhancement by terpenes: Mechanistic view. AAPS Journal 2008; 10, 120-132.
F Nourbakhsh, M Lotfalizadeh, M Badpeyma, A Shakeri and V Soheili. From plants to antimicrobials: Natural products against bacterial membranes. Phytotherapy Research 2022; 36(1), 33-52.
AY Abramov, MV Zamaraeva, AI Hagelgans, RR Azimov and OV Krasilnikov. Influence of plant terpenoids on the permeability of mitochondria and lipid bilayers. Biochimica et Biophysica Acta (BBA) - Biomembranes 2001; 1512(1), 98-110.
J Szymczak, L Sobotta, J Dlugaszewska, M Kryjewski and J Mielcarek. Menthol-modified zinc(II) phthalocyanine regioisomers and their photoinduced antimicrobial activity against Staphylococcus aureus. Dyes and Pigments 2021; 193, 109410.
M Kossakowska-Zwierucho, G Szewczyk, T Sarna and J Nakonieczna. Farnesol potentiates photodynamic inactivation of Staphylococcus aureus with the use of red light-activated porphyrin TMPyP. Journal of Photochemistry and Photobiology B: Biology 2020; 206, 111863.
Y Li and M X Wu. Visualization and elimination of polymicrobial biofilms by a combination of ALA-carvacrol-blue light. Journal of Photochemistry and Photobiology B: Biology 2022; 234, 112525.
M Lu, Y Li and MX Wu. Bacteria-specific pro-photosensitizer kills multidrug-resistant Staphylococcus aureus and Pseudomonas aeruginosa. Communications Biology 2021; 4(1), 408.
M Lu, S Wang, T Wang, S Hu, B Bhayana, M Ishii, Y Kong, Y Cai, T Dai, W Cui and MX Wu. Bacteria-specific phototoxic reactions triggered by blue light and phytochemical carvacrol. Science Translational Medicine 2021; 13(575), eaba3571.
P Manrique, I Montero, M Fernandez-Gosende, N Martinez, CH Cantabrana and D Rios-Covian. Past, present, and future of microbiome-based therapies. Microbiome Research Reports 2024; 3(2), 23.
MB Habib, G Batool, NA Shah, T Muhammad, NS Akbar and A Shahid. Biofilm-mediated infections: Novel therapeutic approaches and harnessing artificial intelligence for early detection and treatment. Microbial Pathogenesis 2025; 203, 107497.
F Pennisi, A Pinto, G E Ricciardi, C Signorelli and V Gianfredi. Artificial intelligence in antimicrobial stewardship: A systematic review and meta-analysis of predictive performance and diagnostic accuracy. European Journal of Clinical Microbiology & Infectious Diseases 2025; 44(3), 463-513.
Natouri, A Barzegar, A Nobakht, A Bagheri, F Eslami, S Jafarirad, N Tohidifar and B Nikzad. Bioactive stem cell-laden 3D nanofibrous scaffolds for tissue engineering. Heliyon 2024; 10(19), e38462.
S Yamakawa and K Hayashida. Advances in surgical applications of growth factors for wound healing. Burns & Trauma 2019; 7, 1-9.
IA Jang, J Kim and W Park. Endogenous hydrogen peroxide increases biofilm formation by inducing exopolysaccharide production in Acinetobacter oleivorans DR1. Scientific Reports 2016; 6(1), 21121.
J Mei, D Xu, L Wang, L Kong, Q Liu, Q Li, X Zhang, Z Su, X Hu, W Zhu and M Ye. Biofilm microenvironment-responsive self-assembly nanoreactors for all-stage biofilm-associated infection through bacterial cuproptosis-like death and macrophage re-rousing. Advanced Materials 2023; 35(36), 2303432.
M Kim and J H Park. Isolation of Aloe saponaria-derived extracellular vesicles and investigation of their potential for chronic wound healing. Pharmaceutics 2022; 14(9), 1905.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






